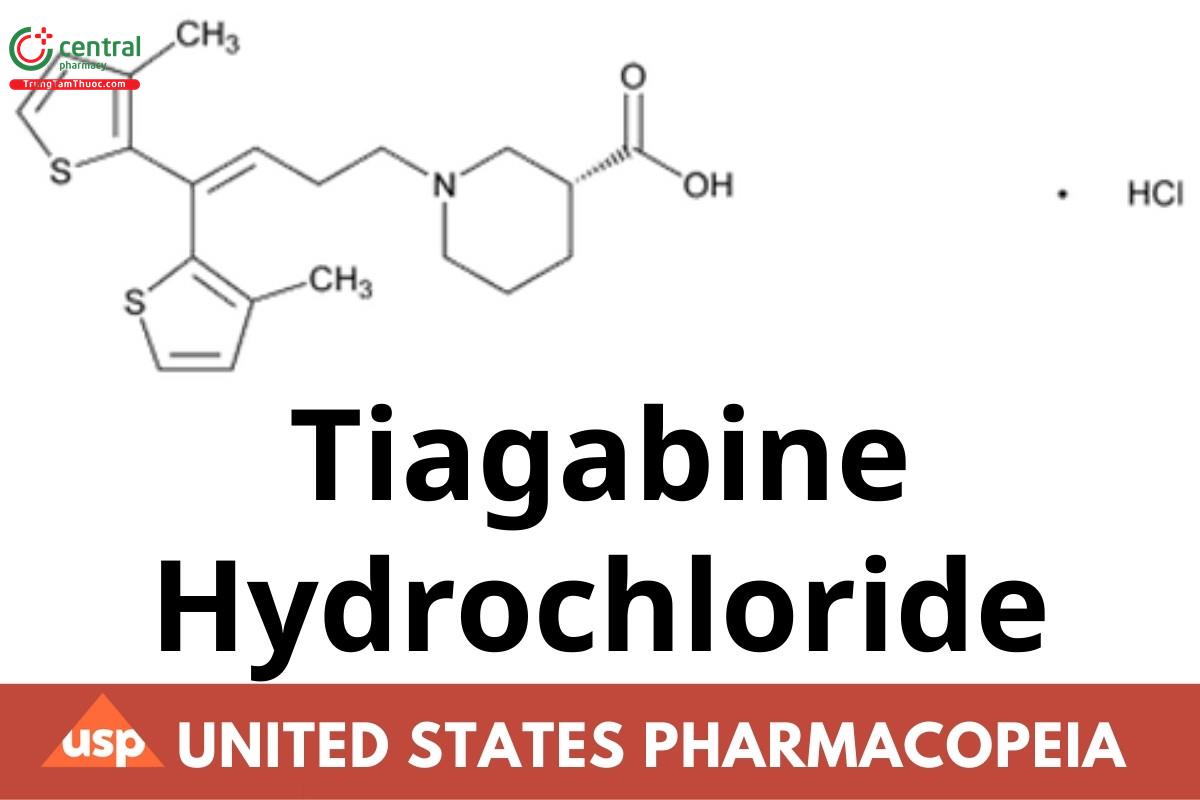
Tiagabine Hydrochloride
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 DEFINITION
Tiagabine Hydrochloride contains NLT 97.5% and NMT 102.5% of tiagabine hydrochloride (C20H25NO2S2 · HCl), calculated on the anhydrous basis.
2 IDENTIFICATION
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy: 197K
Sample: Transfer about 5 mg of Tiagabine Hydrochloride to a test tube. Add 4 mL of 2-propanol, and sonicate to dissolve, if necessary. Evaporate the solvent under inert atmosphere at 50°, using a nitrogen evaporator, for 2 h.
Analysis: The IR spectrum of the Sample corresponds to that of a similarly prepared USP Tiagabine Hydrochloride RS. Acceptance criteria: Meets the requirements
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
PROCEDURE
Solution A: 1.38 g/L of monobasic sodium phosphate. Adjust with phosphoric acid to a pH of 2.0. Mobile phase: Acetonitrile and Solution A (35:65)
Diluent: Methanol and water (50:50)
Internal standard solution: 0.4 mg/mL of butylparaben in Diluent
Standard stock solution: 1 mg/mL of USP Tiagabine Hy drochloride RS in Diluent
Standard solution: 0.1 mg/mL of USP Tiagabine Hydrochloride RS and 0.04 mg/mL of butylparaben in Diluent prepared as follows. Transfer suitable volumes of the Standard stock solution and Internal standard solution into a suitable volumetric flask and dilute with Diluent to volume.
Sample stock solution: 1 mg/mL of Tiagabine Hydrochloride in Diluent
Sample solution: 0.1 mg/mL of Tiagabine Hydrochloride and 0.04 mg/mL of butylparaben in Diluent prepared as follows. Transfer suitable volumes of the Sample stock solution and Internal standard solution into a suitable volumetric flask and dilute with Diluent to volume.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 254 nm
Column: 4.6-mm × 15-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
Run time: NLT 3.5 times the retention time of tiagabine
System suitability
Sample: Standard solution
Suitability requirements
Resolution: NLT 5.5 between tiagabine and butylparaben
Relative standard deviation: NMT 1.5% for the peak response ratios
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of tiagabine hydrochloride (C20H25NO2S2 · HCl) in the portion of Tiagabine Hydrochloride taken:
Result = (RU/RS) × (CS/CU) × 100
RU = peak area ratio of tiagabine hydrochloride to the internal standard from the Sample solution
RS = peak area ratio of tiagabine hydrochloride to the internal standard from the Standard solution
CS = concentration of USP Tiagabine Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Tiagabine Hydrochloride in the Sample solution (mg/mL)
Acceptance criteria: 97.5%–102.5% on the anhydrous basis
4 IMPURITIES
RESIDUE ON IGNITION 〈281〉: NMT 0.2%
LIMIT OF (S)-(+) ISOMER
Mobile phase: n-Hexane, isopropyl alcohol, alcohol, and trifluoroacetic acid (80: 14: 6: 0.5). [NOTE—Increase or decrease the percentage of n- hexane or alcohol, but keep the percentage of isopropyl alcohol constant. Make other adjustments, if necessary (see Chromatography
〈 621〉, System Suitability).]
Standard solution: 0.1 mg/mL of USP Racemic Tiagabine Hydrochloride Mixture RS prepared as follows. Transfer a suitable quantity of USP Racemic Tiagabine Hydrochloride Mixture RS into a suitable volumetric flask, dissolve in a few drops of methanol, and dilute with isopropyl alcohol to volume.
Sample solution: 2 mg/mL of Tiagabine Hydrochloride prepared as follows. Transfer a suitable quantity of Tiagabine Hydrochloride in a suitable volumetric flask, dissolve in a few drops of methanol, and dilute with isopropyl alcohol to volume.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 260 nm
Column: 4.6-mm × 25-cm; 10-µm packing L40
Flow rate: 0.8 mL/min
Injection volume: 10 µL
Run time: NLT 1.5 times the retention of the (R)-(−) isomer
System suitability
Sample: Standard solution
[NOTE—The relative retention times of the (S)-(+) isomer and the (R)-(−) isomer are about 0.76 and 1.0, respectively.]
Suitability requirements
Resolution: NLT 2.0 between the (S)-(+) and (R)-(−) isomers
Analysis
Sample: Sample solution
Calculate the percentage of the (S)-(+) isomer in the portion of Tiagabine Hydrochloride taken:
Result = (rU/rT) × 100
rU = peak response of the (S)-(+) isomer from the Sample solution
rT = sum of the peak responses of the (S)-(+) isomer and (R)-(−) isomer from the Sample solution
Acceptance criteria: NMT 0.5%
ORGANIC IMPURITIES
Solution A: Water adjusted with phosphoric acid to a pH of 2.3 Solution B: Acetonitrile
Mobile phase: See Table 1.
Table 1
Standard stock solution: 1 mg/mL of USP Tiagabine Hydrochloride RS in water
Standard solution: 0.001 mg/mL of USP Tiagabine Hy drochloride RS in water from the Standard stock solution System suitability stock solution: 1 mg/mL of USP Tiagabine Related Compound A RS in water
System suitability solution: 0.1 mg/mL each of USP Tiagabine Hy drochloride RS and USP Tiagabine Related Compound A RS in water prepared from Standard stock solution and System suitability stock solution
Sample solution: 1 mg/mL of Tiagabine Hydrochloride in water
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 254 nm
Column: 4.6-mm × 15-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 20 µL
System suitability
Samples: Standard solution and System suitability solution
Suitability requirements
Resolution: NLT 9.0 between tiagabine hydrochloride and tiagabine related compound A, System suitability solution
Relative standard deviation: NMT 2.0%, Standard solution
Analysis
Sample: Sample solution
Calculate the percentage of each impurity in the portion of Tiagabine Hydrochloride taken:
Result = (rU/rT) × (1/F) × 100
rU = peak response of each impurity from the Sample solution
rT = sum of all the peak responses, excluding the solvent peaks, from the Sample solution
F = relative response factor for each impurity (see Table 2)
Acceptance criteria: See Table 2.
Table 2
a (3R)-1-[3,4-Dihydroxy-4,4-bis(3-methylthiophen-2-yl)butyl]piperidine-3-carboxylic acid.
b (R)-1-[4,4-Bis(3-methylthiophen-2-yl)-3-oxobutyl]piperidine-3-carboxylic acid.
c (R,ZE)-1-[4-(3-Methylthiophen-2-yl)-4-(thiophen-2-yl)but-3-en-1-yl]piperidine-3-carboxylic acid.
d (R)-1-[4-(x-Methylthiophen-2-yl)-4-(y-methylthiophen-2-yl)but-3-en-1-yl]piperidine-3-carboxylic acid; where possible, x,y combinations include (3,4), (4,3), (4,4), (5,5), (4,5), (5,4), (3,5), and (5,3).
e Methyl (R)-1-[4,4-bis(3-methylthiophen-2-yl)but-3-en-1-yl]piperidine-3-carboxylate.
f 4,4-Bis(3-methylthiophen-2-yl)but-3-en-1-ol.
g Bis(3-methylthiophen-2-yl)methanone.
h 4,4-Bis(3-methylthiophen-2-yl)but-3-en-1-yl methanesulfonate.
i 2,2-Bis(3-methylthiophen-2-yl)tetrahydrofuran.
5 SPECIFIC TESTS
WATER DETERMINATION 〈921〉 , Method I: NMT 6.0%
6 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in tight, light-resistant containers. Store at a temperature not higher than 30°. Change to read:
USP REFERENCE STANDARDS 〈11〉
USP Racemic Tiagabine Hydrochloride Mixture RS
(S)-(+),(R)-(−)-1-[4,4-Bis(3-methyl-2-thienyl)-3 butenyl]nipecotic acid, hydrochloride. C20H25NO2S2 · HCl 412.00
USP Tiagabine Hydrochloride RS
(ERR 1-Nov-2021)
USP Tiagabine Related Compound A RS
(R)-Ethyl-1-[4,4-bis(3-methy-2-thienyl)-3-butenyl]-3-piperidinecarboxylate, hydrochloride. C22H29NO2S2 · HCl 440.06


