Zolpidem Tartrate
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
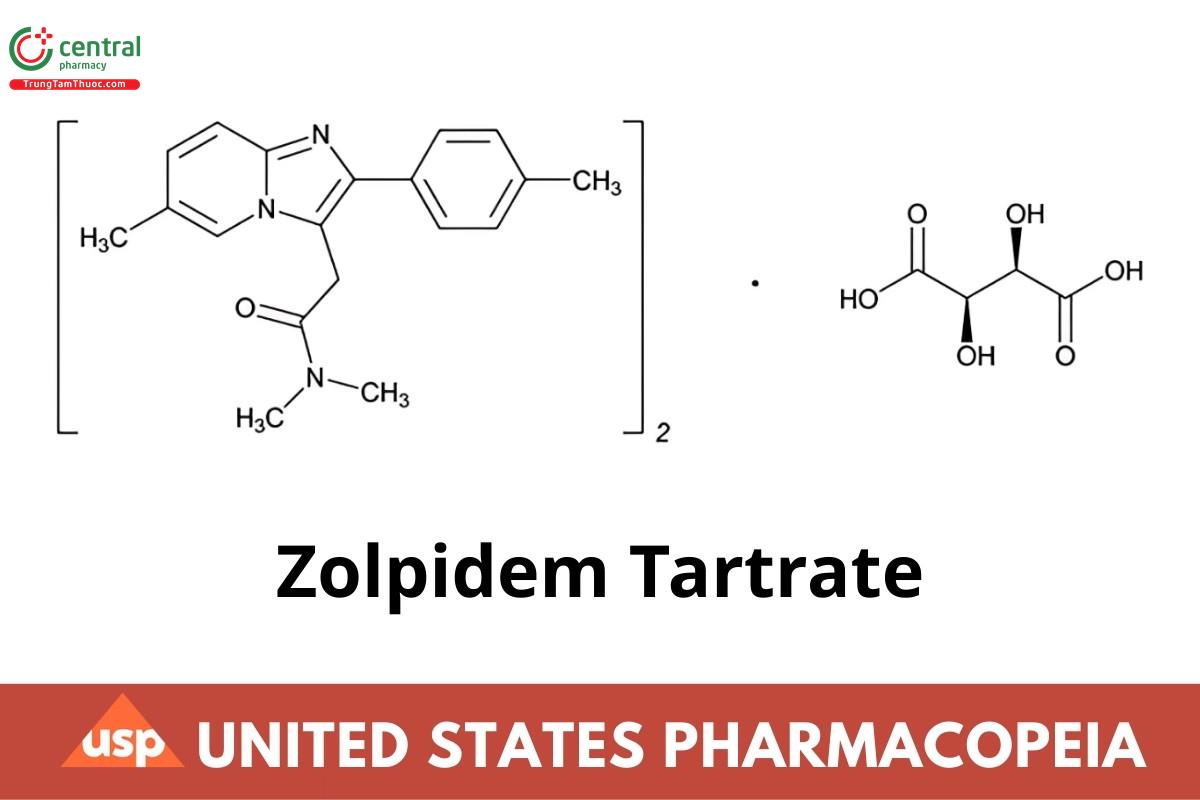
(C19H21N3O)2·C4H6O6 764.87
Imidazo[1,2-a]pyridine-3-acetamide, N,N,6-trimethyl-2-(4-methylphenyl)-, [R-(R*,R*)]-2,3-dihydroxy butanedioate;
N,N,6-Trimethyl-2-p-tolylimidazo[1,2-a]pyridine-3-acetamide L-(+)-tartrate CAS RN®: 99294-93-6; UNII: WY6W63843K.
1 DEFINITION
Zolpidem Tartrate contains NLT 98.0% and NMT 102.0% of zolpidem tartrate [(C19H21N3O)2·C4H6O6], calculated on the anhydrous basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS (197), Infrared Spectroscopy: 197A or 197K (CN 1-May-2020)
Sample: Dissolve 0.10 g of Zolpidem Tartrate in 10 mL of 0.1 M hydrochloric acid. Add 10 mL of water. Add dropwise with stirring 1 mL of diluted ammonia solution. Filter and collect the resulting precipitate. Wash the precipitate with water, and then dry at 100°-105° for 2 h. Use 2 mg of the dried residue.
Analysis: The IR spectrum of the free base thus obtained corresponds to the IR spectrum of similarly prepared USP Zolpidem Tartrate RS.
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
C. REACTION OF TARTRATES
Sample: 0.1 g of Zolpidem Tartrate
Analysis: Dissolve the Sample in 1 mL of methanol with gentle heating. To 0.1 mL of the solution, add 0.1 mL of a 100-g/L solution of potassium bromide, 0.1 mL of a 20-g/L solution of Resorcinol, and 3 mL of sulfuric acid. Heat on a water bath for 5-10 min. A dark blue color develops. Allow to cool, then pour the solution into water.
Acceptance criteria: The dark blue color changes to red.
3 ASSAY
3.1 PROCEDURE
Buffer: 5.6 g/L of phosphoric acid. Adjust with triethylamine to a pH of 5.5.
Mobile phase: Methanol, acetonitrile, and Buffer (23:18:59)
System suitability solution: 0.05 mg/mL each of USP Zolpidem Tartrate RS and USP Zolpidem Related Compound A RS in Mobile phase
Standard solution: 0.1 mg/mL of USP Zolpidem Tartrate RS in Mobile phase
Sample solution: 0.1 mg/mL of Zolpidem Tartrate in Mobile phase
3.2 Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 254 nm
Column: 3.9-mm x 15-cm; 4-µm packing L1
Flow rate: 1.5 mL/min
Injection volume: 20 µL
Run time: NLT 3 times the retention time of zolpidem
3.3 System suitability
Samples: System suitability solution and Standard solution
[NOTE-See Table 1 for the relative retention times.]
Suitability requirements
Resolution: NLT 2.0 between zolpidem related compound A and zolpidem, System suitability solution
Tailing factor: NMT 2.0, Standard solution
Relative standard deviation: NMT 1.0%, Standard solution
3.4 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zolpidem tartrate [(C19H21N3O)2·C4H6O6] in the portion of Zolpidem Tartrate taken:
Result = (rU/rS) x (CS/CU) x 100
rU = peak response of zolpidem from the Sample solution
rS = peak response of zolpidem from the Standard solution
CS = concentration of USP Zolpidem Tartrate RS in the Standard solution (mg/mL)
CU = concentration of Zolpidem Tartrate in the Sample solution (mg/mL)
Acceptance criteria: 98.0%-102.0% on the anhydrous basis
4 IMPURITIES
4.1 RESIDUE ON IGNITION (281)
NMT 0.1%
4.2 ORGANIC IMPURITIES
Buffer, Mobile phase, System suitability solution, and Chromatographic system: Proceed as directed in the Assay.
Standard solution: 0.001 mg/mL of USP Zolpidem Tartrate RS in Mobile phase
Sample solution: 0.5 mg/mL of Zolpidem Tartrate in Mobile phase
System suitability
Samples: System suitability solution and Standard solution
[NOTE-See Table 1 for the relative retention times.]
Suitability requirements
Resolution: NLT 2.0 between zolpidem related compound A and zolpidem, System suitability solution
Relative standard deviation: NMT 10.0%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Zolpidem Tartrate taken:
Result = (rU/rS) x (CS/CU) x 100
rU = peak response of each impurity from the Sample solution
rS = peak response of zolpidem from the Standard solution
CS = concentration of USP Zolpidem Tartrate RS in the Standard solution (mg/mL)
CU = concentration of Zolpidem Tartrate in the Sample solution (mg/mL)
Acceptance criteria: See Table 1. Disregard any peak less than 0.05%.
Table 1
| Name | Relative Retention Time | Acceptance Criteria, NMT (%) |
| Tartaric acida | 0.16 | — |
| Zolpidem related compound A | 0.83 | 0.10 |
| Zolpidem | 1.0 | — |
| Any individual impurity | — | 0.10 |
| Total impurities | — | 0.2 |
a Counter ion peak; not to be reported and not to be included in total impurities.
5 SPECIFIC TESTS
WATER DETERMINATION (921), Method I, Method la: NMT 3.0%
6 ADDITIONAL REQUIREMENTS
6.1 PACKAGING AND STORAGE
Preserve in well-closed containers, and store at controlled room temperature.
6.2 USP REFERENCE STANDARDS (11)
USP Zolpidem Tartrate RS
USP Zolpidem Related Compound A RS
N,N-Dimethyl-2-(7-methyl-2-p-tolylimidazo[1,2-a]pyridin-3-yl)acetamide.
C19H21N3O 307.39


