Zinc Acetate
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
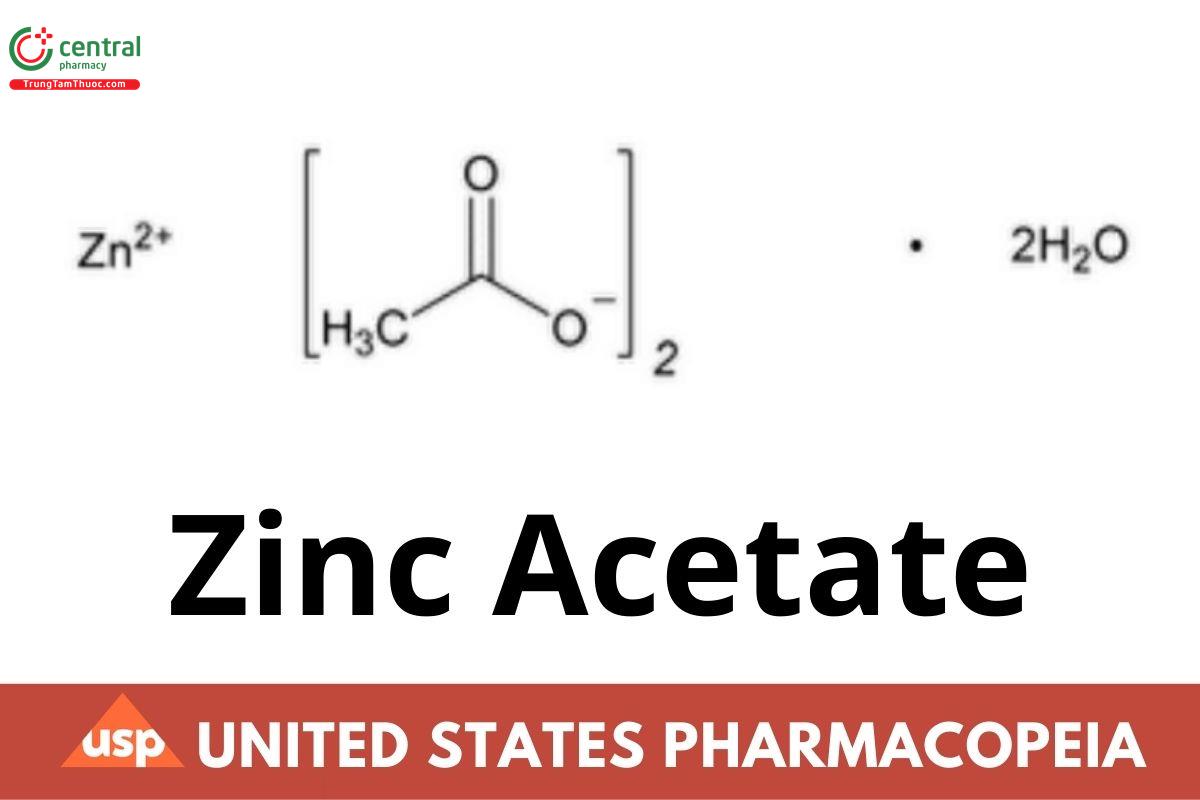
C4H6O4Zn · 2H2O 219.50
C4H6O4Zn 183.47
Acetic acid, zinc salt, dihydrate;
Zinc acetate dihydrate CAS RN®: 5970-45-6; UNII: FM5526K07A.
Zinc acetate anhydrous CAS RN®: 557-34-6; UNII: H2ZEY72PME.
1 DEFINITION
Zinc Acetate contains NLT 98.0% and NMT 102.0% of zinc acetate dihydrate (C4H6O4Zn · 2H2O).
2 IDENTIFICATION
A. The retention time of the zinc peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
B. Identification Tests—General 〈191〉, Chemical Identification Tests, Acetate
Sample solution: 50 mg/mL
Acceptance criteria: Meets the requirements
3 ASSAY
Change to read:
Procedure
Diluent, Mobile phase, and (IRA 1-Jun-2024) Post-column derivatization reagent (IRA 1-Jun-2024) : Proceed as directed in Zinc Determination 〈591〉, Procedure, Ion Chromatographic Method.
Standard solution: 50 µg/mL of USP Zinc Acetate RS in Diluent prepared as follows. Transfer an appropriate portion of homogenized USP Zinc Acetate RS to a suitable volumetric ask. Add Diluent to about 50% of the nal ask volume to dissolve. Sonication may be needed to aid dissolution. Dilute with Diluent to volume.
Sample solution: 50 µg/mL of Zinc Acetate in Diluent, prepared as directed in the Standard solution
Chromatographic system: Proceed as directed in Zinc Determination 〈591〉, Procedure, Ion Chromatographic Method except for the Columns.
Columns
Guard: 4.0-mm × 5-cm; 9-µm packing L100, or 4.0-mm × 0.5-cm; 4.6-μm packing L91
Analytical: 4.0-mm × 25-cm; 9-µm packing L100, or 4.0-mm × 25-cm; 4.6-μm packing L91
System suitability
Sample: Standard solution
Suitability requirements
Tailing factor: NMT 2.0
Relative standard deviation: NMT 0.73% (IRA 1-Jun-2024)
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zinc acetate dihydrate (C4H6O4Zn · 2H2O) in the portion of Zinc Acetate taken:
Result = (rU /rS ) × (CS /CU ) × 100
rU = peak response of zinc from the Sample solution
rS = peak response of zinc from the Standard solution
CS = concentration of USP Zinc Acetate RS in the Standard solution (µg/mL)
CU = concentration of Zinc Acetate in the Sample solution (µg/mL)
Acceptance criteria: 98.0%–102.0%
4 IMPURITIES
4.1 Chloride and Sulfate 〈221〉, Chloride
Sample: 1.5 g
Acceptance criteria: The Sample shows no more chloride than corresponds to 0.10 mL of 0.020 N hydrochloric acid (0.005%). • Chloride and Sulfate 〈221〉, Sulfate
Sample: 1.0 g
Acceptance criteria: The Sample shows no more sulfate than corresponds to 0.10 mL of 0.020 N sulfuric acid (0.010%). • Arsenic 〈211〉, Procedures, Procedure 1 and Procedure 2, Procedure 1: Colorimetry: NMT 3 ppm
Change to read:
Lead 〈251〉, Procedures, Procedure 1: Chemical Method
Standard solution: 20 mL of 0.5 µg/mL of lead in 1% (v/v) nitric acid
Sample solution: Dissolve 0.5 g of Zinc Acetate in 1 mL of 50% (v/v) nitric acid in a separator. Add 3 mL of Ammonium citrate solution and 0.5 mL of Hydroxylamine hydrochloride solution, and render alkaline with ammonium hydroxide to phenol red TS. Add 10 mL of Potassium cyanide solution, and immediately extract the solution with successive 5-mL portions of Dithizone extraction solution, draining off each extract into another separator, until the last portion of the Dithizone extraction solution retains its green color. Shake the combined extracts for 30 s with 20 mL of 1% (v/v) nitric acid, and discard the chloroform layer.
Analysis: To the Standard solution and Sample solution, add 4 mL of Ammonium cyanide solution (IRA 1-Jun-2024) and 2 drops of Hydroxylamine hydrochloride solution. Add 10.0 mL of Standard dithizone solution, and shake the mixture for 30 s. Pass the chloroform layer through acid-washed filter paper into a color-comparison tube. Compare the color of the Sample solution with that of the Standard solution.
Acceptance criteria: The color of the Sample solution does not exceed the color of the Standard solution (0.002%).
4.2 Alkalies and Alkaline Earths
Sample solution: Dissolve 2.0 g in 150 mL of water in a 200-mL volumetric flask. Add sufficient ammonium sulfide TS to precipitate the zinc completely, and dilute with water to volume. Pass through a dry filter, rejecting the first portion of the filtrate.
Analysis: To 100 mL of the filtrate add 5 drops of sulfuric acid. Evaporate to dryness, and ignite.
Acceptance criteria: The weight of the residue does not exceed 2 mg (0.2%).
5 SPECIFIC TESTS
5.1 Insoluble Matter
Sample: 20 g
Analysis: Dissolve the Sample in 150 mL of water containing 1 mL of glacial acetic acid. Filter any undissolved residue, wash with water, and dry at 105°.
Acceptance criteria: NMT 1.0 mg of insoluble matter (0.005%)
5.2 pH 〈791〉
Sample solution: 50 mg/mL
Acceptance criteria: 6.0–8.0
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in tight containers.
USP Reference Standards 〈11〉
USP Zinc Acetate RS


