Sulfamethoxazole
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
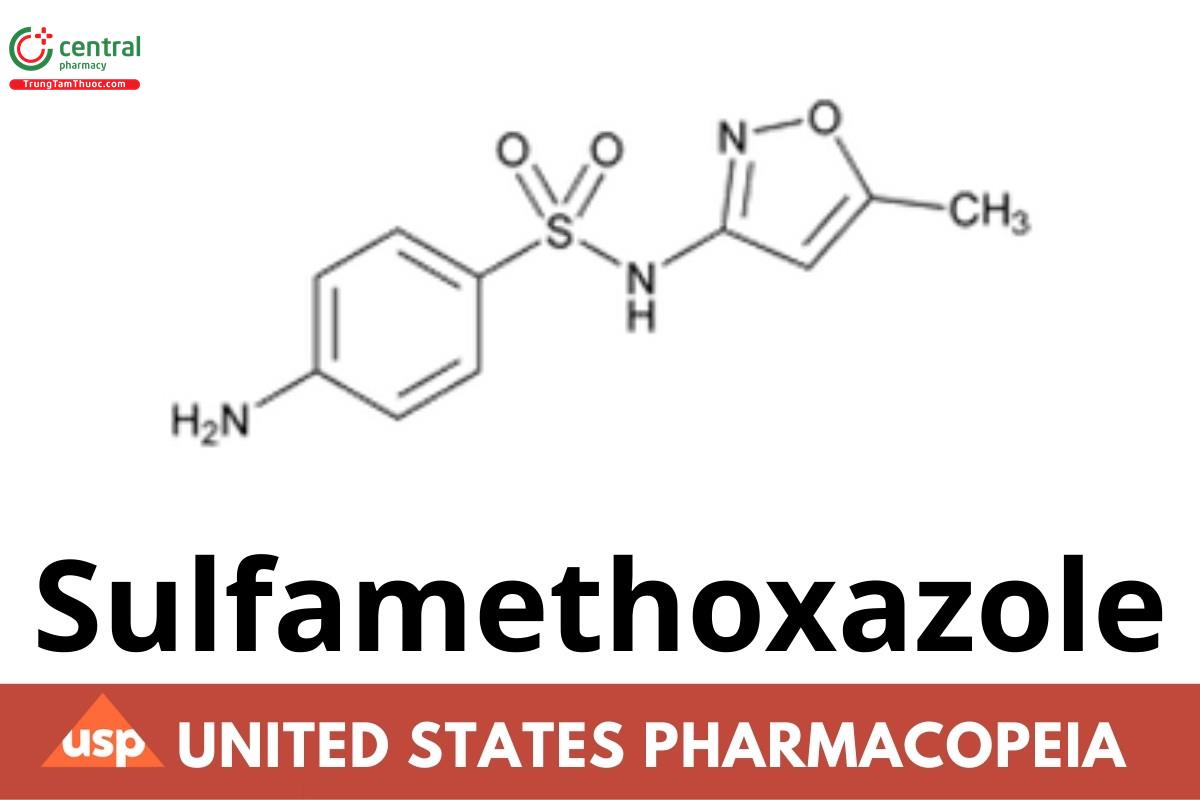
1 DEFINITION
Sulfamethoxazole contains NLT 98.0% and NMT 102.0% of sulfamethoxazole (C10H11N3O3S), calculated on the dried basis.
2 IDENTIFICATION
Change to read:
A. SPECTROSCOPIC IDENTIFICATION TESTS 〈197〉 , Infrared Spectroscopy: 197K or 197A (IRA 1-May-2021)
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
3 ASSAY
Change to read:
4 PROCEDURE
Buffer: 13.6 g/L of potassium dihydrogen phosphate adjusted with a 20-g/L solution of potassium hydroxide to a pH of 5.3 Mobile phase: Methanol and Buffer (30:70)
Standard solution: 0.1 mg/mL (IRA 1-May-2021) of USP Sulfamethoxazole RS (IRA 1-May-2021) in Mobile phase. Sonicate at 45° with intermittent shaking to dissolve before final dilution.
System suitability solution: 0.1 mg/mL each of USP Sulfamethoxazole RS and USP Sulfamethoxazole Related Compound A RS in Mobile phase. Sonicate at 45° with intermittent shaking to dissolve before final dilution. (IRA 1-May-2021)
Sample solution: 0.1 mg/mL of Sulfamethoxazole in Mobile phase. Sonicate at 45° with intermittent shaking to dissolve before final dilution.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 210 nm
Column: 4-mm × 25-cm; 5-µm packing L7
Flow rate: 0.9 mL/min
Injection volume: 20 µL System suitability
Samples: System suitability solution and (IRA 1-May-2021) Standard solution
[NOTE—The relative retention times for sulfamethoxazole and sulfamethoxazole related compound A are 1.0 and 1.2, respectively.] Suitability requirements
Resolution: NLT 3.5 between sulfamethoxazole and sulfamethoxazole related compound A, System suitability solution (IRA 1-May-2021)
Relative standard deviation: NMT 0.73%, Standard solution (IRA 1-May-2021)
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of sulfamethoxazole (C10H11N3O3S) in the portion of Sulfamethoxazole taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of sulfamethoxazole from the Sample solution
rS = peak response of sulfamethoxazole from the Standard solution
CS = concentration of USP Sulfamethoxazole RS in the Standard solution (mg/mL)
CU = concentration of Sulfamethoxazole in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–102.0% on the dried basis
5 IMPURITIES
RESIDUE ON IGNITION 〈281〉: NMT 0.1%
Change to read:
ORGANIC IMPURITIES
Buffer, Mobile phase, System suitability solution, (IRA 1-May-2021) and Chromatographic system: Proceed as directed in the Assay.
(IRA 1-May-2021)
Peak identification solution: 1 µg/mL each of USP Sulfamethoxazole Related Compound A RS, USP Sulfamethoxazole Related Compound B RS, USP Sulfamethoxazole Related Compound C RS, USP Sulfanilic Acid RS, and USP Sulfanilamide RS in Mobile phase. Sonicate, if necessary, to dissolve before final dilution.
Sensitivity solution: 0.3 µg/mL of USP Sulfamethoxazole RS in Mobile phase. Sonicate, if necessary, to dissolve before final dilution. (IRA 1- May-2021)
Standard solution: 1 µg/mL each of USP Sulfamethoxazole RS and USP Sulfamethoxazole Related Compound F RS in Mobile phase. Sonicate,
if necessary, to dissolve before final dilution.
Sample solution: 1 mg/mL of Sulfamethoxazole in Mobile phase. Sonicate at 45° with intermittent shaking to dissolve before final dilution. System suitability
Samples: System suitability solution, Sensitivity solution, (IRA 1-May-2021) and Standard solution Suitability requirements
Resolution: NLT 3.5 between sulfamethoxazole and sulfamethoxazole related compound A, (IRA 1-May-2021) System suitability solution Relative standard deviation: NMT 5.0% each (IRA 1-May-2021) for sulfamethoxazole and sulfamethoxazole related compound F, (IRA 1-May-2021) Standard solution
Signal-to-noise ratio: NLT 10, Sensitivity solution (IRA 1-May-2021)
Analysis
Samples: Peak identification solution, Standard solution, and Sample solution
Calculate the percentage of sulfamethoxazole related compound F in the portion of Sulfamethoxazole taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of sulfamethoxazole related compound F from the Sample solution
rS = peak response of sulfamethoxazole related compound F from the Standard solution
CS = concentration of USP Sulfamethoxazole Related Compound F RS in the Standard solution (mg/mL)
CU = concentration of Sulfamethoxazole in the Sample solution (mg/mL)
Calculate the percentage of any other (IRA 1-May-2021) individual impurity in the portion of Sulfamethoxazole taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak response of any other (IRA 1-May-2021) individual impurity from the Sample solution
rS = peak response of sulfamethoxazole from the Standard solution
CS = concentration of USP Sulfamethoxazole RS in the Standard solution (mg/mL)
CU = concentration of Sulfamethoxazole in the Sample solution (mg/mL)
Acceptance criteria: See Table 1. The reporting threshold is 0.03%.
Table 1
| Name | Relative Retention Time | Acceptance Criteria, NMT (%) |
| Sulfanilic acid | 0.26 | 0.10 |
| Sulfanilamide | 0.35 | 0.10 |
| Sulfamethoxazole related compound F | 0.45 | 0.10 |
| Sulfamethoxazole related compound C | 0.50 | 0.10 |
| Sulfamethoxazole | 1.0 | — |
| Sulfamethoxazole related compound A | 1.2 | 0.10 |
| Sulfamethoxazole related compound B | 2.1 | 0.10 |
| Any individual unspecified impurity | — | 0.10 |
| Total impurities | — | 0.30 |
6 SPECIFIC TESTS
LOSS ON DRYING 〈731〉
Analysis: Dry at 105° for 4 h.
Acceptance criteria: NMT 0.5%
7 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in well-closed, light-resistant containers. Store at room temperature.
Change to read:
USP REFERENCE STANDARDS 〈11〉
USP Sulfamethoxazole RS
USP Sulfamethoxazole Related Compound A RS
N-{4-[N-(5-Methylisoxazol-3-yl)sulfamoyl]phenyl}acetamide. C12H13N3O4S 295.31
USP Sulfamethoxazole Related Compound B RS
4-Amino-N-{4-[N-(5-methylisoxazol-3-yl)sulfamoyl]phenyl}benzenesulfonamide. C16H16N4O5S2 408.45
USP Sulfamethoxazole Related Compound C RS
5-Methylisoxazol-3-amine. C4H6N2O 98.11 (IRA 1-May-2021)
USP Sulfamethoxazole Related Compound F RS
4- Amino-N-(3-methylisoxazol-5-yl)benzenesulfonamide. C10H11N3O3S 253.28
USP Sulfanilamide RS
4- Aminobenzenesulfonamide. C6H8N2O2S 172.20
USP Sulfanilic Acid RS
4-Aminobenzenesulfonic acid. C6H7NO3S 173.19


