Sulfaquinoxaline
If you find any inaccurate information, please let us know by providing your feedback here
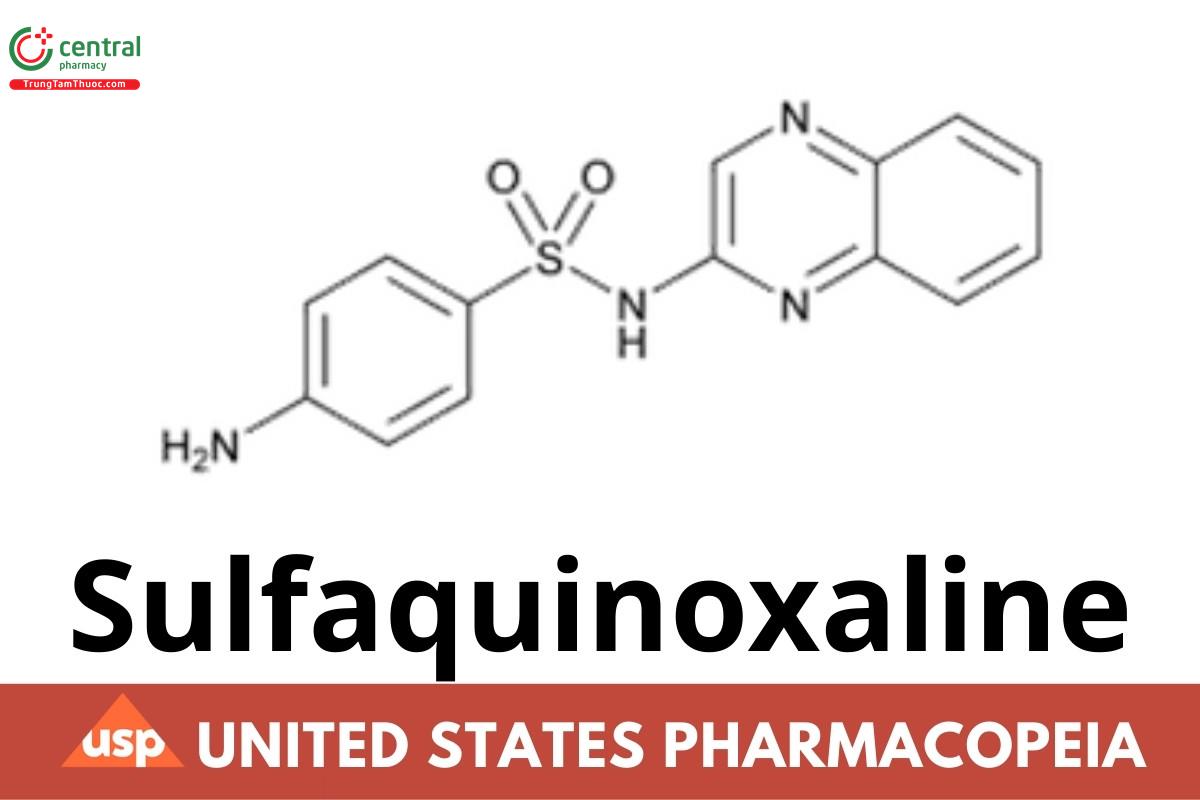
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 DEFINITION
Sulfaquinoxaline contains NLT 98.0% and NMT 101.0% of C14H12N4O2S, calculated on the dried basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy : 197K (CN 1-M -2020)
Change to read:
B. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Ultraviolet-Visible Spectroscopy : 197U (CN 1-MAY-2020)
Sample solution: 10 µg/mL in 0.01 N sodium hydroxide Acceptance criteria: Meets the requirements
3 ASSAY
PROCEDURE
Mobile phase: 2 g/L of monobasic ammonium phosphate in a mixture of acetonitrile, glacial acetic acid, tetrahydrofuran, ammonium hydroxide, and water (400:10:5:2:583). Pass through a filter of 0.5-µm or finer pore size.
Standard solution: 0.06 mg/mL of USP Sulfaquinoxaline RS in 0.01 N sodium hydroxide
Sample solution: 0.06 mg/mL of Sulfaquinoxaline in 0.01 N sodium hydroxide
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 254 nm
Column: 4-mm × 25-cm; packing L1 Flow rate: 1 mL/min
Injection size: 15 µL System suitability
Sample: Standard solution Suitability requirements
Tailing factor: NMT 1.2
Relative standard deviation: NMT 1.0% Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of sulfaquinoxaline (C14H12N4O2S) in the portion of Sulfaquinoxaline taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response from the Sample solution
rS = peak response from the Standard solution
CS = concentration of USP Sulfaquinoxaline RS in the Standard solution (mg/mL)
CU = concentration of Sulfaquinoxaline in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–101.0% on the dried basis IMPURITIES
RESIDUE ON IGNITION 〈281〉: NMT 0.1%
ORGANIC IMPURITIES
Sample solution: 4 mg/mL, prepared as follows. Dissolve 400 mg of Sulfaquinoxaline in 4 mL of 1 N sodium hydroxide, dilute with methanol to 100 mL, and mix.
Standard solution A: 0.12 mg/mL of USP Sulfaquinoxaline Related Compound A RS in methanol Standard solution B: 0.04 mg/mL of sulfanilamide in methanol
Chromatographic system
(See Chromatography 〈621〉, Thin-Layer Chromatography.)
Mode: TLC
Adsorbent: 0.25-mm layer of chromatographic silica gel mixture
Application volume: 5 µL
Developing solvent system: Chloroform, methanol, and ammonium hydroxide (60:40:20)
Analysis: Separately apply each solution to the TLC plate, and proceed as directed in the chapter. When the solvent front has moved about three-fourths the length of the plate, remove the plate from the chamber, mark the solvent front, allow it to air-dry, and examine the plate under short-wavelength UV light.
Acceptance criteria: No spot corresponding to sulfaquinoxaline related compound A in the chromatogram of the Sample solution is more intense than the principal spot in the chromatogram of Standard solution A (NMT 3.0%); and no spot, other than the principal spot and the sulfaquinoxaline related compound A spot, in the chromatogram of the Sample solution is more intense than the principal spot in the chromatogram of Standard solution B (NMT 1.0%).
4 SPECIFIC TESTS
ACIDITY
Sample: 2 g
Analysis: Digest the Sample with 100 mL of water at about 70° for 5 min, cool to about 20°, and filter. Titrate 50 mL of the filtrate with 0.1 N sodium hydroxide VS to a pH of 7.0.
Acceptance criteria: NMT 0.2 mL is required
LOSS ON DRYING 〈731〉: Dry a sample at 105° for 4 h: it loses NMT 1.0% of its weight.
5 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in well-closed containers, protected from light.
LABELING: Label it to indicate that it is for veterinary use only.
USP REFERENCE STANDARDS 〈11〉
USP Sulfaquinoxaline Related Compound A RS
N1,N2-Diquinoxalin-2-ylsulfanilamide.
C22H16N6SO2 428.50
USP Sulfaquinoxaline RS


