Tetracycline
If you find any inaccurate information, please let us know by providing your feedback here
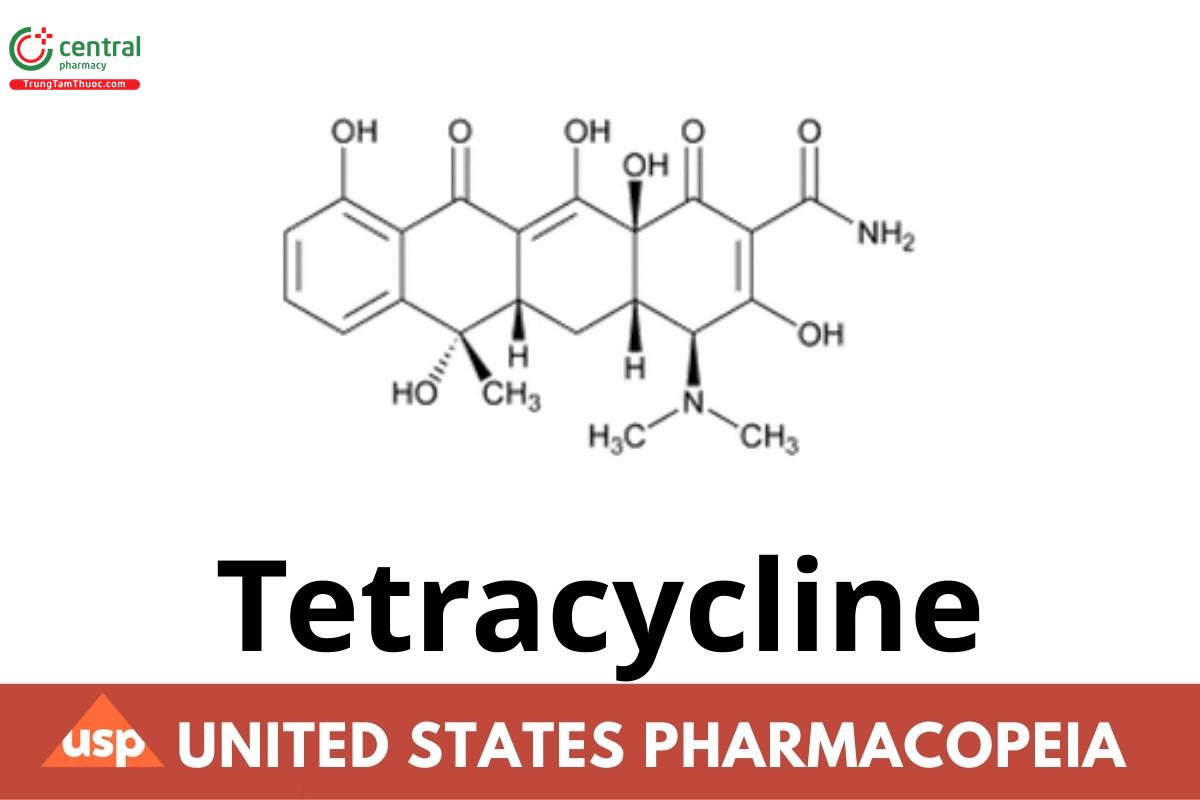
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 DEFINITION
Tetracycline has a potency equivalent to NLT 975 µg/mg of tetracycline hydrochloride (C22H24N2O8 · HCl), calculated on the anhydrous basis.
2 IDENTIFICATION
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Ultraviolet-Visible Spectroscopy: 197U Sample solution: 20 µg/mL in 0.25 N sodium hydroxide
Analytical wavelength: 380 nm
Analysis: Measure the absorptivity 6 min after preparation.
Acceptance criteria: Absorptivity, calculated on the anhydrous basis and taking into account the potency of the Reference Standard, is between 104.5% and 111.95% of the absorptivity of USP Tetracy cline Hy drochloride RS.
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
PROCEDURE
Solution A: Dilute 1 mL of phosphoric acid with water to 1 L.
Solution B: Acetonitrile
Mobile phase: See Table 1.
Table 1
System suitability solution: 25 µg/mL each of USP Anhy drotetracy cline Hy drochloride RS, USP Epitetracy cline Hy drochloride RS, and USP 4- Epianhydrotetracycline Hydrochloride RS, and 100 µg/mL of USP Tetracycline Hydrochloride RS in Solution A
Standard solution: 100 µg/mL of USP Tetracy cline Hy drochloride RS in Solution A
Sample solution: 90 µg/mL of Tetracycline in Solution A Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 280 nm
Column: 4.6-mm × 15-cm; 3-µm packing L60. [NOTE—Column with L1 packing is also suitable.] Temperatures
Autosampler: 10°
Column: 50°
Flow rate: 1.0 mL/min
Injection volume: 10 µL
System suitability
Samples: System suitability solution and Standard solution
Suitability requirements
Resolution: NLT 2.5 between epitetracycline and tetracycline; NLT 2.5 between anhydrotetracycline and 4-epianhydrotetracycline, System suitability solution
Tailing factor: NMT 1.5, Standard solution
Relative standard deviation: NMT 0.73%, Standard solution Analysis
Samples: Standard solution and Sample solution
Calculate the potency equivalent, in µg/mg, of tetracycline hydrochloride (C22H24N2O8 · HCl) in the portion of Tetracycline taken:
Result = (rU/rS) × (CS/CU) × P
rU = peak response of tetracycline from the Sample solution
rS = peak response of tetracycline from the Standard solution
CS = concentration of USP Tetracy cline Hy drochloride RS in the Standard solution (µg/mL)
CU = concentration of Tetracycline in the Sample solution (µg/mL)
P = potency of USP Tetracycline Hydrochloride RS (µg/mg) Acceptance criteria: NLT 975 µg/mg on the anhydrous basis
4 IMPURITIES
Change to read:
ORGANIC IMPURITIES
Solution A, Solution B, Mobile phase, Sample solution, and Chromatographic system: Proceed as directed in the Assay.
System suitability solution: 25 µg/mL each of USP Anhydrotetracycline Hydrochloride RS, USP Epitetracycline Hydrochloride RS, USP 4- Epianhy drotetracy cline Hy drochloride RS, and USP Tetracy cline Hy drochloride RS in Solution A
Standard solution 1: 0.5 µg/mL of USP Anhy drotetracy cline Hy drochloride RS and 3 µg/mL of USP Epitetracy cline Hy drochloride RS in Solution A
Standard solution 2: 2 µg/mL of USP 4-Epianhy drotetracy cline Hy drochloride RS in Solution A
Standard solution 3: 0.1 µg/mL of USP Tetracy cline Hy drochloride RS in Solution A (IRA 1-May-2022)
System suitability
Samples: System suitability solution, Standard solution 1, Standard solution 2, and Standard solution 3 (IRA 1-May-2022)
Suitability requirements
Resolution: NLT 2.5 between epitetracycline and tetracycline; NLT 2.5 between anhydrotetracycline and 4-epianhydrotetracycline, System suitability solution
Relative standard deviation: NMT 2.8% for anhydrotetracycline and epitetracycline, Standard solution 1; NMT 2.8% for 4- epianhydrotetracycline, Standard solution 2; NMT 2.8% for tetracycline, Standard solution 3▲ (IRA 1-May-2022)
Analysis
Samples: Standard solution 1, Standard solution 2, Standard solution 3, (IRA 1-May-2022) and Sample solution
Calculate the percentage of anhydrotetracycline hydrochloride and epitetracycline hydrochloride in the portion of Tetracycline taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of anhydrotetracycline or epitetracycline from the Sample solution
rS = peak response of the corresponding USP Reference Standard from Standard solution 1
CS = concentration of the corresponding USP Reference Standard in Standard solution 1 (µg/mL)
CU = concentration of Tetracycline in the Sample solution (µg/mL)
Calculate the percentage of 4-epianhydrotetracycline hydrochloride in the portion of Tetracycline taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of 4-epianhydrotetracycline from the Sample solution
rS = peak response of 4-epianhydrotetracycline from Standard solution 2
CS = concentration of the USP 4-Epianhy drotetracy cline Hy drochloride RS in Standard solution 2 (µg/mL)
CU = concentration of Tetracycline in the Sample solution (µg/mL) (IRA 1-May-2022)
Calculate the percentage of 2-acetyl analog or any unspecified impurity in the portion of Tetracycline taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × 100
rU = peak response of 2-acetyl analog or any unspecified impurity from the Sample solution
rS = peak response of tetracycline from Standard solution 3 (IRA 1-May-2022)
CS = concentration of USP Tetracycline Hydrochloride RS in Standard solution 3 (IRA 1-May-2022) (µg/mL)
CU = concentration of Tetracycline in the Sample solution (µg/mL)
Mr1 = molecular weight of tetracycline, 444.44
Mr2 = molecular weight of tetracycline hydrochloride, 480.90
Acceptance criteria: See Table 2. The reporting threshold is (IRA 1-May-2022) 0.05%.
Table 2
a 2-Acetyl-2-decarbamoyltetracycline; also known as (4S,4aS,5aS,6S,12aS)-2-Acetyl-4-(dimethylamino)-3,6,10,12,12a-pentahydroxy-6- methyl-4a,5a,6,12a-tetrahydrotetracene-1,11(4H,5H)-dione.
5 SPECIFIC TESTS
OPTICAL ROTATION 〈781S〉 , Procedures, Specific Rotation
Sample solution: 5 mg/mL of tetracycline in 0.1 N hydrochloric acid Acceptance criteria: −260° to −280° on the anhydrous basis
CRYSTALLINITY 〈695〉: Meets the requirements
PH 〈791〉
Sample solution: Prepare in an aqueous suspension (1 in 100). Acceptance criteria: 3.0–7.0
WATER DETERMINATION 〈921〉 , Method I: NMT 13.0%
6 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in tight, light-resistant containers.
LABELING: Label it to indicate that it is to be used in the manufacture of nonparenteral drugs only. Change to read:
USP REFERENCE STANDARDS 〈11〉
USP Anhydrotetracycline Hydrochloride RS
(4S,4aS,12aS)-4-(Dimethylamino)-3,10,11,12a-tetrahydroxy-6-methyl-1,12-dioxo-1,4,4a,5,12,12a-hexahydrotetracene-2-carboxamide monohydrochloride.
C22H22N2O7 · HCl 462.88 (IRA 1-May-2022)
USP 4-Epianhydrotetracycline Hydrochloride RS
(4R,4aS,12aS)-4-(Dimethylamino)-3,10,11,12a-tetrahydroxy-6-methyl-1,12-dioxo-1,4,4a,5,12,12a-hexahydrotetracene-2-carboxamide hydrochloride.
C22H22N2O7 · HCl 462.88 (IRA 1-May-2022)
USP Epitetracycline Hydrochloride RS
(4R,4aS,5aS,6S,12aS)-4-(Dimethylamino)-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2- carboxamide monohydrochloride.
C22H24N2O8 · HCl 480.90 (IRA 1-May-2022)
USP Tetracycline Hydrochloride RS


