Zoledronic Acid
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
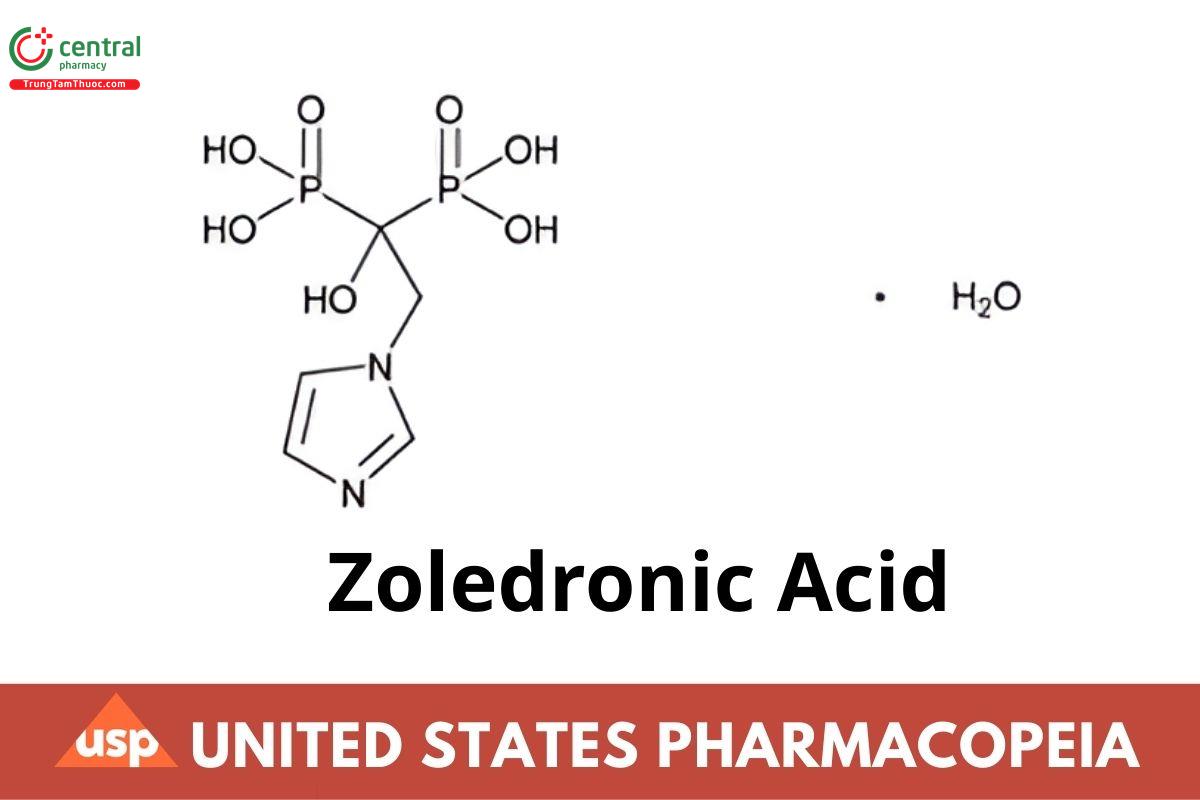
C5H10N2O7P2 · H2O 290.10
Phosphonic acid, [1-hydroxy-2-(1H-imidazol-1-yl)ethylidene]bis-, monohydrate;
(1-Hydroxy-2-imidazol-1-ylethylidene)diphosphonic acid, monohydrate CAS RN®: 165800-06-6.
Anhydrous CAS RN®: 118072-93-8. 272.09
1 DEFINITION
Zoledronic acid contains NLT 98.0% and NMT 102.0% of zoledronic acid (C5H10N2O7P2), calculated on the anhydrous basis.
2 IDENTIFICATION
A. Spectroscopic Identification Tests 〈197〉, Infrared Spectroscopy: 197K or 197A
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
Procedure
Perchloric acid solution: Carefully add 10 mL of perchloric acid to 1 L of water, then add 2 mL of phosphoric acid, and mix.
Buffer: 0.037 g/L of edetate disodium and 10.8 g/L of octanesulfonic acid sodium salt in Perchloric acid solution
Mobile phase: Acetonitrile and Buffer (4:96)
System suitability solution: 2 μg/mL each of USP Imidazole RS, USP Zoledronic Acid Related Compound A RS, and sodium nitrate; 5 μg/mL of USP Zoledronic Acid Related Compound B RS; and 1.2 mg/mL of USP Zoledronic Acid RS in Mobile phase
Standard solution: 1 mg/mL of USP Zoledronic Acid RS in Mobile phase. Sonicate with occasional shaking for about 15 min to achieve complete dissolution.
Sample solution: 1 mg/mL of Zoledronic Acid in Mobile phase. Sonicate with occasional shaking for about 15 min to achieve complete dissolution.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
[Note - Plastic HPLC vials are recommended.]
Preconditioning of the instrument: Rinse the instrument without a column with a mixture of acetic acid and water (25:75) at 5 mL/min for 20 min. Then, rinse with water at 5 mL/min for about 2 h.
Preconditioning of the column: Rinse with Mobile phase at 0.6 mL/min for about 1 h. During the rinsing, inject the Sample solution 10–15 times, applying a run time of about 3 min for each injection.
Mode: LC
Detector: UV 215 nm
Column: 4.6-mm × 25-cm; 5-μm packing L11
Flow rate: 0.6 mL/min
Injection volume: 5 μL
Run time: NLT 3 times the retention time of zoledronic acid
System suitability
Samples: System suitability solution and Standard solution
Suitability requirements
Resolution: NLT 1.0 between the nitrate and zoledronic acid related compound B peaks, System suitability solution
Tailing factor: NMT 1.4 for the zoledronic acid peak, System suitability solution
Relative standard deviation: NMT 0.73%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zoledronic acid (C5H10N2O7P2) in the portion of Zoledronic Acid taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of zoledronic acid from the Sample solution
rS = peak response of zoledronic acid from the Standard solution
CS = concentration of USP Zoledronic Acid RS in the Standard solution (mg/mL)
CU = concentration of Zoledronic Acid in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–102.0% on the anhydrous basis
4 IMPURITIES
Change to read:
Organic Impurities
Perchloric acid solution, Buffer, Mobile phase, and System suitability solution: Prepare as directed in the Assay.
Standard stock solution: 0.4 mg/mL of USP Zoledronic Acid RS in Mobile phase. Sonicate with occasional shaking for about 15 min to achieve complete dissolution.
Sensitivity solution: 1 μg/mL of USP Zoledronic Acid RS in Mobile phase from Standard stock solution
Standard solution: 0.002 mg/mL of USP Zoledronic Acid RS in Mobile phase from Standard stock solution
Sample solution: 2 mg/mL of Zoledronic Acid in Mobile phase. Sonicate with occasional shaking for about 30 min to achieve complete dissolution.
Chromatographic system: Proceed as directed in the Assay, except for Injection volume and Run time.
Injection volume: 10 μL
Run time: NLT 5 times the retention time of zoledronic acid
System suitability
Samples: System suitability solution, Sensitivity solution, and Standard solution
Suitability requirements
Resolution: NLT 1.0 between the nitrate and zoledronic acid related compound B peaks, System suitability solution
Tailing factor: NMT 1.4 for the zoledronic acid peak, Standard solution
Relative standard deviation: NMT 5.0%, Standard solution
Signal-to-noise ratio: NLT 10, Sensitivity solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of any individual impurity in the portion of Zoledronic Acid taken:
Result = (rU/rS) × (CS/CU) × (1/F) x 100
rU = peak response of any individual impurity (ERR 1-Mar-2022) from the Sample solution
rS = peak response of zoledronic acid from the Standard solution
CS = concentration of USP Zoledronic Acid RS in the Standard solution (mg/mL)
CU = concentration of Zoledronic Acid in the Sample solution (mg/mL)
F = relative response factor (see Table 1)
Acceptance criteria: See Table 1. The reporting threshold is 0.05%.
Table 1
| Name | Relative Retention Time | Relative Response Factor | Acceptance Criteria NMT (%) |
|---|---|---|---|
| Sodium nitrate | 0.6 | — | — |
| Zoledronic acid related compound B | 0.7 | 0.53 | 0.5 |
| Zoledronic acid | 1.0 | — | — |
| Zoledronic acid related compound A | 3.1 | 3.0 | 0.15 |
| Imidazole | 4.4 | 4.4 | 0.15 |
| Any unspecified impurity | — | 1.0 | 0.10 |
| Total impurities | — | — | 0.8 |
Limit of Phosphate and Phosphite (if present)
Mobile phase: Add 0.47 mL of formic acid to 2.5 L of water and adjust with 2 N sodium hydroxide to a pH of 3.5.
Phosphoric acid stock solution: 0.65 mg/mL of phosphoric acid in Mobile phase
Phosphorus acid stock solution: 0.50 mg/mL of phosphorous acid in Mobile phase
System suitability solution: 13 μg/mL of phosphoric acid from Phosphoric acid stock solution, 10 μg/mL of phosphorous acid from Phosphorus acid stock solution, and 1 mg/mL of USP Zoledronic Acid RS in Mobile phase
Standard solution: 0.013 mg/mL of phosphoric acid from Phosphoric acid stock solution and 10 μg/mL of phosphorous acid from Phosphorus acid stock solution
Sample solution: 1 mg/mL of Zoledronic Acid in Mobile phase
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: Refractive index
Column: 4.6-mm × 15-cm; 5-μm packing L31
Temperatures
Column: 35°
Detector: 35°
Flow rate: 1 mL/min
Injection volume: 100 μL
Run time: NLT 4 times the retention time of zoledronic acid
System suitability
Samples: System suitability solution and Standard solution
Suitability requirements
Resolution: NLT 1.5 between the zoledronic acid and phosphate peaks, System suitability solution
Relative standard deviation: NMT 10.0% for the phosphate and phosphite peaks, Standard solution
Analysis
Samples: Mobile phase, Standard solution, and Sample solution. [Note—Disregard any peak observed in the blank.]
Calculate the percentage of phosphate (determined as phosphoric acid) and phosphite (determined as phosphorus acid) in the portion of Zoledronic Acid taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of phosphate or phosphite from the Sample solution
rS = peak response of phosphate or phosphite from the Standard solution
CS = concentration of phosphoric acid or phosphorus acid in the Standard solution (mg/mL)
CU = concentration of Zoledronic Acid in the Sample solution (mg/mL)
Acceptance criteria: See Table 2.
Table 2
| Name | Relative Retention Time | Acceptance Criteria NMT (%) |
|---|---|---|
| Zoledronic acid | 1.0 | — |
| Phosphatea | 1.4 | 0.50 |
| Phosphiteb | 2.0 | 0.50 |
a Determined as phosphoric acid.
b Determined as phosphorus acid.
5 SPECIFIC TESTS
Bacterial Endotoxins Test 〈85〉: Where the label states that Zoledronic Acid must be subjected to further processing during the preparation of injectable dosage forms, the levels of bacterial endotoxins are such that the requirement under the relevant dosage form monograph(s) in which Zoledronic Acid is used can be met.
Microbial Enumeration Tests 〈61〉 and Tests for Specified Microorganisms 〈62〉: The total aerobic microbial count does not exceed 102 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g.
Water Determination 〈921〉, Method I, Method Ia: For the monohydrate form, 5.0%–7.5%. For the anhydrous form, NMT 0.5%. [Note - Hydranyl solvent and Hydranal titrant 5 are suitable.]
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in well-closed containers. Store at room temperature.
Labeling: Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms to ensure acceptable levels of bacterial endotoxins, it is so labeled.
USP Reference Standards 〈11〉
USP Imidazole RS
1H-Imidazole.
C3H4N268.08
USP Zoledronic Acid RS
USP Zoledronic Acid Related Compound A RS
2-(1H-Imidazol-1-yl)acetic acid.
C5H6N2O2 126.12
USP Zoledronic Acid Related Compound B RS
1-Hydroxy-2-[1-(2-hydroxy-2,2-diphosphonoethyl)-1H-imidazol-3-ium-3-yl]-1-phosphonoethylphosphonate.
C7H16N2O14P4 476.10


