THE APPLICATION OF MOISTURE VAPOR TRANSMISSION RATES FOR SOLID ORAL DOSAGE FORMS IN PLASTIC PACKAGING SYSTEMS
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
A common cause for failure to meet specifications of packaged solid oral dosage forms (SODFs) is their instability as a result of moisture uptake during storage. Failures are usually related to out-of-specification or out-of-range values in any of the following quality attributes (alone or in combination with other physical properties): assay, related substances, degradation by-products, and dissolution rate. Therefore, the moisture barrier properties of the packaging system must be characterized properly to demonstrate the relative moisture vapor transmission rate (MVTR) performance required to protect SODFs from moisture uptake.
Common packaging systems used for SODFs are plastic bottles that include a plastic closure with a foil seal over the bottle opening, or formed plastic and aluminum blisters, with a foil seal over the blister cavity. Plastic packaging systems are considered to be permeable, and therefore the moisture barrier properties must be characterized.
Containers-Performance Testing (671) includes desiccant-based MVTR methods for SODF packaging systems that align with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) accelerated storage conditions for stability testing [40°/75% relative humidity (RH)] and a water-based method that aligns with the ICH storage condition of 40"/25% RH. These methods have the accuracy to measure the performance of high-barrier packaging systems, which has been traditionally difficult to measure. They provide specific MVTR values for packaging systems with sufficient sensitivity and precision to allow clear differentiation across packaging systems. This chapter provides guidance on the application of these methods to determine MVTR and the use of MVTR data to support demonstrating the equivalence of packaging systems when packaging SODFs.
1.1 Scope
For packaging systems containing more than one unit of product, the concept of normalizing the MVTR of the packaging system to the number of units contained in the system (MVTR/unit) allows for a relative comparison of barrier performance. This ratio can be used to demonstrate equivalent moisture protection, when comparing a variety of packaging systems that contain different numbers of units. Thus, to complement-and in some cases, reduce-product stability testing for SODFs, the use of MVTR/unit can provide evidence that a proposed packaging system may provide adequate moisture protection to maintain product stability. However, annual stability may never be replaced by MVTR studies and changes based upon MVTR studies are made with some risk that stability failures may occur.
This chapter is intended to:
- Describe the methodology of providing evidence for the moisture barrier equivalency of two similar packaging systems (e.g., bottle A to bottle B, blister 1 to blister 2), based on the MVTR measurements described in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems
- Define the upper limit of the MVTR for an SODF
- Compare the desiccant-based and water-based MVTR methods
- Explain how to leverage MVTR data to complement and, possibly, reduce stability studies when a new packaging system, a new packaging configuration for a marketed drug product, or a change to an existing packaging system is introduced
The scope of this chapter is restricted to:
- SODFS
- MVTR based on Method 1, Method 2, and Method 3 (desiccant-based) described in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems
- MVTR based on Method 4 (water-based) described in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems
- Repackaging of SODFs
This chapter is applicable to:
- Pharmaceutical manufacturers
- Packagers
- Repackagers
2 PACKAGING SYSTEMS
Pharmaceutical products are manufactured in different dosage forms and are packaged in a packaging system for storage and distribution. A packaging system performs multiple functions, among which protection is the most fundamental. Such protection ensures that the quality of the packaged drug product is maintained and not affected by physical hazards during the shipping and distribution process or by environmental hazards such as moisture, light, and oxygen throughout the shelf life of the drug product.
For SODFs, the most commonly used packaging systems are plastic bottles and blisters. A number of different plastic materials are used to manufacture SODF packaging, including cyclic olefins, polyethylene (PE), polyethylene terephthalate (PET), polyethylene terephthalate G (PETG), polypropylene (PP), and polyvinyl chloride (PVC). A general chemical description of these plastics is contained in Evaluation of Plastic Packaging Systems and Their Materials of Construction with Respect to Their User Safety Impact (1661), and the specifications for use in oral dosage packaging systems are defined in Plastic Materials of Construction (661.1). Two other plastic materials used to form blisters are polyvinylidene chloride (PVDC) and polychlorotrifluoroethylene (PCTFE).
Chapter (671) classifies these packaging systems into three main categories: multiple-unit containers, single-unit containers, and unit-dose containers as defined in Packaging and Storage Requirements (659). In the context of this chapter, packaging systems for SODFs are categorized as:
1. Multiple-unit container. a sealed plastic bottle that contains more than one unit of drug product. An example of a multiple-unit container is a high-density polyethylene (HDPE) bottle containing 30 tablets, Method 1 or Method 4 in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems should be used to determine the MVTR of the multiple-unit container.
2. Single-unit or unit-dose container: A sealed blister card that contains 1 or more blister cavities and 1 unit of drug product per blister cavity. An example of such a container is a sealed PVDC blister card containing 7 cavities. Method 2 or Method 4 in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems should be used to determine the MVTR of the container unless the container is a low-barrier blister (see Glossary), in which case Method 3 or Method 4 in Containers-Performance Testing (671), Moisture Vapor Transmission for Plastic Packaging Systems should be followed.
3 DESICCANT VERSUS WATER MVTR METHOD
Traditionally, the MVTR of container-closure systems for solid oral drug products has been determined by using desiccant-filled test containers stored in an environmental chamber to create a difference in RH or vapor pressure between the inside and outside of the test containers. The use of desiccants can be problematic because an inadequate amount of desiccant or incompletely dried desiccant may result in variability in the vapor-pressure differences between the inside and outside of the container during the study. This potential variability in the vapor-pressure difference can increase variability in MVTR determinations.
3.1 Use of Desiccants for MVTR Determination
According to MVTR Method 1, Method 2, and Method 3 described in (671), test containers are filled with sufficient anhydrous desiccant to maintain an inside RH close to 0%. This RH is maintained if the prescribed desiccant preparations are handled properly. However, mishandling of the desiccant during shipping to the filling site or during filling of the blisters will compromise the dryness of the desiccant. Desiccant-filled test containers are held in an environmental chamber under controlled environmental conditions, and weight determinations are made over time. The inside RH remains constant during testing if adequate anhydrous desiccant is present, which becomes more challenging to achieve with a low-barrier blister (Method 3) because the desiccant can become saturated before the completion of testing. The weight gain of the desiccant is determined during this steady-state condition and is translated into a permeation rate MVTR (i.e., water vapor) for the test packaging system.
Percent relative humidity (%RH) is used to express the environmental chamber conditions and the headspace inside the test containers. To obtain reliable MVTR determinations, humidity must remain constant inside and outside the test container throughout the study. Results from desiccant-filled test containers do not account for potential variability introduced if the internal RH of the test containers neither starts at 0% nor remains at 0% during the study. The desiccant methods are designed to maintain the internal RH below 10% throughout the study. However, this internal RH can range between 0% and 10%, which can translate to the variability of the internal RH. In test containers filled with water, the RH is maintained at a constant (100%) from the beginning to the end of the MVTR study, thereby reducing potential variability.
3.2 Use of Water as an Alternative Fill Medium
Method 4, as described in (671), is an alternative approach for the determination of MVTR for packaging systems for SODFs. While desiccant-filled test containers stored in a 40°/75% RH chamber achieve a vapor-pressure difference of 65%-75% if the internal RH created by the desiccant is between 0% and 10%, water-filled test containers stored in an environmental chamber at 40°/25% RH achieve and maintain a constant vapor-pressure difference of 75%.
There are potentially multiple benefits of the MVTR method using water as the test medium. With water, the variability of the fill amount does not result in variability of internal RH, and no interaction with container walls is known to exist for the common blister plastic and lidding materials. The stability of the internal RH afforded by water facilitates holding the samples or reusing them at a later time, or in multiple studies (e.g., different storage conditions using the same samples). The concerns about shipping desiccant-filled samples and delays in testing are mitigated by the use of water. Additional measurements or extensions of studies are possible without concern about depleting the water.
The use of water eliminates special handling requirements to protect desiccant-filled blister samples from excessive exposure to moisture before testing. Also, samples filled with aqueous dye can be used for leak testing before or after MVTR testing, which can enhance visual detection of gross leaks during the study.
Sample preparation techniques are important for filling blisters with water. The best practice for delivering water into the cavities on the packaging line is to use a manual syringe filled with water, this method allows the delivery of a few drops of water to each cavity without causing sealing problems. However, there are concerns about introducing an inadequate amount of water. Chapter (671) describes how to estimate the amount of water to avoid the total loss of water before the completion of the test. In contrast to the desiccant method in which the saturation of desiccant is a concern, the water-filled method allows low-barrier blisters to be tested over an extended period in the same manner as high-barrier blisters.
Table 1 summarizes recommended test methods to measure the MVTR of different packaging systems. Method 3 in (671) is not recommended for measuring the MVTR of low-barrier blisters due to the rationales discussed earlier in this chapter.
Table 1. Recommended MVTR Test Methods for Different Packaging Systems
| Packaging System | Test Method (recommended) |
| Low-barrier blister | 〈671〉, Method 4 |
| High-barrier blister | 〈671〉, Method 2 or Method 4 |
| Ultra-high barrier blister | 〈671〉, Method 2 or Method 4 |
| Bottle containers (e.g., multiple-unit containers) | 〈671〉, Method 1 or Method 4 |
3.2.1 COMPARISON OF MVTR RESULTS FOR WATER AND DESICCANT METHOD
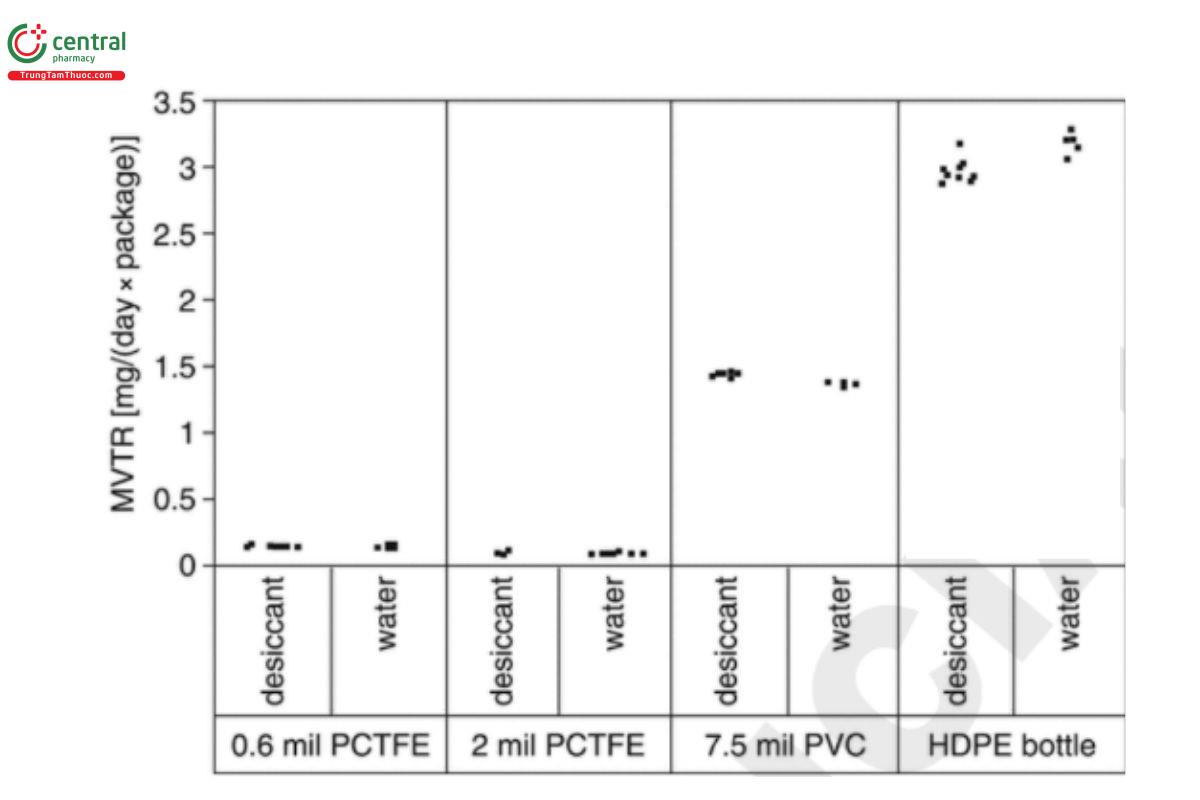
Several sets of data are provided in Table 2 (1). The user should use an appropriate statistical tool to analyze the data and prove the equivalency from the statistical perspective. The raw data for each set are shown graphically in Figure 1.
Table 2. Summary of MVTR Studies Comparing Desiccant-Filled and Water-Filled Samples
| Sample Description | Fill Medium | Chamber Conditions | Number of Test Containers | Average MVTR |
| 7.5 mil PVC | Molecular sieves | 40°/75% RH | 10 | 1.46 mg/(day × cavity) |
| 7.5 mil PVC | Water | 40°/25% RH | 5 | 1.39 mg/(day × cavity) |
| 0.6 mil PCTFE | Calcium chloride | 40°/75% RH | 10 | 0.161 mg/(day × cavity) |
| 0.6 mil PCTFE | Water | 40°/25% RH | 6 | 0.162 mg/(day × cavity) |
| 2 mil PCTFE | Molecular sieves | 40°/75% RH | 10 | 0.104 mg/(day × cavity) |
| 2 mil PCTFE | Water | 40°/25% RH | 10 | 0.105 mg/(day × cavity) |
| 325-mL HISᵃ HDPE bottle | Calcium chloride | 40°/75% RH | 10 | 2.97 mg/(day × package) |
| 325-mL HISᵃ HDPE bottle | Water | 40°/25% RH | 5 | 3.20 mg/(day × package) |
ᵃ Heat induction sealed
Change to read:
4 EQUIVALENCY AND APPLICATION OF MVTR DETERMINATION METHOD
4.1 Description
For SODF packaging systems containing more than one unit of product, the concept of normalizing the MVTR of the packaging system to the number of SODF units contained in the system allows for the comparison of the barrier performance and the demonstration of equivalent moisture protection for each SODF unit when comparing various packaging systems containing varying numbers of SODF units.
4.2 Rationale
The moisture permeability of a packaging system can be a product Critical Quality Attribute (CQA) for SODFs. Once the stability performance has been assessed and suitability demonstrated in one or more packaging systems, a maximum demonstrated level of moisture permeability of the packaging systems, defined by MVTR normalized to the number of dosage units in the container (MVTR/unit), can be established. The MVTR/unit links the performance of the packaging system to the quality of the product in the container. Having established the maximum demonstrated level of MVTR/unit for a given SODF, and having demonstrated the suitability of one or more packaging systems through stability testing and/or other means, the MVTR/unit may be used as a measure of moisture protection to support changes to the packaging systems.
Use of a proper moisture permeation test method and a product-centric measurement of MVTR/unit for blisters and bottles permits the pooling of performance data from both packaging systems to establish a maximum demonstrated level for moisture permeability as a product CQA and accompanies stability study data to establish drug product quality attributes. For the pharmaceutical manufacturer, the product-centric model enables and facilitates a scientifically justified determination of the maximum demonstrated level of MVTR/unit unique to the product. This presents the ability to demonstrate that the new packaging system provides the equivalent or better MVTR/unit for a specific product, with the potential of not having to wait for the results of stability testing, providing that suitability of the bottle or blister packaging system is demonstrated through other means [such as compliance with the appropriate Code of Federal Regulations (CFR) and USP tests) and the packaging systems have previously been used in a regulatory-approved SODF.
4.3 Application
By utilizing MVTR/unit and product-specific stability studies, scientifically justified limits for packaging moisture permeability can be defined. The moisture permeation test results and the maximum demonstrated level derived from those tests should be discussed in the Pharmaceutical Development section of the regulatory submission to support the selection of the packaging systems and materials of construction. It is also important to indicate the control strategy and the steps that will be taken to ensure that the packaging system attributes, and specifically the MVTR, will be met in day-to-day production. Moisture permeation through a material is affected by the properties of the material and its thickness. The MVTR can be controlled through acceptance specifications to confirm material identity and the weight or thickness of the material, and in-process control checks to confirm seal quality.
A maximum demonstrated level for moisture permeability of packaging systems can be ascertained based on the MVTR/unit for each package configuration and the acceptable product stability performance. Consider the following hypothetical example for SODF product X, in which MVTR and stability testing were performed on unit-dose blisters, using two different blister films, and an HDPE packaging system with a fill count of 30 tablets. In this example, the dissolution rate is a drug product CQA, and a decrease in the dissolution rate caused by moisture ingress was observed on stability.
Table 3. MVTR for Different Packaging Systems (tested according to (671))
| Packaging System | MVTR/Container (mg/day) | MVTR/Unit (mg/day) | Drug Product Dissolution |
| 250-µm PVC unit-dose blister | 3.1 | 3.1 | Fail |
| 250-µm PVC / 60-g/m² PVDC unit-dose blister | 0.8 | 0.8 | Pass |
| 60-cc HDPE bottle, 30-count | 1.2 | 0.04 | Pass |
In Table 3 (2), PVC blisters with an MVTR of 3.1 mg/day/tablet were unable to provide adequate protection against moisture ingress, and the product failed to meet the specification for dissolution rate. However, the use of a higher-barrier PVDC-coated PVC blister packaging or a 30-count 60-cc HDPE bottle, with MVTR values of 0.8 mg/day/tablet and 0.04 mg/day/tablet, respectively (Table 3) (2), did provide adequate protection. A maximum demonstrated moisture permeability of ≤0.8 mg (ERR 1-Dec-2020)/day/tablet was established.
The maximum demonstrated level of MVTR may be leveraged in order to introduce new packaging presentations without the need of specific drug product stability testing prior to implementation, MVTR is one of many factors in assessing packaging system change including introducing new packaging configuration (e.g., HDPE bottle with a new count), the composition of the container-closure system, product requirements, and stability. However, MVTR may be useful in a product development setting, to decide on the packaging components before packaging exhibit batches, or in a pharmacy setting where the product is being repackaged.
For example, after launch, a commercial need may arise for a bottle containing a weekly dose of 7 tablets or a bottle containing 90 tablets. These two presentations may be considered based on the data shown in Table 3 (2) and Table 4 (2).
Table 4. MVTR for Different Bottle Sizes and Counts (Tested According to (671))
| Packaging System | MVTR/Container (mg/day) | MVTR/Unit (mg/day) | Maximum Demonstrated MVTR/Unit (mg/day) |
| 40-cc HDPE bottle, 7-count | 0.95 | 0.14 | 0.8 |
| 100-cc HDPE bottle, 90-count | 1.7 | 0.019 | 0.8 |
5 REPACKAGING OF SODFS
5.1 Repackaging an SODF into a Unit-Dose Container
Multiple-unit packages of SODFs (e.g., unit-of-use or bulk packaging systems) may be reprocessed or repackaged as instructed by the manufacturer. A blister package may not be reprocessed or repackaged after it has been deblistered from a unit-dose container. If the manufacturer's labeling includes "Protect from Moisture", the repackager must use a high-barrier or an ultra-high barrier film. If light protection is required for the SODF, the repackager should follow the requirements for spectral transmission established in (671). This testing should be conducted on the formed blister because the light-protective properties of the film are compromised once the film is thinned during the forming process. It is recommended that these tests, in conjunction with any guidance provided by the manufacturer, be considered for any packaging system used to repackage an SODF. The packaging system used to repackage an SODF must be at least as protective or provide better protection than the packaging system used by the original manufacturer of SODFs, taking into consideration the planned shelf life of the repackaged drug.
In the event that a product should not be repackaged, the manufacturer may state this in the labeling.
5.2 Determination of Equivalence for Packaging Systems for SODFS
The suitability of a repackaging system for an SODF may be characterized by establishing equivalency compared to the original packaging system. Equivalence is established when the repackaging system is at least as protective as the original packaging system in terms of MVTR, oxygen transmission, light transmission, and compatibility of the materials of construction. Packaging system equivalency extends to any special protective materials, such as those used for light protection; seals; auxiliary packaging components, such as fillers (see Auxiliary Packaging Components (670)); and desiccants and their canister or sachet, which are associated with the original packaging system (see Packaging and Storage Requirements (659). General Definitions. Packaging Definitions. Equivalent container-closure system).
For moisture, the determination of MVTR should be made using a method provided in (671). (Method 1, Method 2, or Method 4) for both the original packaging system and the packaging system used to repackage the SODF to provide evidence of equivalence.
6 GLOSSARY
Blister: Formed, lidded, and sealed plastic or foil dome that contains the capsule or tablet (usually a single-unit or unit-dose).
Blister card: A contiguous group of blisters formed and sealed with lid in place. The number of blisters per card commonly ranges from 1 to 10 but may be more. The blister card may sometimes be referred to as a packaging system.
High-barrier blister: A blister whose moisture vapor transmission rate, when tested at 40" and an RH difference of 75% between inside and outside the sealed blister, is less than 1.0 mg/cavity-day, but greater than 0.01 mg/cavity-day. An example of a high-barrier blister is a thermoformed PCTFE blister sealed with foil.
Low-barrier blister: A blister whose moisture vapor transmission rate, when tested at 40° and an RH difference of 75% between inside and outside the sealed blister, is greater than 1.0 mg/cavity-day. An example of a low barrier blister is a thermoformed PVC blister sealed with foil.
Ultra-high barrier blister: A blister whose moisture vapor transmission rate, when tested at 40° and an RH difference of 75% between inside and outside the sealed blister, is less than 0.01 mg/cavity-day. An example of an ultra-high barrier blister is a cold-formed foil blister sealed with foil.
Cavity: Formed, lidded, and sealed plastic or foil dome (see Blister).
Equivalency: A claim that two container-closure packaging systems for a particular dose of an SODF are suitable for maintaining the stability of the drug in accordance with its specifications. In this chapter, equivalency refers to equivalent packaging systems from a moisture barrier perspective.
Equivalent container-closure system: See Packaging and Storage Requirements (659), General Definitions, Packaging Definitions, Equivalent container-closure system.
Moisture vapor transmission rate: The steady-state moisture vapor transmission (in unit time) through a packaging system, under specific conditions of temperature and humidity. These test methods use gravimetric measurement to determine the rate of weight gain as a result of moisture vapor transmission into the packaging system and subsequent uptake by a desiccant enclosed within the packaging system or the rate of weight loss as a result of moisture vapor transmission out of the packaging system and subsequent loss of water enclosed within the packaging system.
Test specimen (or specimen): For multiple-unit containers, the bottle is the test specimen; and for single-unit or unit-dose containers, the blister card containing multiple blister cavities is the test specimen. For blisters, more than one card may be bundled to form a test specimen.
Test unit: For multiple-unit containers, the bottle is the test unit as well as being the test specimen; and for single-unit or unit-dose containers, the test unit is a group of test specimens (blister cards) processed together for temperature and humidity exposure and for weighing at each time point. The purpose of the test unit for single-unit or unit-dose containers is to gain the advantage of additive weight gain resulting from more blister cavities than are present on a single card. The test unit, when applied to bottles, is used to maintain congruence of naming among the three test methods.
7 REFERENCES
1. Sparks DL, Yoon S. Containers-Performance Testing (671): Use of water-filled test containers for water vapor transmission rate determinations. Pharm Forum. 2015;41(1).
2. Bergum J, Chen Y, Hunt D, Lockhart H, Malinowski D, Moreton C, O'Reilly D, and Wiltulski F. Development and application of MVTR/unit data in regulatory submissions. Pharm Forum. 2013;39(6)


