Ziprasidone Hydrochloride
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
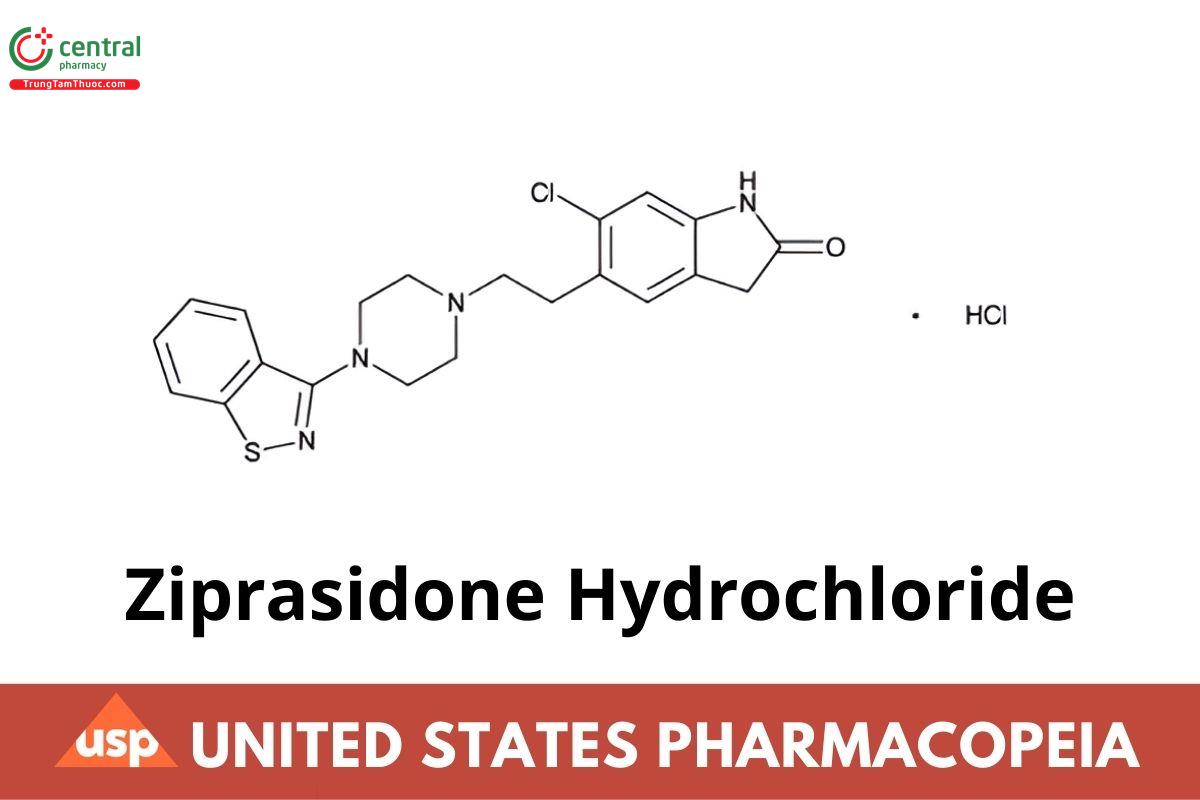
C21H21ClN4OS · HCl 449.39
C21H21ClN4OS · HCl · H2O 467.41
2H-Indol-2-one, 5-{2-[4-(1,2-benzisothiazol-3-yl)-1-piperazinyl]ethyl}-6-chloro-1,3-dihydro-, monohydrochloride;
5-{2-[4-(1,2-Benzisothiazol-3-yl)-1-piperazinyl]ethyl}-6-chloro-2-indolinone monohydrochloride.
Monohydrate CAS RN®: 138982-67-9; UNII: 216X081ORU.
Anhydrous CAS RN®: 122883-93-6; UNII: JAL53626GG.
1 DEFINITION
Ziprasidone Hydrochloride contains NLT 97.5% and NMT 102.0% of ziprasidone hydrochloride (C21H21ClN4OS · HCl), calculated on the anhydrous and solvent-free basis.
2 IDENTIFICATION
A. Spectroscopic Identification Tests 〈197〉, Infrared Spectroscopy: 197A or 197K. If a difference appears in the spectra of the analyte and the Reference Standard, dissolve equal portions of the analyte and the Reference Standard in equal volumes of methanol separately, evaporate to dryness, and record new spectra using the residues.
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
C. Identification Tests - General 〈191〉, Chemical Identification Tests, Chloride
Sample solution: Suspend 30 mg of the analyte in 2 mL of water, acidify with 0.15 mL of 2 N nitric acid TS, and filter. Use the filtrate.
Acceptance criteria: Meets the requirements
3 ASSAY
Procedure
Buffer: 6.8 g/L of monobasic potassium phosphate in water. Adjust with phosphoric acid to a pH of 3.0.
Mobile phase: Methanol and Buffer (40:60)
Diluent: Methanol and water (60:40)
Standard solution: 0.23 mg/mL of USP Ziprasidone Hydrochloride RS in Diluent
Sample solution: 0.23 mg/mL of Ziprasidone Hydrochloride in Diluent. [Note—Because of its hygroscopic nature, the anhydrous form may be dried at 70° under vacuum for 2 h prior to the preparation of the Sample solution.]
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 229 nm
Column: 4.6-mm × 15-cm; 5-μm packing L7
Column temperature: 40°
Flow rate: 1.5 mL/min
Injection volume: 20 μL
Run time: NLT 2 times the retention time of ziprasidone
System suitability
Sample: Standard solution
Suitability requirements
Tailing factor: NMT 2.0
Relative standard deviation: NMT 0.73%
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of ziprasidone hydrochloride (C21H21ClN4OS · HCl) in the portion of Ziprasidone Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of ziprasidone from the Sample solution
rS = peak response of ziprasidone from the Standard solution
CS = concentration of USP Ziprasidone Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Ziprasidone Hydrochloride in the Sample solution (mg/mL)
Acceptance criteria: 97.5%–102.0% on the anhydrous and solvent-free basis
4 IMPURITIES
Residue on Ignition 〈281〉: NMT 0.2%
Limit of Tetrahydrofuran
[Note - Perform this test only if tetrahydrofuran is likely to be present as the result of the manufacturing process.]
Standard solution: 0.05 mg/mL of tetrahydrofuran in dimethyl sulfoxide. Transfer 4 mL of this solution to a 20-mL headspace vial, and seal.
Sample solution: Transfer 40 mg of Ziprasidone Hydrochloride and 4.0 mL of Dimethyl sulfoxide to a 20-mL headspace vial, seal, and mix.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: GC with headspace injector
Detector: Flame ionization
Column: 30-m × 0.32-mm fused silica; coated with a 1.8-μm film of phase G43
Temperatures
Injection port: 180°
Detector: 260°
Column: See Table 1.
Table 1
| Initial Temperature (°) | Temperature Ramp (°/min) | Final Temperature (°) | Hold Time at Final Temperature (min) |
|---|---|---|---|
| 40 | 0 | 40 | 5 |
| 40 | 2 | 90 | 0 |
| 90 | 30 | 225 | 2 |
Carrier gas: Helium
Flow rate: 1.6 mL/min
Injection volume: 2 mL
Injection type: Headspace; split ratio, 30:1
Vial treatment: Maintain at 105° for 60 min prior to injection
System suitability
Sample: Standard solution
Suitability requirements
Relative standard deviation: NMT 5%
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of tetrahydrofuran in the portion of Ziprasidone Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of tetrahydrofuran from the Sample solution
rS = peak response of tetrahydrofuran from the Standard solution
CS = concentration of tetrahydrofuran in the Standard solution (mg/mL)
CU = concentration of Ziprasidone Hydrochloride in the Sample solution (mg/mL)
Acceptance criteria: NMT 0.4% of tetrahydrofuran
Change to read:
Organic Impurities
Buffer: 6.8 g/L of monobasic potassium phosphate in water
Solution A: Methanol and Buffer (33:67). Adjust with phosphoric acid to a pH of 3.0.
Solution B: Acetonitrile, methanol, and Buffer (55:5:40). Adjust with potassium hydroxide solution (ERR 1-Sep-2023) to a pH of 6.0.
Mobile phase: See Table 2.
Table 2
| Time (min) | Solution A (%) | Solution B (%) |
|---|---|---|
| 0 | 100 | 0 |
| 15 | 100 | 0 |
| 20 | 85 | 15 |
| 30 | 85 | 15 |
| 40 | 55 | 45 |
| 55 | 40 | 60 |
| 65 | 25 | 75 |
| 70 | 20 | 80 |
| 71 | 100 | 0 |
| 75 | 100 | 0 |
Diluent: Acetonitrile, methanol, and water (40:10:50). Adjust with phosphoric acid to a pH of 2.5.
System suitability solution: 0.5 mg/mL of USP Ziprasidone Hydrochloride RS and 0.05 mg/mL each of USP Ziprasidone Related Compound A RS, USP Ziprasidone Related Compound B RS, USP Ziprasidone Related Compound C RS, USP Ziprasidone Related Compound D RS, and USP Ziprasidone Related Compound F RS in Diluent. [Note - Ziprasidone related compound F is stable for 6 h at 10° in this solution.]
Sensitivity solution: 0.5 μg/mL of USP Ziprasidone Hydrochloride RS in Diluent
Standard solution: 0.001 mg/mL of USP Ziprasidone Hydrochloride RS and 0.002 mg/mL each of USP Ziprasidone Related Compound A RS, USP Ziprasidone Related Compound B RS, USP Ziprasidone Related Compound C RS, and USP Ziprasidone Related Compound D RS in Diluent
Sample solution: 1.0 mg/mL of Ziprasidone Hydrochloride in Diluent. [Note—Because ziprasidone related compound F is stable only for 6 h at 10°, it is recommended that the freshly prepared Sample solution be analyzed.]
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 229 nm
Column: 4.6-mm × 15-cm; 5-μm packing L7
Temperatures
Autosampler: 10°
Column: 30°
Flow rate: 1.5 mL/min
Injection volume: 10 μL
System suitability
Samples: System suitability solution, Sensitivity solution, and Standard solution
[Note - The relative retention times in Table 3 are provided as information that could aid to peak assignment.]
Table 3
| Name | Relative Retention Time |
|---|---|
| Ziprasidone related compound A | 0.22 |
| Chloroindolinonea | 0.59 |
| Ziprasidone related compound B | 0.70 |
| Ziprasidone related compound F | 0.84 |
| Ziprasidone | 1.0 |
| Ziprasidone related compound C | 1.84 |
| Ziprasidone related compound D | 2.18 |
a 6-Chloroindolin-2-one.
Suitability requirements
Resolution: NLT 1.5 between ziprasidone related compound B and related compound F; NLT 2.0 between ziprasidone related compound F and ziprasidone, System suitability solution
Tailing factor: NMT 1.5 for ziprasidone, Standard solution
Relative standard deviation: NMT 5.0% for ziprasidone, ziprasidone related compound B, ziprasidone related compound C, and ziprasidone related compound D, Standard solution
Signal-to-noise ratio: NLT 10, Sensitivity solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of ziprasidone related compounds A, B, C, and D, relative to the content of ziprasidone free base, in the portion of Ziprasidone Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × 100
rU = peak response of ziprasidone related compounds A, B, C, and D from the Sample solution
rS = peak response of the corresponding USP Reference Standard from the Standard solution
CS = concentration of the corresponding USP Reference Standard in the Standard solution (mg/mL)
CU = concentration of Ziprasidone Hydrochloride in the Sample solution (mg/mL)
Mr1 = molecular weight of ziprasidone hydrochloride in the sample taken, 467.41 for the monohydrate, 449.39 (ERR 1-Sep-2023) for the anhydrous form
Mr2 = molecular weight of ziprasidone free base, 412.94
Calculate the percentage of chloroindolinone, ziprasidone related compound F, and any unspecified impurity, relative to the content of ziprasidone free base, in the portion of Ziprasidone Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × (1/F) × (Mr1/Mr2) × (Mr3/Mr1) × 100
rU = peak response of chloroindolinone, ziprasidone related compound F, or any unspecified impurity from the Sample solution
rS = peak response of ziprasidone from the Standard solution
CS = concentration of USP Ziprasidone Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Ziprasidone Hydrochloride in the Sample solution (mg/mL)
F = relative response factor (see Table 4)
Mr1 = molecular weight of ziprasidone free base, 412.94
Mr2 = molecular weight of ziprasidone hydrochloride anhydrous form, 449.39 (ERR 1-Sep-2023)
Mr3 = molecular weight of ziprasidone hydrochloride in the sample taken, 467.41 for the monohydrate, 449.39 (ERR 1-Sep-2023) for the anhydrous form
Acceptance criteria: See Table 4. The reporting threshold is 0.05%.
Table 4
| Name | Relative Response Factor | Acceptance Criteria, NMT (%) |
|---|---|---|
| Ziprasidone related compound A | — | 0.1 |
| Chloroindolinonea | 0.47 | 0.15 |
| Ziprasidone related compound B | — | 0.2 |
| Ziprasidone related compound F | 0.83 | 0.2 |
| Ziprasidone related compound C | — | 0.2 |
| Ziprasidone related compound D | — | 0.2 |
| Any unspecified impurity | 1.0 | 0.1 |
| Total impurities | — | 0.5 |
a A process impurity specific to a manufacturing process; may not be present in all manufacturing processes.
5 SPECIFIC TESTS
Water Determination 〈921〉, Method I
Monohydrate: 3.7%–5.0%
Anhydrous: NMT 2.0%
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in tight, light-resistant containers, and store at room temperature.
Labeling: If this is an anhydrous form, it is so labeled.
USP Reference Standards 〈11〉
USP Ziprasidone Hydrochloride RS
USP Ziprasidone Related Compound A RS
3-(Piperazin-1-yl)benzoisothiazole monohydrochloride;
also known as 3-(Piperazin-1-yl)benzo[d]isothiazole monohydrochloride.
C11H13N3S · HCl 255.77
USP Ziprasidone Related Compound B RS
5-{2-[4-(Benzoisothiazol-3-yl)piperazin-1-yl]ethyl}-6-chloroindoline-2,3-dione;
also known as 5-{2-[4-(Benzo[d]isothiazol-3-yl)piperazin-1-yl]ethyl}-6-chloroindoline-2,3-dione.
C21H19ClN4O2S 426.92
USP Ziprasidone Related Compound C RS
5,5′-Bis{2-[4-(benzoisothiazol-3-yl)piperazin-1-yl]ethyl}-6,6′-dichloro-3-hydroxy-3,3′-biindoline-2,2′-dione;
also known as 5,5′-Bis{2-[4-(benzo[d]isothiazol-3-yl)piperazin-1-yl]ethyl}-6,6′-dichloro-3-hydroxy-3,3′-biindoline-2,2′-dione.
C42H40Cl2N8O3S2 839.85
USP Ziprasidone Related Compound D RS
3-(Benzoisothiazol-3-yl)-5-{2-[4-(benzoisothiazol-3-yl)piperazin-1-yl]ethyl}-6-chloroindolin-2-one;
also known as 3-(Benzo[d]isothiazol-3-yl)-5-{2-[4-(benzo[d]isothiazol-3-yl)piperazin-1-yl]ethyl}-6-chloroindolin-2-one.
C21H21ClN4OS 546.11
USP Ziprasidone Related Compound F RS
Sodium 2-(2-amino-5-{2-[4-(benzisothiazol-3-yl)piperazin-1-yl]ethyl}-4-chlorophenyl)acetate monohydrate;
also known as Sodium 2-(2-amino-5-{2-[4-(benzo[d]isothiazol-3-yl)piperazin-1-yl]ethyl}-4-chlorophenyl)acetate monohydrate.
C21H22ClN4NaO2S · H2O 470.95


