Zinc Gluconate
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
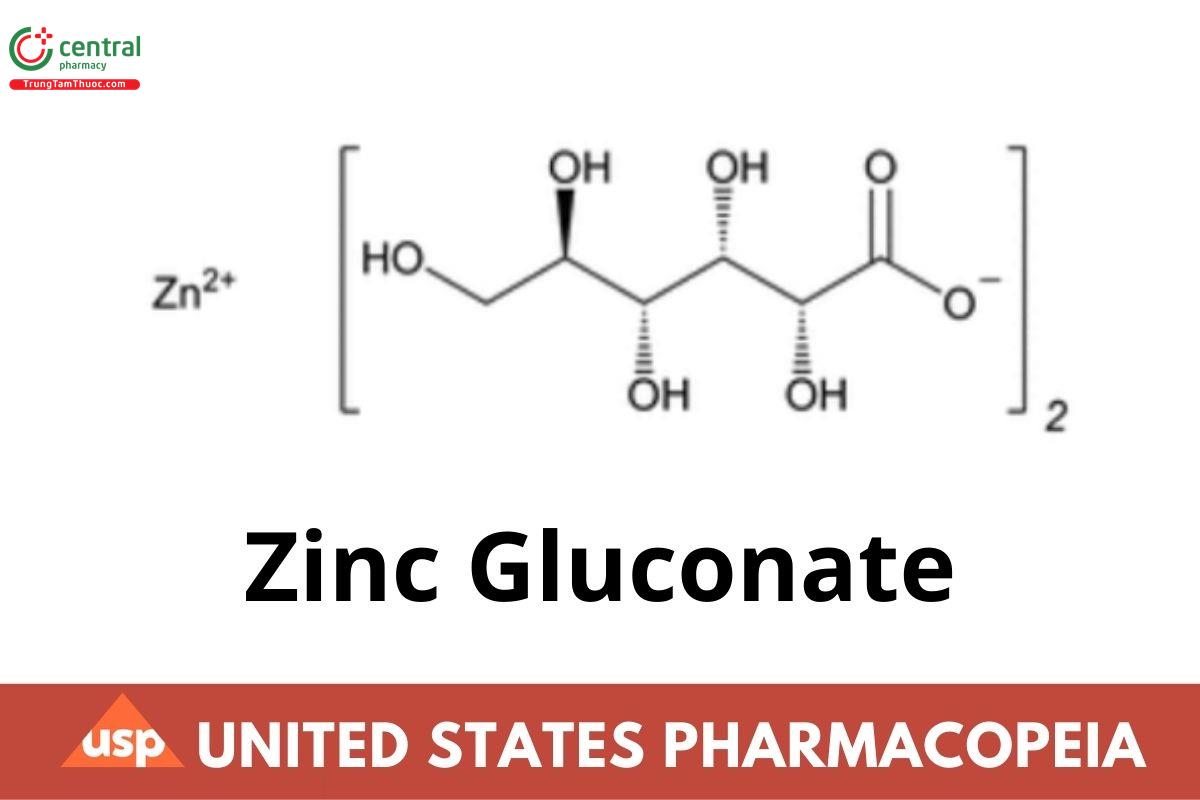
C12H22O14Zn 455.67
Bis(d-gluconato-O1,O2) zinc;
Zinc d-gluconate (1:2) CAS RN®: 4468-02-4.
1 DEFINITION
Zinc Gluconate contains NLT 97.0% and NMT 102.0% of zinc gluconate (C12H22O14Zn), calculated on the anhydrous basis.
2 IDENTIFICATION
A. Identification Tests—General, Zinc 〈191〉: A 100-mg/mL solution meets the requirements.
B. Thin-Layer Chromatography
Standard solution: 10 mg/mL of USP Potassium Gluconate RS
Sample solution: 10 mg/mL of Zinc Gluconate, heating in a water bath at 60°, if necessary, to dissolve
2.1 Chromatographic system
(See Chromatography 〈621〉, Thin-Layer Chromatography.)
Mode: TLC
Adsorbent: 0.25-mm layer of chromatographic silica gel
Application volume: 5 µL
Developing solvent system: Alcohol, ethyl acetate, ammonium hydroxide, and water (50:10:10:30)
Spray reagent: Dissolve 2.5 g of ammonium molybdate in 50 mL of 2 N sulfuric acid in a 100-mL volumetric flask, add 1.0 g of ceric sulfate, swirl to dissolve, and dilute with 2 N sulfuric acid to volume.
2.2 Analysis
Samples: Standard solution and Sample solution
Develop the chromatogram until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, and dry at 110° for 20 min. Allow to cool, and spray with the Spray reagent. Heat the plate at 110° for about 10 min. Acceptance criteria: The principal spot of the Sample solution corresponds in color, size, and RF value to that of the Standard solution.
3 ASSAY
Procedure
Sample: 700 mg of Zinc Gluconate
Blank: 100 mL of water
Titrimetric system
(See Titrimetry 〈541〉.)
Mode: Direct titration
Titrant: 0.05 M edetate disodium VS
Endpoint detection: Visual
Analysis: Dissolve the Sample in 100 mL of water. Add 5 mL of ammonia–ammonium chloride buffer TS and 0.1 mL of eriochrome black TS. Titrate with Titrant until the solution is deep blue in color. Perform a blank determination.
Calculate the percentage of zinc gluconate (C12H22O14Zn) in the Sample taken:
Result = {[(VS − VB ) × M × F]/W} × 100
VS = Titrant volume consumed by the Sample (mL)
VB = Titrant volume consumed by the Blank (mL)
M = Titrant molarity (mmol/mL)
F = equivalency factor, 455.7 mg/mmol
W = Sample weight (mg)
Acceptance criteria: 97.0%–102.0% on the anhydrous basis
4 IMPURITIES
4.1 Chloride and Sulfate, Chloride 〈221〉
Standard solution: 0.7 mL of 0.020 N hydrochloric acid
Sample: 1.0 g
Acceptance criteria: NMT 0.05%
4.2 Chloride and Sulfate, Sulfate 〈221〉
Standard solution: 1.0 mL of 0.020 N sulfuric acid
Sample: 2.0 g
Acceptance criteria: NMT 0.05%
Change to read:
4.3 Arsenic 〈211〉, Procedures, Procedure 1 (CN 1-Jun-2023)
Test preparation: 1 g in 35 mL of water
Acceptance criteria: NMT 3 ppm
4.4 Limit of Lead
[Note—For the preparation of all aqueous solutions and for the rinsing of glassware before use, use water that has been passed through a strong-acid, strong-base, mixed-bed ion-exchange resin. Select all reagents to have as low a content of lead as practicable, and store all reagent solutions in containers of borosilicate glass. Cleanse glassware before use by soaking in warm 8 N nitric acid for 30 min and by rinsing with deionized water.]
Ascorbic acid–sodium iodide solution: 100 mg/mL of ascorbic acid and 192.5 mg/mL of sodium iodide
Trioctylphosphine oxide solution: 50 mg/mL of trioctylphosphine oxide in 4-methyl-2-pentanone.
[Caution—This solution causes irritation. Avoid contact with eyes, skin, and clothing. Take special precautions in disposing of unused portions of solutions to which this reagent is added.]
Standard solution: Transfer 5.0 mL of lead nitrate stock solution TS, to a 100-mL volumetric flask, and dilute with water to volume. Transfer 2.0 mL of the resulting solution to a 50-mL volumetric flask, and add 10 mL of 9 N hydrochloric acid and 10 mL of water. Add 20 mL of Ascorbic acid–sodium iodide solution and 5.0 mL of Trioctylphosphine oxide solution. Shake for 30 s, and allow to separate. Add water to bring the organic solvent layer into the neck of the flask, shake again, and allow to separate. The organic layer is the Standard solution, and it contains 2 µg/mL of lead.
Sample solution: To a 50-mL volumetric flask add 1.0 g of Zinc Gluconate, 10 mL of 9 N hydrochloric acid, 10 mL of water, 20 mL of Ascorbic acid–sodium iodide solution, and 5.0 mL of Trioctylphosphine oxide solution. Shake for 30 s, and allow to separate. Add water to bring the organic solvent layer into the neck of the flask, shake again, and allow to separate. The organic layer is the Sample solution.
Blank: To a 50-mL volumetric flask add 10 mL of 9 N hydrochloric acid, 10 mL of water, 20 mL of Ascorbic acid–sodium iodide solution, and 5.0 mL of Trioctylphosphine oxide solution. Shake for 30 s, and allow to separate. Add water to bring the organic solvent layer into the neck of the flask, shake again, and allow to separate. The organic layer is the Blank, and it contains 0 µg/mL of lead.
4.4.1 Instrumental conditions
(See Atomic Absorption Spectroscopy 〈852〉.)
Mode: Atomic absorption spectrophotometry
Analytical wavelength: 283.3 nm
Lamp: Lead hollow-cathode
Flame: Air–acetylene
4.4.2 System suitability
Samples: Standard solution and Blank
Suitability requirements: The absorbance of the Standard solution and the absorbance of the Blank are significantly different. Analysis
Samples: Standard solution, Sample solution, and Blank
Concomitantly determine the absorbances of the Blank, Standard solution, and the Sample solution. Use the Blank to set the instrument to zero.
Acceptance criteria: NMT 10 ppm: the absorbance of the Sample solution does not exceed that of the Standard solution. • Limit of Cadmium
Standard stock solution: 0.1372 mg/mL of cadmium nitrate
Standard solution: Pipet 25 mL of the Standard stock solution into a 100-mL volumetric flask, add 1 mL of hydrochloric acid, dilute with water to volume, and mix. It contains 12.5 µg/mL of cadmium (Cd).
Sample stock solution: Transfer 10.0 g of Zinc Gluconate into a 50-mL volumetric flask, and dissolve in and dilute with water to volume. Sample solution A: Transfer 5.0 mL of the Sample stock solution to a 25-mL volumetric flask, and dilute with water to volume. This solution contains 0 µg/mL of added cadmium from the Standard solution.
Sample solution B: Transfer 5.0 mL of the Sample stock solution to a 25-mL volumetric flask, add 2.0 mL of the Standard solution, and dilute with water to volume. This solution contains 1.0 µg/mL of added cadmium from the Standard solution.
Sample solution C: Transfer 5.0 mL of the Sample stock solution to a 25-mL volumetric flask, add 4.0 mL of the Standard solution, and dilute with water to volume. This solution contains 2.0 µg/mL of added cadmium from the Standard solution.
Blank: Water
Instrumental conditions
(See Atomic Absorption Spectroscopy 〈852〉.)
Mode: Atomic absorption spectrophotometry
Analytical wavelength: 228.8 nm
Lamp: Cadmium hollow-cathode
Flame: Air–acetylene
4.4.3 Analysis
Samples: Sample solution A, Sample solution B, and Sample solution C
Determine the absorbances. Correct the absorbance values of Sample solution A, Sample solution B, and Sample solution C from the Blank. Plot the corrected absorbances of Sample solution A, Sample solution B, and Sample solution C versus their added cadmium concentrations, in µg/mL. Draw the straight line best fitting the three points, and extrapolate the line until it intercepts the concentration axis. From the intercept, determine the amount, in µg/mL, of cadmium in Sample solution A.
Calculate the content of cadmium in the portion of Zinc Gluconate taken:
Result = (C × V)/W
C = concentration of cadmium in Sample solution A (µg/mL), determined from the intercept of the linear regression line
V = volume of solvent taken to prepare Sample solution A (mL)
W = weight of Zinc Gluconate taken to prepare Sample solution A (g)
Acceptance criteria: NMT 5 ppm
4.5 Reducing Substances
Sample: 1.0 g of Zinc Gluconate
Blank: 10 mL of water
Titrimetric system
(See Titrimetry 〈541〉.)
Mode: Residual titration
Titrant 0.1 N iodine VS
Back titrant: 0.1 N sodium thiosulfate VS
Endpoint detection: Visual
Analysis: Transfer the Sample to a 250-mL conical flask, dissolve in 10 mL of water, and add 25 mL of alkaline cupric citrate TS. Cover the flask, boil gently for 5 min, accurately timed, and cool rapidly to room temperature. Add 25 mL of 0.6 N acetic acid, 10.0 mL of Titrant, and 10 mL of 3 N hydrochloric acid, and titrate with Back titrant, adding 3 mL of starch TS as the endpoint is approached. Perform the blank determination.
Calculate the percentage of reducing substances (as dextrose) in the Sample taken:
Result = {[(VB − VS ) × N × F]/W} × 100
VB = Back titrant volume consumed by the Blank (mL)
VS = Back titrant volume consumed by the Sample (mL)
N = Back titrant normality (mEq/mL)
F = equivalency factor, 27 mg/mEq
W = Sample weight (mg)
Acceptance criteria: NMT 1.0%
5 SPECIFIC TESTS
pH 〈791〉
Sample solution: 10 mg/mL
Acceptance criteria: 5.5–7.5
Water Determination, Method Ib 〈921〉: NMT 11.6%
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in well-closed containers.
USP Reference Standards 〈11〉
USP Potassium Gluconate RS


