Zonisamide
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
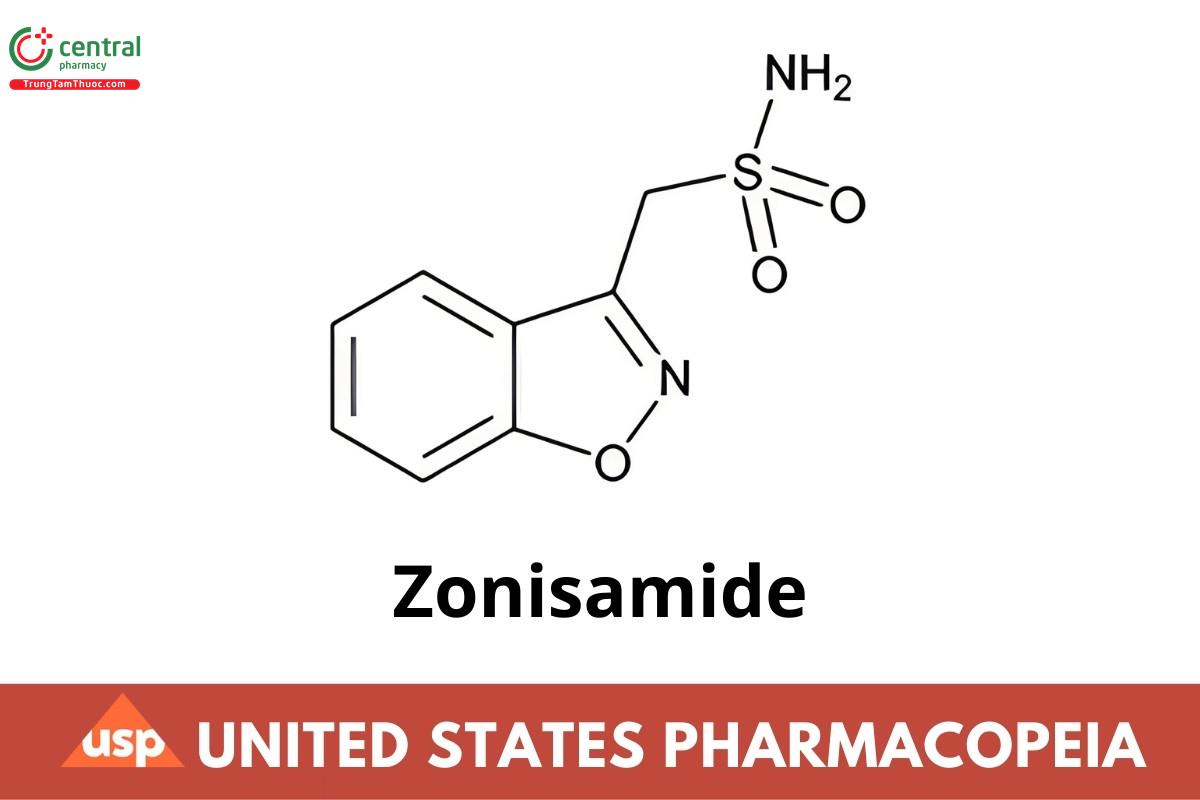
C8H8N2O3S 212.23
1,2-Benzisoxazole-3-methanesulfonamide CAS RN®: 68291-97-4; UNII: 459384H98V.
1 DEFINITION
Zonisamide contains NLT 98.0% and NMT 102.0% of C8H8N2O3S, calculated on the anhydrous basis.
2 IDENTIFICATION
Change to read:
A. Spectroscopic Identification Tests 〈197〉, Infrared Spectroscopy: 197K (ERR 1-Oct-2020)
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
Procedure
Buffer: 1.36 g/L of monobasic potassium phosphate in water. Adjust the pH to 3.0 ± 0.1 with 10% phosphoric acid.
Mobile phase: Acetonitrile, methanol, and Buffer (1:1:8)
Standard solution: 0.1 mg/mL of USP Zonisamide RS in Mobile phase
Sample solution: 0.1 mg/mL of Zonisamide in Mobile phase
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 240 nm
Column: 4.6-mm × 25-cm; 5-μm packing L1
Flow rate: 1.5 mL/min
Injection size: 20 μL
System suitability
Sample: Standard solution
Suitability requirements
Column efficiency: NLT 5000 theoretical plates
Tailing: NMT 1.8
Relative standard deviation: NMT 2.0%
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of C8H8N2O3S in the portion of Zonisamide taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of zonisamide from the Sample solution
rS = peak response of zonisamide from the Standard solution
CS = concentration of USP Zonisamide RS in the Standard solution (mg/mL)
CU = concentration of Zonisamide in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–102.0% on the anhydrous basis
4 IMPURITIES
Inorganic Impurities
Residue on Ignition 〈281〉: NMT 0.1%
Organic Impurities
Procedure
Mobile phase: Prepare as directed in the Assay.
Standard solution: 1 μg/mL of USP Zonisamide RS and 1.5 μg/mL of USP Zonisamide Related Compound A RS in Mobile phase
Sample solution: 1.0 mg/mL of Zonisamide in Mobile phase
Chromatographic system: Prepare as directed in the Assay.
Run time: 2 times the retention time of the zonisamide peak
System suitability
Sample: Standard solution
Suitability requirements
Resolution: NLT 10.0 between zonisamide related compound A and zonisamide
Relative standard deviation: NMT 10.0% for both zonisamide and zonisamide related compound A peaks
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zonisamide related compound A in the portion of Zonisamide taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × 100
rU = peak response of zonisamide related compound A from the Sample solution
rS = peak response of zonisamide related compound A from the Standard solution
CS = concentration of USP Zonisamide Related Compound A RS in the Standard solution (mg/mL)
CU = concentration of zonisamide in the Sample solution (mg/mL)
Mr1 = molecular weight of zonisamide related compound A (free acid), 213.23
Mr2 = molecular weight of USP Zonisamide Related Compound A RS (sodium salt), 235.23
Calculate the percentage of any unspecified impurity in the portion of Zonisamide taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of each impurity from the Sample solution
rS = peak response of zonisamide from the Standard solution
CS = concentration of USP Zonisamide RS in the Standard solution (mg/mL)
CU = concentration of Zonisamide in the Sample solution (mg/mL)
Acceptance criteria
Individual impurities: See Impurity Table 1.
Total impurities: NMT 0.3%
Impurity Table 1
| Name | Relative Retention Time | Acceptance Criteria, NMT (%) |
|---|---|---|
| Zonisamide related compound Aa | 0.51 | 0.15 |
| Zonisamide | 1.0 | — |
| Any unspecified individual impurity | — | 0.10 |
a 1,2-Benzisoxazole-3-methane sulfonic acid.
5 SPECIFIC TESTS
Water Determination, Method I 〈921〉: NMT 0.8%
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in well-closed containers, and store at room temperature.
USP Reference Standards 〈11〉
USP Zonisamide RS
USP Zonisamide Related Compound A RS
1,2-Benzisoxazole-3-methanesulfonic acid sodium salt.
C8H6NNaO4S 235.19 CAS RN®: CAS-73101-64-1.


