Tiamulin Fumarate
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
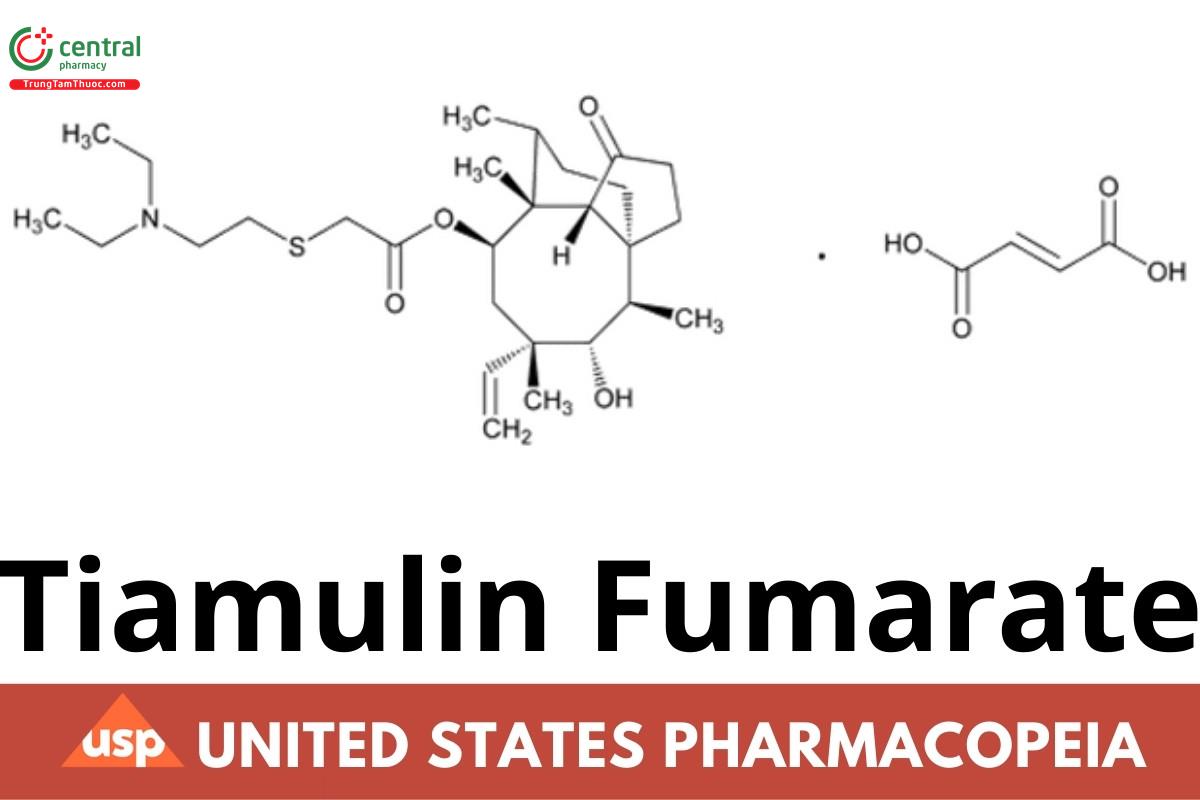
1 DEFINITION
Tiamulin Fumarate contains NLT 97.0% and NMT 102.0% of tiamulin fumarate (C28H47NO4S4 · C4H4O4), calculated on the dried basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy: 197A or 197K[NOTE—When using 197K, intimately mix Tiamulin Fumarate with potassium bromide, but do not grind.] (CN 1-MAY-2020)
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
PROCEDURE
Solution A: 6% perchloric acid
Buffer: Transfer 10 g of ammonium carbonate to a 1000-mL volumetric flask, and dissolve in 800 mL of water. Add 24 mL of Solution A, and dilute with water to volume.
Mobile phase: Methanol, acetonitrile, and Buffer (49:23:28)
System suitability solution: 0.08 mg/mL of USP Tiamulin Fumarate RS and 0.08 mg/mL of USP Tiamulin Related Compound A RS in Mobile phase
Standard solution: 4 mg/mL of USP Tiamulin Fumarate RS in Mobile phase Sample solution: 4 mg/mL of Tiamulin Fumarate in Mobile phase
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 212 nm
Column: 4.6-mm × 25-cm; 5-µm packing L1
Column temperature: 30 ± 3°
Flow rate: 1.2 mL/min
Injection volume: 20 µL
System suitability
Samples: System suitability solution and Standard solution Suitability requirements
[NOTE—The tiamulin related compound A peak elutes before the tiamulin fumarate peak.]
Resolution: NLT 2.0 between tiamulin related compound A and tiamulin fumarate, System suitability solution
Tailing factor: NMT 2.0, Standard solution
Relative standard deviation: NMT 2.0%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of tiamulin fumarate (C28H47NO4S4 · C4H4O4) in the portion of Tiamulin Fumarate taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response from the Sample solution
rS = peak response from the Standard solution
CS = concentration of USP Tiamulin Fumarate RS in the Standard solution (mg/mL)
CU = concentration of Tiamulin Fumarate in the Sample solution (mg/mL)
Acceptance criteria: 97.0%–102.0% on the dried basis
4 OTHER COMPONENTS
CONTENT OF FUMARATE
Sample solution: 450 mg of Tiamulin Fumarate in 60 mL of a mixture of alcohol and water (1:1)
Analysis: Titrate the Sample solution with 0.1 N sodium hydroxide VS, using a glass–calomel electrode (see Titrimetry 〈541〉). Perform a blank determination, and make any necessary correction. Each milliliter of 0.1 N sodium hydroxide is equivalent to 5.8 mg of fumarate.
Acceptance criteria: 83.7–87.3 mg
5 IMPURITIES
RESIDUE ON IGNITION 〈281〉: NMT 0.1%
ORGANIC IMPURITIES
Solution A, Buffer, Mobile phase, System suitability solution, Standard solution, Sample solution, Chromatographic system, and System suitability: Proceed as directed in the Assay.
Analysis
Sample: Sample solution
Calculate the area percentage of each impurity, relative to tiamulin fumarate, in the portion of Tiamulin Fumarate taken:
Result = (rU/rT) × 100
rU = peak response of each impurity from the Sample solution
rT = peak response of tiamulin fumarate from the Sample solution
Acceptance criteria: See Table 1. Disregard the peak due to fumarate at a relative retention time of 0.15.
Table 1
6 SPECIFIC TESTS
OPTICAL ROTATION 〈781S〉 , Procedures, Specific Rotation
Sample solution: 5.0 mg/mL in dioxane
Acceptance criteria: +24° to +28°, on the dried basis, measured at 20°
PH 〈791〉
Sample solution: 10 mg/mL
Acceptance criteria: 3.1–4.1
LOSS ON DRYING 〈731〉
Analysis: Dry under vacuum at 105° for 3 h. Acceptance criteria: NMT 0.5%
CLARITY AND COLOR OF SOLUTION
Sample: 5 g
Analysis: Transfer the Sample to a 100-mL volumetric flask. Dissolve in and dilute with water to volume.
Acceptance criteria: The solution is clear and colorless, and its absorbances determined in a 1-cm cell at 400 and 650 nm are NMT 0.150 and 0.030 absorbance units, respectively.
7 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in tight, light-resistant containers, and store at room temperature.
LABELING: Label it to indicate that it is for veterinary use only.
USP REFERENCE STANDARDS 〈11〉
USP Tiamulin Fumarate RS
USP Tiamulin Related Compound A RS
Tosyl pleuromutilin.


