Sulindac
If you find any inaccurate information, please let us know by providing your feedback here
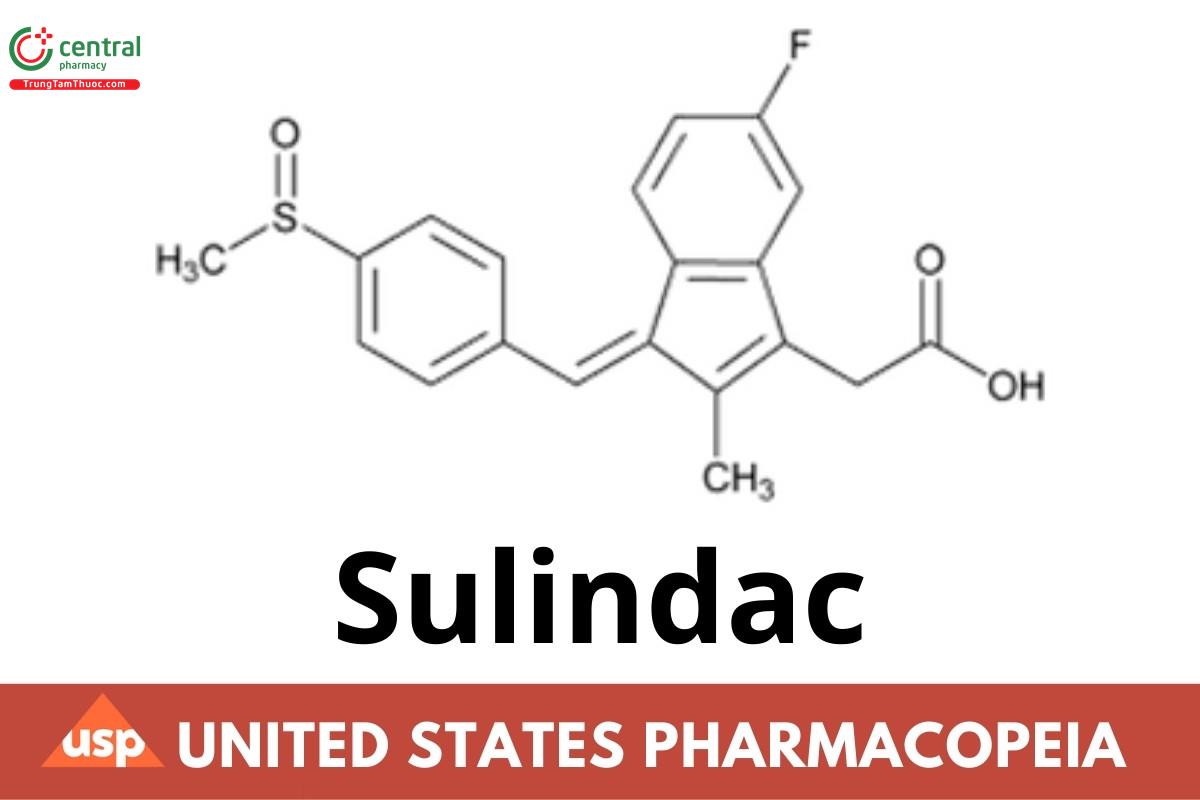
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 DEFINITION
Sulindac contains NLT 98.0% and NMT 102.0% of sulindac (C20H17FO3S), calculated on the dried basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy : 197M (CN 1-M -2020)
Change to read:
B. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Ultraviolet-Visible Spectroscopy : 197U (CN 1-MAY-2020)
Analytical wavelength: 284 nm
Medium: Hydrochloric acid in methanol (1 in 120) Sample solution: 15 µg/mL in Medium
Acceptance criteria: Absorptivities, calculated on the dried basis, do not differ by more than 3.0%.
C. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
PROCEDURE
Solution A: 0.1% Formic acid in water Solution B: 0.1% Formic acid in acetonitrile
Mobile phase: See Table 1.
Table 1
Diluent: Acetonitrile and water (50:50)
System suitability solution: 0.003 mg/mL each of USP Sulindac RS, USP Sulindac Related Compound A RS, USP Sulindac Related Compound B RS, and USP Sulindac Related Compound C RS in Diluent
Standard solution: 0.2 mg/mL of USP Sulindac RS in Diluent
Sample solution: 0.2 mg/mL of Sulindac in Diluent
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: 330 nm
Column: 2.1-mm × 15-cm; 1.7-µm packing L1
Column temperature: 45°
Flow rate: 0.3 mL/min
Injection volume: 2 µL
System suitability
Samples: System suitability solution and Standard solution
[NOTE—The relative retention times for sulindac related compound A, sulindac related compound B, and sulindac related compound C are 1.25, 1.34, and 1.67, respectively.]
Suitability requirements
Resolution: NLT 4.0 between sulindac and sulindac related compound A and between sulindac related compound A and sulindac related compound B, System suitability solution
Tailing factor: NMT 2.0, Standard solution
Relative standard deviation: NMT 0.73%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of sulindac (C20H17FO3S) in the portion of Sulindac taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response from the Sample solution
rS = peak response from the Standard solution
CS = concentration of USP Sulindac RS in the Standard solution (mg/mL)
CU = concentration of Sulindac in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–102.0% on the dried basis
4 IMPURITIES
RESIDUE ON IGNITION 〈281〉: NMT 0.1%
RESIDUAL SOLVENTS 〈467〉
Solvent: Dimethylsulfoxide
Acceptance criteria: Meets the requirements except that chloroform is NMT 500 ppm
ORGANIC IMPURITIES
Solution A, Solution B, Mobile phase, Diluent, and Chromatographic system: Proceed as directed in the Assay.
Standard solution: 0.6 µg/mL of USP Sulindac RS and 3 µg/mL each of USP Sulindac Related Compound A RS, USP Sulindac Related Compound B RS, and USP Sulindac Related Compound C RS in Diluent. Sonicate for 2–5 min.
Sample solution: 600 µg/mL of Sulindac in Diluent. Sonicate for 2–5 min. System suitability
Sample: Standard solution Suitability requirements
Resolution: NLT 4.0 between sulindac and sulindac related compound A and between sulindac related compound A and sulindac related compound B
Relative standard deviation: NMT 2.0% for any peak Analysis
Samples: Standard solution and Sample solution
Calculate the percentages of sulindac related compound A, sulindac related compound B, and sulindac related compound C in the portion of Sulindac taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of the specified impurity from the Sample solution
rS = peak response of the corresponding related compound from the Standard solution
CS = concentration of the corresponding related compound in the Standard solution (µg/mL)
CU = concentration of Sulindac in the Sample solution (µg/mL)
Calculate the percentage of any individual unspecified impurity in the portion of Sulindac taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of any individual unspecified impurity from the Sample solution
rS = peak response of sulindac from the Standard solution
CS = concentration of USP Sulindac RS in the Standard solution (µg/mL)
CU = concentration of Sulindac in the Sample solution (µg/mL)
Acceptance criteria: See Table 2.
Table 2
a (E)-2-{5-Fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid.
b (Z)-2-{5-Fluoro-2-methyl-1-[4-(methylsulfonyl)benzylidene]-1H-inden-3-yl}acetic acid.
c (Z)-2-{5-Fluoro-2-methyl-1-[4-(methylthio)benzylidene]-1H-inden-3-yl}acetic acid.
5 SPECIFIC TESTS
LOSS ON DRYING 〈731〉
Analysis: Dry under vacuum at 100° for 2 h. Acceptance criteria: NMT 0.5%
6 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in well-closed containers.
USP REFERENCE STANDARDS 〈11〉
USP Sulindac RS
USP Sulindac Related Compound A RS
(E)-2-{5-Fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1H-inden-3-yl}acetic acid. C20H17FO3S 356.41
USP Sulindac Related Compound B RS
(Z)-2-{5-Fluoro-2-methyl-1-[4-(methylsulfonyl)benzylidene]-1H-inden-3-yl}acetic acid. C20H17FO4S 372.41
USP Sulindac Related Compound C RS
(Z)-2-{5-Fluoro-2-methyl-1-[4-(methylthio)benzylidene]-1H-inden-3-yl}acetic acid. C20H17FO2S 340.41


