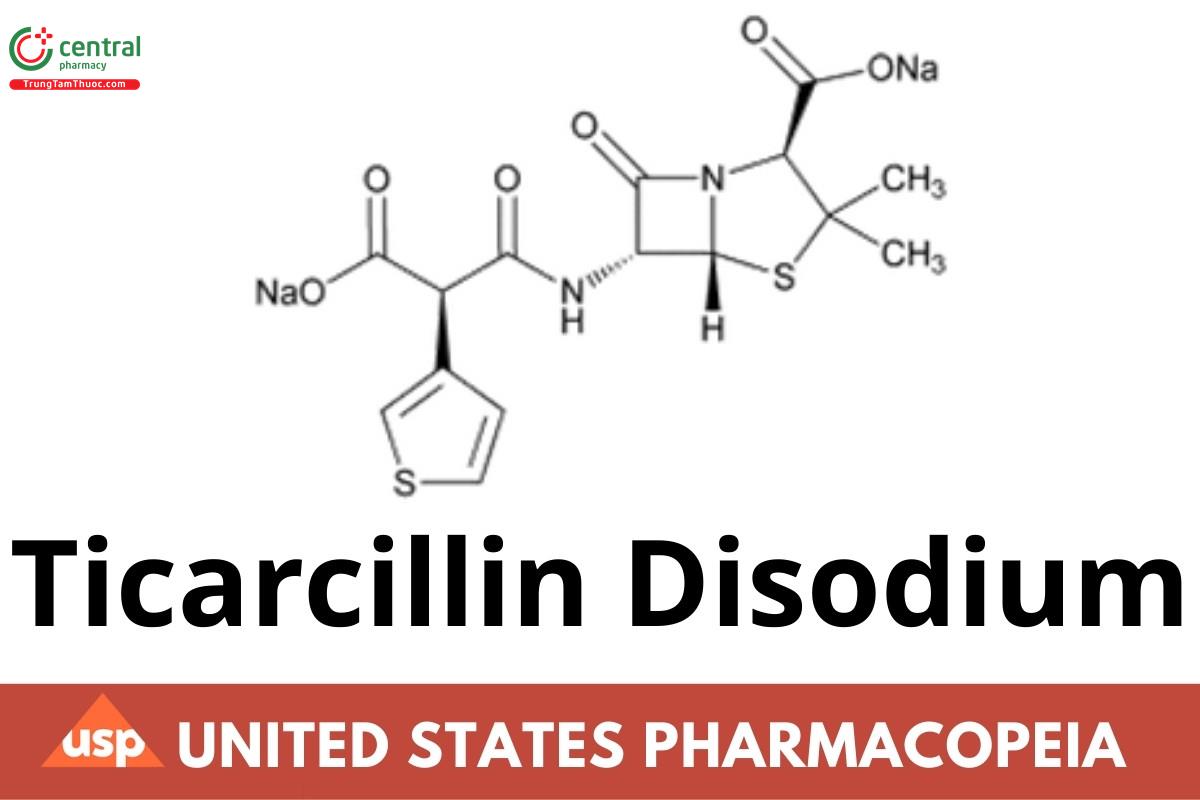
Ticarcillin Disodium
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 DEFINITION
Ticarcillin Disodium has a potency equivalent to NLT 800 µg/mg of ticarcillin (C15H16N2O6S2), calculated on the anhydrous basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy : 197K (CN 1-M -2020)
Solution A: Acetone and water (90:10)
Standard: Dissolve 50 mg of USP Ticarcillin Monosodium Monohydrate RS in 1 mL of water, add 0.1 mL of hydrochloric acid, swirl, and allow to stand in iced water for 10 min. Pass through a filter. Wash the residue with 2 mL of water and dissolve in Solution A. Evaporate almost to dryness, then dry in an oven at 60° for 30 min.
Sample: Dissolve 50 mg of Ticarcillin Disodium in 1 mL of water, add 0.1 mL of hydrochloric acid, swirl, and allow to stand in iced water for 10 min. Pass through a filter. Wash the residue with 2 mL of water and dissolve in Solution A. Evaporate almost to dryness, then dry in an oven at 60° for 30 min.
Acceptance criteria: Meets the requirements
B. Meets the requirements of the test for Optical Rotation, Specific Rotation
C. The retention times of the major peaks of the Sample solution correspond to those of the Standard solution, as obtained in the Assay.
D. IDENTIFICATION TESTS—GENERAL, Sodium〈191〉
Sample solution: 50 mg/mL
Acceptance criteria: Meets the requirements
3 ASSAY
PROCEDURE
Buffer: 1.3 g/L of dibasic ammonium phosphate in water adjusted with phosphoric acid to a pH of 7.0 Mobile phase: Methanol and Buffer (20:80)
Standard solution: 0.1 mg/mL of USP Ticarcillin Monosodium Monohy drate RS in Mobile phase
Sample solution: 0.1 mg/mL of Ticarcillin Disodium in Mobile phase
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 220 nm
Column: 4-mm × 25-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
System suitability
Sample: Standard solution
Suitability requirements
Resolution: NLT 1.5 between the two ticarcillin peaks
Relative standard deviation: NMT 1.0% for the sum of the ticarcillin peaks
Analysis
Samples: Standard solution and Sample solution
Calculate the quantity, in µg/mg, of ticarcillin (C15H16N2O6S2) in the portion of Ticarcillin Disodium taken:
Result = (rU/rS) × (CS/CU) × P
rU = sum of the responses of the two ticarcillin peaks from the Sample solution
rS = sum of the responses of the two ticarcillin peaks from the Standard solution
CS = concentration of USP Ticarcillin Monosodium Monohydrate RS in the Standard solution (mg/mL)
CU = concentration of Ticarcillin Disodium in the Sample solution (mg/mL)
P = potency of ticarcillin in USP Ticarcillin Monosodium Monohydrate RS (µg/mg) Acceptance criteria: NLT 800 µg/mg on the anhydrous basis
4 IMPURITIES
DIMETHYLANILINE 〈223〉: Meets the requirements
ORGANIC IMPURITIES
Solution A: 1.3 g/L of dibasic ammonium phosphate in water adjusted with phosphoric acid to a pH of 7.0 Solution B: Methanol and Solution A (50:50)
Mobile phase: See Table 1.
Table 1
System suitability solution: 0.02 mg/mL of USP Ticarcillin Monosodium Monohydrate RS in Solution A Sample solution: 1 mg/mL of Ticarcillin Disodium in Solution A
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 220 nm
Column: 4-mm × 25-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
System suitability
Sample: System suitability solution
Suitability requirements
Resolution: NLT 2.0 between the two ticarcillin peaks
Relative standard deviation: NMT 5.0% for the sum of the ticarcillin peaks
Analysis
Sample: Sample solution
Calculate the percentage of each impurity in the portion of Ticarcillin Disodium taken:
Result = (rU/rT) × 100
rU = peak response of each impurity from the Sample solution
rT = sum of the responses of the two ticarcillin peaks from the Sample solution
Acceptance criteria: See Table 2.
Table 2
a Decarboxy ticarcillin; (2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[[(thiophen-3-yl)acetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid.
5 SPECIFIC TESTS
OPTICAL ROTATION, Specific Rotation〈781S〉 Sample solution: 10 mg/mL in water Acceptance criteria: +172° to +187°
PH 〈791〉
Sample solution: 10 mg/mL of ticarcillin from Ticarcillin Disodium in water Acceptance criteria: 6.0–8.0
WATER DETERMINATION, Method I〈921〉: NMT 6.0%
STERILITY TESTS 〈71〉: Where the label states that Ticarcillin Disodium is sterile, it meets the requirements in Test for Sterility of the Product to Be Examined, Membrane Filtration.
BACTERIAL ENDOTOXINS TEST 〈85〉: Where the label states that Ticarcillin Disodium is sterile or must be subjected to further processing during the preparation of injectable dosage forms, it contains NMT 0.05 USP Endotoxin Units/mg of ticarcillin.
6 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in tight containers and store in a refrigerator.
LABELING: Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
USP REFERENCE STANDARDS 〈11〉
USP Ticarcillin Monosodium Monohydrate RS C15H15N2NaO6S2 · H2O 424.42


