Zidovudine
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
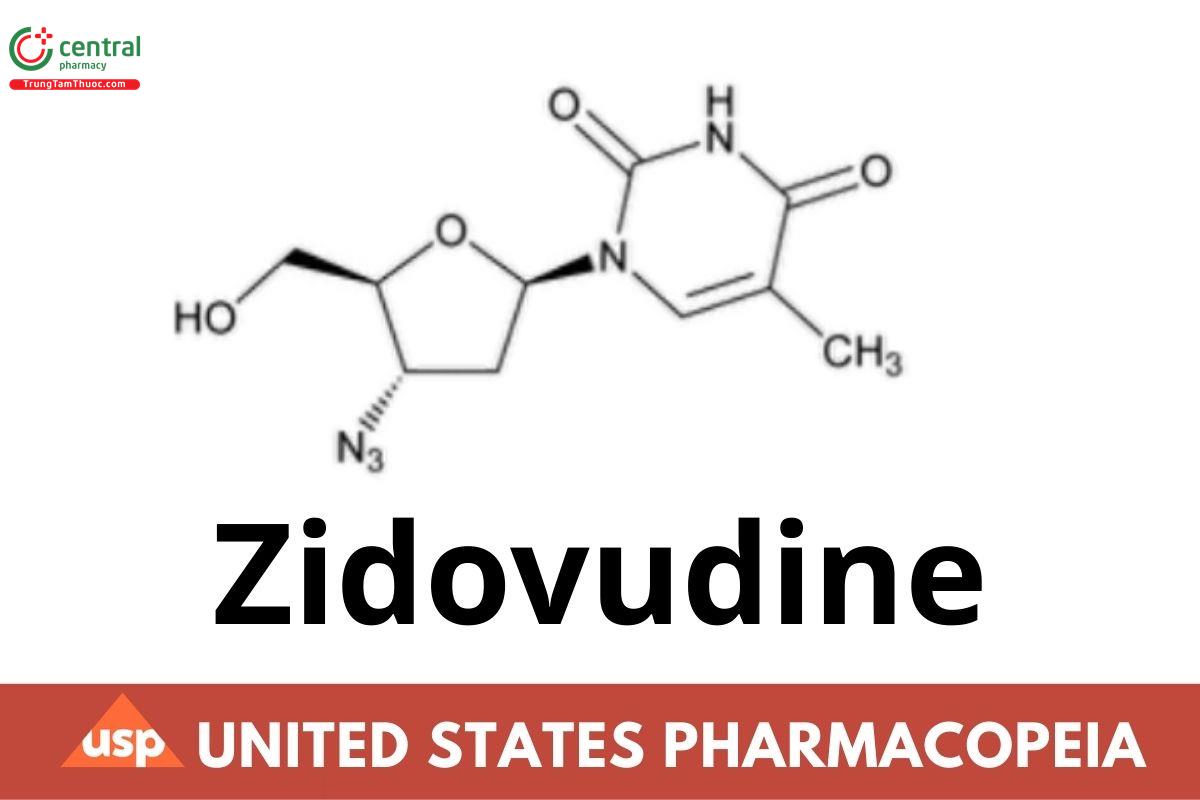
C10H13N5O4 267.24
Thymidine, 3′-azido-3′-deoxy-;
3′-Azido-3′-deoxythymidine CAS RN®: 30516-87-1.
1 DEFINITION
Zidovudine contains NLT 97.0% and NMT 102.0% of zidovudine (C10H13N5O4), calculated on the anhydrous basis.
2 IDENTIFICATION
A. Spectroscopic Identification Tests 〈197〉, Infrared Spectroscopy: 197K
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
Change to read:
Procedure
Buffer: 2 g/L of ammonium acetate in water. Adjust with dilute acetic acid or dilute ammonium hydroxide (1 in 10) to a pH of 6.8.
Solution A: Acetonitrile
Mobile phase: See Table 1.
Table 1
Time (min) | Buffer (%) | Solution A (%) |
0 | 95 | 5 |
3 | 95 | 5 |
18 | 85 | 15 |
28 | 30 | 70 |
43 | 30 | 70 |
45 | 95 | 5 |
Diluent: Acetonitrile, methanol, and Buffer (4:20:76)
Standard solution: 0.2 mg/mL of USP Zidovudine RS in Diluent
Sample solution: 0.2 mg/mL of Zidovudine in Diluent
3.1 Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 265 nm
Column: 4.6-mm × 25-cm; 5-µm packing L1
Flow rate: 1.5 mL/min
Injection volume: 20 µL
3.2 System suitability
Sample: Standard solution
Suitability requirements
Tailing factor: NMT 2.0
Relative standard deviation: NMT 2.0% (USP 1-May-2022)
3.3 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of zidovudine (C10H13N5O4) in the portion of Zidovudine taken:
Result = (rU /rS ) × (CS /CU ) × 100
rU = peak response of zidovudine from the Sample solution
rS = peak response of zidovudine from the Standard solution
CS = concentration of USP Zidovudine RS in the Standard solution (mg/mL)
CU = concentration of Zidovudine in the Sample solution (mg/mL)
Acceptance criteria: 97.0%–102.0% on the anhydrous basis
4 IMPURITIES
Residue on Ignition 〈281〉: NMT 0.25%
Delete the following:
4.1 Limit of Triphenylmethanol and Other Impurities
Standard solution: 0.1 mg/mL each of USP Zidovudine RS and triphenylmethanol in methanol
Sample solution: 20 mg/mL of Zidovudine in methanol
Chromatographic system
(See Chromatography 〈621〉, Thin-Layer Chromatography.)
Mode: TLC
Adsorbent: 0.25-mm layer of chromatographic silica gel mixture containing a fluorescent indicator having an optimal intensity at 254 nm
Application volume: 10 µL
Developing solvent system: Chloroform and methanol (9:1)
Spray reagent: 5 mg/mL of carbazole in alcohol and sulfuric acid (95:5)
Analysis: Develop the chromatogram until the solvent front has moved three-fourths of the length of the plate. Remove the plate from the chamber, mark the solvent front, and allow the solvent to evaporate. Examine the plate under short-wavelength UV light, and compare the intensities of any secondary spots observed in the chromatogram of the Sample solution with those of the principal spot in the chromatogram of the Standard solution.
Acceptance criteria 1: No secondary spot from the chromatogram of the Sample solution is larger or more intense than the principal spot from the Standard solution, and the sum of the intensities of the secondary spots obtained from the Sample solution corresponds to NMT 3.0%.
Spray the plate with Spray reagent, heat for 10 min at 120°, and compare the intensities of any secondary spots observed in the chromatogram of the Sample solution with those of the principal spots in the chromatogram of the Standard solution.
Acceptance criteria 2: No spot corresponding to triphenylmethanol (RF about 2.3 relative to the RF of zidovudine) is more intense than the corresponding spot from the Standard solution. No secondary spot from the chromatogram of the Sample solution is larger or more intense than the principal spot obtained from the Standard solution. The sum of the intensities of the secondary spots from the Sample solution corresponds to NMT 3.0%. (USP 1-May-2022)
Delete the following:
4.2 Limit of Zidovudine Related Compounds B and C
Mobile phase, Standard solution, Sample solution, Chromatographic system, and System suitability: Proceed as directed in the Assay.
Analysis
Sample: Sample solution
Calculate the percentage of zidovudine related compound B and zidovudine related compound C in the portion of Zidovudine taken:
Result = (rU /rT ) × 100
rU = peak response for zidovudine related compound B or zidovudine related compound C
rT = sum of the responses of all of the peaks
Acceptance criteria
Individual impurities: NMT 1.0% for zidovudine related compound B and NMT 2.0% for zidovudine related compound C Total impurities: NMT 3.0% for all impurities from the tests for Limit of Triphenylmethanol and Other Impurities and Limit of Zidovudine Related Compounds B and C. (USP 1-May-2022)
Add the following:
4.3 Organic Impurities
Buffer, Solution A, Mobile phase, Diluent, and Chromatographic system: Proceed as directed in the Assay.
System suitability solution: 1 mg/mL of USP Zidovudine RS and 2 µg/mL of USP Zidovudine Related Compound B RS in Diluent Standard solution: 1 µg/mL of USP Zidovudine RS in Diluent
Sample solution: 1 mg/mL of Zidovudine in Diluent
4.3.1 System suitability
Samples: System suitability solution and Standard solution
[Note—See Table 2 for the relative retention times.]
Suitability requirements
Resolution: NLT 2.0 between zidovudine and zidovudine related compound B, System suitability solution
Relative standard deviation: NMT 5.0%, Standard solution
4.3.2 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of any individual impurity in the portion of Zidovudine taken:
Result = (rU /rS ) × (CS /CU ) × (1/F) × 100
rU = peak response of each impurity from the Sample solution
rS = peak response of zidovudine from the Standard solution
CS = concentration of USP Zidovudine RS in the Standard solution (mg/mL)
CU = concentration of Zidovudine in the Sample solution (mg/mL)
F = relative response factor (see Table 2)
Acceptance criteria: See Table 2. Disregard the triphenylmethanol peak, any peak eluting after triphenylmethanol impurity, and any peak less than 0.05%.
Table 2
Name | Relative Retention Time | Relative Response Factor | Acceptance Criteria, NMT (%) |
Zidovudine related compound Ca | 0.2 | 1.6 | 1.0 |
Stavudineb | 0.5 | 1.0 | 0.3 |
Zidovudine | 1.0 | — | — |
Zidovudine related compound Bc | 1.05 | 1.0 | 1.0 |
Zidovudine related compound G (dimer)d | 1.5 | 1.0 | 0.15 |
Triphenylmethanol | 2.0 | — | —e |
Any individual unspecified impurity | — | — | 0.10 |
Total impuritiesf | — | — | 3.0 |
a Thymine; also known as 5-Methylpyrimidine-2,4(1H,3H)-dione.
b 1-[(2R,5S)-5-(Hydroxymethyl)-2,5-dihydrofuran-2-yl]-5-methylpyrimidine-2,4(1H,3H)-dione.
c 3'-Chloro-3'-deoxythymidine.
d 1-{3-[3-(3-Azido-2,3-dideoxy-β-d-pentofuranosyl)-5-methyl-2,6-dioxo-3,6-dihydropyrimidin-1-yl]-2,3-dideoxy-β-d-pentofuranosyl]-5- methylpyrimidine-2,4-dione.
e See the test for Limit of Triphenylmethanol.
f Includes impurities from the tests for Organic Impurities and Limit of Triphenylmethanol.
(USP 1-May-2022)
Add the following:
4.4 Limit of Triphenylmethanol
Mobile phase: Acetonitrile and water (70:30)
System suitability solution: 0.01 mg/mL of USP Zidovudine RS and 5 µg/mL of USP Triphenylmethanol RS prepared as follows. Transfer a suitable amount of USP Zidovudine RS and USP Triphenylmethanol RS to a suitable volumetric ask and add 10% of the nal ask volume of acetonitrile. Dilute with Mobile phase to volume.
Standard stock solution: 0.5 mg/mL of USP Triphenylmethanol RS in acetonitrile
Standard solution: 5 µg/mL of USP Triphenylmethanol RS in Mobile phase from Standard stock solution
Sample solution: 5 mg/mL of Zidovudine prepared as follows. Transfer a suitable amount of Zidovudine to a suitable volumetric ask and add 10% of the nal ask volume of acetonitrile. Dilute with Mobile phase to volume.
4.4.1 Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 210 nm
Column: 4.6-mm × 15-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
Run time: NLT 10 times the retention time of zidovudine
4.4.2 System suitability
Samples: System suitability solution and Standard solution
Suitability requirements
Resolution: NLT 5.0 between zidovudine and triphenylmethanol, System suitability solution
Relative standard deviation: NMT 5.0%, Standard solution
4.4.3 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of triphenylmethanol in the portion of Zidovudine taken:
Result = (rU /rS ) × (CS /CU ) × 100
rU = peak response of triphenylmethanol from the Sample solution
rS = peak response of triphenylmethanol from the Standard solution
CS = concentration of USP Triphenylmethanol RS in the Standard solution (mg/mL)
CU = concentration of Zidovudine in the Sample solution (mg/mL)
Acceptance criteria: NMT 0.25% (USP 1-May-2022)
5 SPECIFIC TESTS
Water Determination 〈921〉, Method I: NMT 1.0%
Optical Rotation 〈781S〉, Procedures, Specific Rotation
Sample solution: 10 mg/mL, in alcohol
Acceptance criteria: +60.5° to +63°
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in tight, light-resistant containers. Store at 25°, excursions permitted between 15° and 30°. Change to read:
USP Reference Standards 〈11〉
USP Triphenylmethanol RS
Triphenylmethanol.
C19H16O 260.33
(USP 1-May-2022)
USP Zidovudine RS
USP Zidovudine Related Compound B RS
3′-Chloro-3′-deoxythymidine.
C10H13ClN2O4 260.68
10 13 2 4
(USP 1-May-2022)


