Sulisobenzone
If you find any inaccurate information, please let us know by providing your feedback here
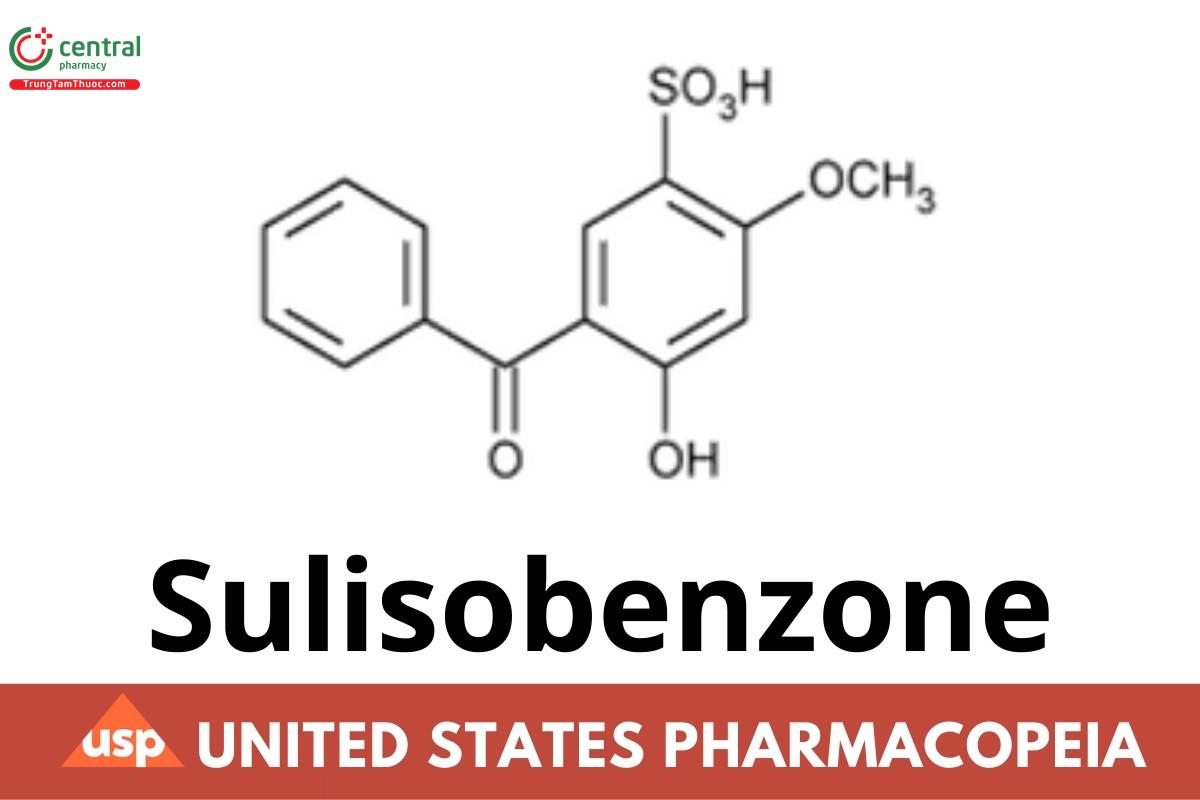
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
Sulisobenzone contains not less than 97.0 percent and not more than 103.0 percent of C14H12O6S, calculated on the anhydrous basis.
Packaging and storage—Preserve in tight, light-resistant containers.
USP REFERENCE STANDARDS 〈11〉—
USP Sulisobenzone RS
Change to read:
SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Ultraviolet-Visible Spectroscopy: 197U (CN 1-May-2020) — Solution: 10 µg per mL.
Medium: water.
Absorptivities, calculated on the anhydrous basis, do not differ by more than 3.0%. WATER DETERMINATION, Method I 〈921〉: not more than 2.0%.
Assay—Accurately weigh about 0.25 g of Sulisobenzone, and dissolve in 5 mL of water in a conical flask. Add 50 mL of dehydrated isopropyl alcohol, titrate with 0.1 N tetrabutylammonium hydroxide VS, and determine the two endpoints potentiometrically. V1 and V2 are the volumes, in mL, of titrant needed to reach the first and second endpoints, respectively. To determine the blank value caused by tertiary amines present as impurities in the titrant, dissolve V1/5 mL of 0.5 N hydrochloric acid VS in 50 mL of dehydrated isopropyl alcohol, and subsequently add (5 − V1/5) mL of water. Titrate with 0.1 N tetrabutylammonium hydroxide VS, and determine the two endpoints potentiometrically. V1B and V2B are the volumes, in mL, of titrant needed to reach the first and second endpoints, respectively. The blank value VBV , in mL, is obtained by the formula:
V2B − V1B
Calculate the percentage of C14H12O6S in the portion of Sulisobenzone taken by the formula:
308.31C[2V1 − (V2 − VBV)](1/W)(0.001)100
in which 308.31 is the molecular weight of Sulisobenzone; C is the concentration, in moles per L, of the 0.1 N tetrabutylammonium hydroxide VS; W is the weight, in g, of Sulisobenzone taken; 0.001 is the conversion factor for converting mL to L; and 100 is the conversion factor to percentage.


