Zolmitriptan
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
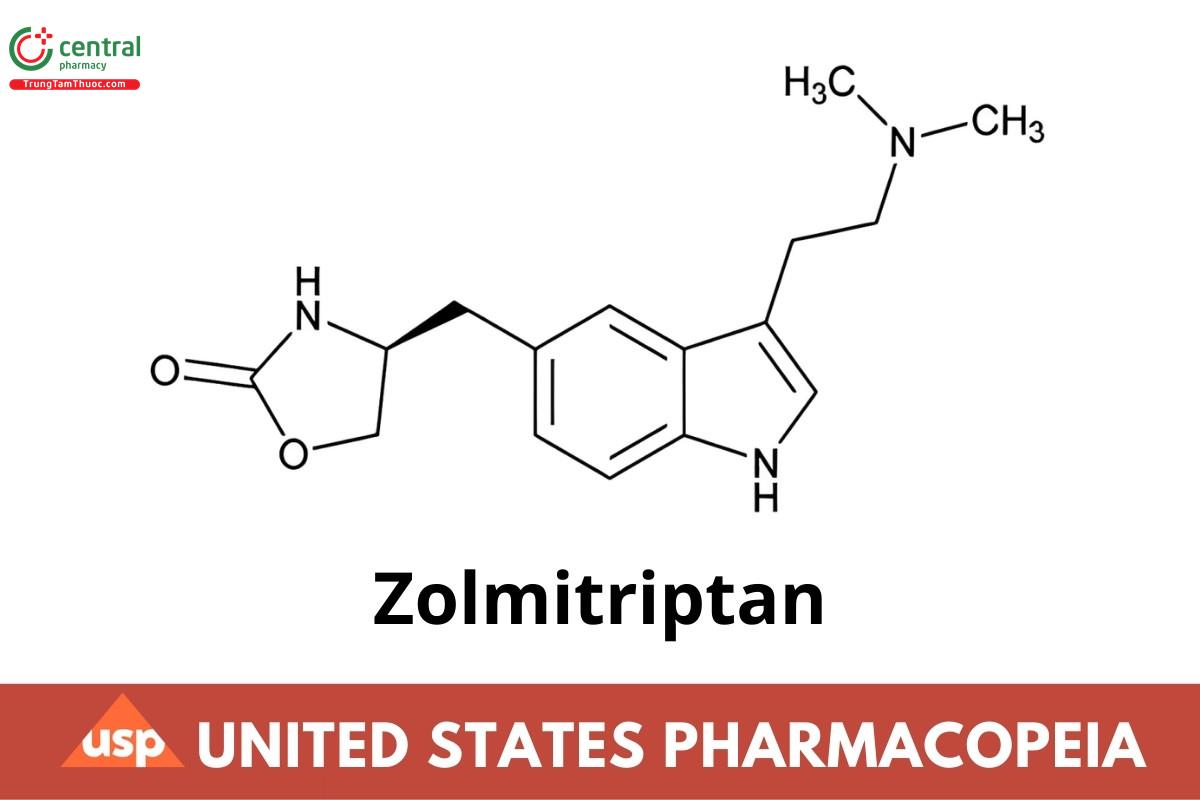
C16H21N3O2 287.36
2-Oxazolidinone, 4-[[3-[2-(dimethylamino)ethyl]-1H-indol-5-yl]methyl]-, (S)-;
(S)-4-({3-[2-(Dimethylamino)ethyl]indol-5-yl}methyl)oxazolidin-2-one;
(S)-4-[[3-[2-(Dimethylamino)ethyl]indol-5-yl]methyl]-2-oxazolidinone CAS RN®: 139264-17-8; UNII: 2FS66TH3YW.
1 DEFINITION
Zolmitriptan contains NLT 97.0% and NMT 102.0% of zolmitriptan (C16H21N3O2), calculated on the anhydrous and solvent-free basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS (197), Infrared Spectroscopy: 197K (CN 1-May-2020)
B. The migration time of the major peak of the Sample solution corresponds to that of the System suitability solution, as obtained in the test for Limit of Zolmitriptan R-Isomer and Other Impurities.
3 ASSAY
Change to read:
3.1 PROCEDURE
Mobile phase: Acetonitrile and water (135:865). For every liter of the mixture add 1 mL of trifluoroacetic acid and 0.25 mL of triethylamine.
System suitability solution: 0.12 µg/mL of USP Zolmitriptan Related Compound E RS and 25 µg/mL of USP Zolmitriptan RS in Mobile phase
Standard solution: 0.025 mg/mL of USP Zolmitriptan RS in Mobile phase
Sample solution: 0.025 mg/mL of Zolmitriptan in Mobile phase
3.2 Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 225 nm
Column: 4.0-mm x 12.5-cm; 5-µm packing 11
Flow rate: 1 mL/min
Injection volume: 20 µL
Run time: NLTA (IRA 1-May-2020) 3 times the retention time of zolmitriptan
3.3 System suitability
Sample: System suitability solution
[NOTE-The relative retention times for zolmitriptan and zolmitriptan related compound E are 1.0 and 1.6, respectively.]
Suitability requirements
Resolution: NLT 5 between zolmitriptan and zolmitriptan related compound E
Tailing factor: NMT 2.0 for zolmitriptan
Relative standard deviation: NMT 0.73% (IRA 1-May-2020)
3.4 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zolmitriptan (C16H21N3O2) in the portion of Zolmitriptan taken:
Result = (rU/rS) x (CS/CU) x 100
rU = peak response of zolmitriptan (IRA 1-May-2020) from the Sample solution
rS = peak response of zolmitriptan (IRA 1-May-2020) from the Standard solution
CS = concentration of USP Zolmitriptan RS in the Standard solution (mg/mL)
CU = concentration of Zolmitriptan in the Sample solution (mg/mL)
Acceptance criteria: 97.0%-102.0% on the anhydrous and solvent-free basis
4 IMPURITIES
4.1 RESIDUE ON IGNITION (281)
NMT 0.1%
4.2 LIMIT OF ZOLMITRIPTAN RELATED COMPOUND H
Perform this test only if zolmitriptan related compound H is a known process impurity. If this test is performed, then this is to be included in total impurities.
Standard solution: 0.1 mg/mL of USP Zolmitriptan Related Compound H RS in methanol
Sample solution: 200 mg/mL of Zolmitriptan in methanol
Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: GC
Detector: Flame ionization
Column: 0.32-mm x 15-m; 1-µm coating of dimethylpolysiloxane phase G1
Temperatures
Injection port: 200°
Column: 130°
Detector: 250°
Carrier gas: Helium at 6 mL/min
Injection volume: 3 µL
Injection type: Split, split ratio 10:1
System suitability
Sample: Standard solution
Suitability requirements
Tailing factor: NMT 2.0
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zolmitriptan related compound H in the portion of Zolmitriptan taken:
Result = (rU/rS) x (CS/CU) x 100
rU = peak response of zolmitriptan related compound H from the Sample solution
rS = peak response of zolmitriptan related compound H from the Standard solution
CS = concentration of USP Zolmitriptan Related Compound H.RS in the Standard solution (mg/mL)
CU = concentration of Zolmitriptan in the Sample solution (mg/mL)
Acceptance criteria: NMT 0.1% of zolmitriptan related compound H
Change to read:
4.3 LIMIT OF ZOLMITRIPTAN R-ISOMER AND OTHER IMPURITIES
Buffer: 19.1 g/L of sodium borate decahydrate in water. Adjust with phosphoric acid to a pH of 2.2.
Run buffer: 50 mg/mL of hydroxypropyl-B-cyclodextrin in Buffer
Diluent: 0.02 M hydrochloric acid
Internal standard solution: 0.05 mg/mL of tryptamine hydrochloride in Diluent
System suitability solution: 0.01 mg/mL of tryptamine hydrochloride from the Internal standard solution; 1 mg/mL of USP Zolmitriptan RS; and 0.01 mg/mL each of USP Zolmitriptan Related Compound F RS, USP Zolmitriptan Related Compound G RS, and USP Zolmitriptan R-Isomer RS in Diluent
Standard solution: 0.01 mg/mL of tryptamine hydrochloride from Internal standard solution and 0.001 mg/mL of USP Zolmitriptan RS in Diluent
Sample solution: 0.01 mg/mL of tryptamine hydrochloride from Internal standard solution and 1 mg/mL of Zolmitriptan in Diluent. Filter the solution and protect from light.
Capillary rinsing procedure: Use separate Run buffer vials for the capillary rinse and sample analysis. Condition the capillary by rinsing with 0.1 N sodium hydroxide followed by Run buffer before each injection. [NOTE-It may be suitable to rinse with 0.1 N sodium hydroxide using a pressure of 20 psi for NLT 1 min and then to rinse with Run buffer using a pressure of 20 psi for NLT 3 min.]
Electrophoretic system
Mode: CE
Detector: UV 200 nm
Column: 75-µm (ID) x 50-cm effective length, 60-cm total-length capillary uncoated fused-silica
Capillary temperature: 25°
Injection pressure: 0.5 psi for 5 s
Applied voltage: 15 kV
Run time: NLT 1.5 times the migration time of zolmitriptan
System suitability
Samples: System suitability solution and Standard solution
[NOTE-The relative migration time for tryptamine is 0.78; (IRA 1-May-2020) see Table 1 for the other (IRA 1-May-2020) relative migration times.]
Suitability requirements
Resolution: NLT 1.5 between zolmitriptan and zolmitriptan R-isomer, (IRA 1-May-2020) System suitability solution
Relative standard deviation: NMT 15% (IRA 1-May-2020) for the peak response ratio of zolmitriptan and tryptamine, (IRA 1-May-2020) Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the corrected peak responses:
Result = r/m
r = peak response
m = migration time of the peak (min)
Calculate the percentage of each impurity in the portion of Zolmitriptan taken:
Result = (RU/RS) x (CS/CU) x (1/F) × 100
RU = corrected peak response ratio of the impurity to the internal standard from the Sample solution
RS = corrected peak response ratio of zolmitriptan to the internal standard from the Standard solution
CS = concentration of USP Zolmitriptan RS in the Standard solution (mg/mL)
CU = concentration of Zolmitriptan in the Sample solution (mg/mL)
F = relative response factor (see Table 1)
Acceptance criteria: See Table 1. Disregard the peak due to zolmitriptan related compound E. The reporting threshold is 0.10%.
Table 1
| Name | Relative Migration Time | Relative Response Factor | Acceptance Criteria, NMT (%) |
| Zolmitriptan related compound G | 0.68 | 0.52 | 0.1 |
| Zolmitriptan related compound F | 0.71 | 0.41 | 1.2 |
| (IRA 1-May-2020) | (IRA 1-May-2020) | (IRA 1-May-2020) | (IRA 1-May-2020) |
| Zolmitriptan | 1.0 | — | — |
| Zolmitriptan R-isomer | 1.07 | 1.0 | 0.2 |
| Any individual unspecified impurity | — | 1.0 | 0.1 |
Change to read:
4.4 ORGANIC IMPURITIES
Mobile phase: Acetonitrile and water (135:865). For every liter of the mixture add 1 mL of trifluoroacetic acid and 0.25 mL of triethylamine.
System suitability solution: 0.5 µg/mL of USP Zolmitriptan Related Compound E RS and 0.1 mg/mL of USP Zolmitriptan RS in Mobile phase
Sample solution: 0.1 mg/mL of Zolmitriptan in Mobile phase
Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 225 nm
Column: 4.0-mm x 12.5-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
Run time: NLT 6 times the retention time of zolmitriptan
System suitability
Sample: System suitability solution
[NOTE-See Table 2 for relative retention times.]
Suitability requirements
Resolution: NLT 5 between zolmitriptan and zolmitriptan related compound E (IRA 1-May-2020)
Tailing factor: NMT 3.0 for zolmitriptan
Analysis
Sample: Sample solution
Calculate the percentage of each impurity in the portion of Zolmitriptan taken:
Result = (rU/rT) x 100
rU = peak response of each impurity from the Sample solution
rT = sum of the peak responses from the Sample solution
Acceptance criteria: See Table 2. The reporting threshold is 0.05%.
Table 2
| Name | Relative Retention Time | Acceptance Criteria, NMT (%) |
| Zolmitriptan related compound Ba | 0.36 | 0.2 |
| Zolmitriptan | 1.0 | — |
| Zolmitriptan related compound E | 1.6 | 0.2 |
| Zolmitriptan related compound Fb | 2.3 | — |
| Any individual unspecified impurity | — | 0.1 |
| Total impuritiesc | — | 0.5 |
a (S)-2-Amino-3-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}propan-1-ol.
b Included for identification purposes only. Not reported here as it is monitored under the Limit of Zolmitriptan R-Isomer and Other Impurities test. Not to be included in total impurities.
c Includes all impurities except zolmitriptan related compound F. Also includes zolmitriptan related compound H from the test for Limit of Zolmitriptan Related Compound H if it is a known process impurity, as well as impurities from the test for Limit of Zolmitriptan R-Isomer and Other Impurities with the exception of zolmitriptan related compound F.
5 SPECIFIC TESTS
WATER DETERMINATION (921), Method I, Method la: NMT 1.0%
6 ADDITIONAL REQUIREMENTS
6.1 PACKAGING AND STORAGE
Preserve in well-closed containers. Store at room temperature.
Change to read:
6.2 USP REFERENCE STANDARDS (11)
USP Zolmitriptan RS
USP Zolmitriptan R-Isomer RS
(R)-4-({3-[2-(Dimethylamino)ethyl]indol-5-yl}methyl)oxazolidin-2-one;
Also known as (IRA 1-May-2020) (R)-4-[[3-[2-(Dimethylamino) ethyl]indol-5-yl]methyl]-2-oxazolidinone.
C16H21N3O2 287.36
USP Zolmitriptan Related Compound E RS
(S)-N,N-Dimethyl-2-{5-[(2-oxooxazolidin-4-yl)methyl]-1H-indol-3-yl}ethanamine oxide.
C16H21N3O3 303.36
USP Zolmitriptan Related Compound F RS
2,2'-[4-(Dimethylamino)butane-1,1-diyl]bis{5-[(S)-(2-oxooxazolidin-4-yl)methyl]-3-(2-dimethylaminoethyl)indole}.
C38H53N7O4 671.89 (IRA 1-May-2020)
USP Zolmitriptan Related Compound G RS
(S)-4-(4-Aminobenzyl)oxazolidin-2-one.
C10H12N2O2 192.22 (IRA 1-May-2020)
USP Zolmitriptan Related Compound H RS
4,4-Diethoxy-N,N-dimethylbutan-1-amine.
C10H23NO2 189.30


