Storage and Transportation of Investigational Drug Products
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
Clinical trials are drug studies that are performed to determine if an investigational medicine meets the effectiveness and safety criteria as has been outlined in the protocol for the clinical trial. Investigational drug products (IDPs) are products not commercially available for the indication or dosage being tested; there may be situations when a commercially available product may be used in a clinical trial as a positive control (comparator), a new indication, or a rescue medication. The term IDP is used throughout this document. Where IDP is used, others may be more familiar with the European Union term investigational medicinal product (IMP). The definition of an IMP is provided in European Commission Directive 2001/20/EC, Article 2 (d), as "a pharmaceutical form of an active substance or Placebo being tested or used as a reference in a clinical trial, including products already with a marketing authorization but used or assembled (formulated or packaged) in a way different from the authorized form, or when used for an unauthorized indication, or when used to gain further information about the authorized form". For the purposes of this chapter, we will consider the two terms as equivalent. The pre-commercial nature of IDPs means the manufacturing ingredients, including active pharmaceutical ingredients (APIs), excipients, the clinical trial dosage, and any associated stability and packaging components may not be as defined as the final approved finished product. The Food and Drug Administration (FDA) current Good Manufacturing Practice regulations for finished drug products apply equally to commercial products and IDPs.
IDP distribution differs from commercial distribution in that the quantities for IDPs are often small (e.g., as little as one or two bottles or unit dose packages) and there are numerous final destinations, such as clinics and remote clinical settings. Another difference from a commercial drug product is the known stability of the IDP that is often a new chemical/molecular entity in the early stages of clinical trials and has not been through the robust stability program of commercial products. The IDP also creates a unique challenge when compared to a commercial product, which is ensuring the proper distribution of every packaging system, because a temperature excursion may jeopardize the entire clinical trial outcome.
Change to read:
2 SCOPE
Due to global security issues, regulations may vary and/or change for the distribution of drug products and for IDPs as well. Sponsors, clinical trial site personnel, or their designees are responsible for control of IDPs and should maintain an understanding of the current country distribution requirements. This chapter will address the aspects of storage and distribution that are unique to IDPs (e.g., unblinding, comparators, and academic studies); other chapters of interest may be Monitoring Devices-Time, Temperature, and Humidity (1079.3) (CN-1-Dec-2023) Risks and Mitigation Strategies for the Storage and Transportation of Finished Drug Products (1079), and Good Distribution Practices for Bulk Pharmaceutical Excipients (1197). This guidance chapter applies to all IDPs, including drug device combinations as well as non-commercial clinical IDPs, but does not apply to medical devices. The non-commercial clinical trial may have limited financial commercial return and is often performed in an academic setting or by a compounding pharmacy with limited industry participation (see Figure 1).
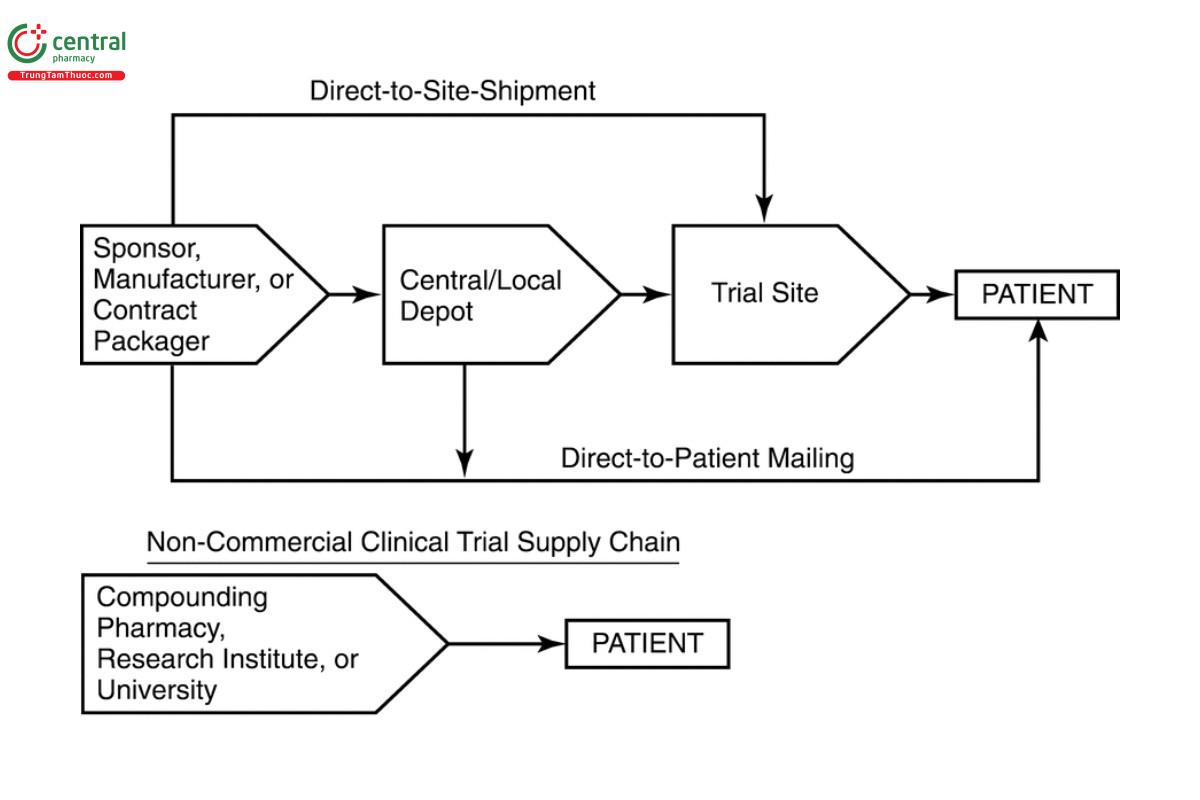
3 KEY FACTORS FOR IDP DISTRIBUTION
3.1 Number of Clinical Trial Sites
The larger the number of clinical trial sites, the greater the risk involved, as this may mean more countries, shipments, and customs challenges. The IDP sponsor company and distributor should be aware of customs requirements in all countries where the IDP is to be distributed. IDPs may be transported from one clinical trial site to another, and the distribution of these IDPs must undergo the same distribution requirements as any other drug products being transported.
3.2 Audits
3.2.1 CLINICAL TRIAL SITES
Clinical trial sites must store IDPs in an area that is safe and secure with limited access, and ensure products are maintained at their labeled storage conditions. A site qualification audit will ensure that the sites have adequate space and segregation for both IDPs that have not been dispensed to patients, as well as IDPs that are awaiting return to a sponsor or contractor for final reconciliation and disposition. Depending on the regulatory requirements of the countries where the studies are being conducted, calibrated thermometers or an automated temperature tracking and alarm system should be in place to document site compliance.
3.2.2 DEPOTS AND SUBCONTRACTORS
These places and individuals should follow both the sponsor requirements and associated country regulations in which the study is being conducted.
3.3 Available Product Quantities
In early stage development (i.e., Phase 1 and early Phase 2), there are typically fewer APIs and IDPs available. Disruptions in the supply chain will have greater impact and consequences to the timely completion of clinical trials, and distribution delays (especially for biologics) may impact enrollment and study completion.
3.4 Expiration/Retest Dates
As the stability profile is being developed during the early phases of development, expiry or re-test date extensions should be anticipated. IDP expiry or re-test dates should follow local regulatory requirements throughout the life of the study. Expiry or re-test dates can be tracked through a validated electronic system, though IDP expiry or re-test dates at global sites or depots may need to be manually updated.
3.5 Qualification of Packaging for IDP and Track and Trace
There is no difference in the packaging qualifications for IDPs and commercially available drug products.
Determining what the required environmental conditions of IDP shipments will be during the early stages of a clinical research protocol is of particular importance, as this is the best time to define the robust stability profile of the study drug. Important distribution risk factors to consider include the distance, time, temperature, and the number and type of handoffs that may occur. Packaging that is appropriate to the clinical phase should be used to protect the contents. When that is not possible, the intent is to use qualified components (i.e., components that have been tested to show positive outcomes for maintaining temperatures for a wide variety of products in a wide variety of environmental conditions) to ensure the IDPs inside the shipping containers are controlled and remain at the correct temperature during the journey from warehouse (or depot) to clinical trial site. There are, however, sophisticated track and trace technologies that permit the trial sponsor, packaging contractor, clinical research organization, and the clinical trial site to monitor (track) the IDP along the supply chain.
Performance qualification (PQ) of packaging systems for shipping IDPs should be carried out reflecting the actual load configuration, duration of travel, and expected environmental conditions. There should be no difference in the PQ rational for investigational and commercial drug products because the risks of storage and transport are not related to the marketing authorization. Details of PQ as a strategy for mitigating risks are provided in (1079).
3.6 IDP Supply Chain Challenges
One of the larger IDP supply chain challenges is with the number of clinical trial supply shipments that are made in support of clinical trials. In IDP distribution circles, these shipments are referred to as "outbound" shipments. The faster the trial enrolls, the larger the number of shipments. With each shipment we now increase the risk of damage as well as temperature excursions. Each of these would mean shipping additional IDPs to support the trial and added cost to the sponsor company.
Among the challenges for the IDP supply chain are the frequency and size of shipments. These are influenced by such factors as number of patients, frequency of IDP administration to patients, the number and size of patient kits in each shipment, and the expense associated with each shipment.
3.7 Timing
Clinical trial studies that are longer in duration than the original plan may mean more IDP shipments and the need to ensure these extensions continue being in compliance with current good distribution practices. There are instances when IDP inventory remains in the investigational site when enrollment is complete, even though enrolled patients continue to take their remaining treatment. In other instances, IDP may remain in the institution while awaiting return/destruction instructions from the sponsor. The guidelines in this chapter apply to both instances.
3.8 Key Global Challenges for IDPs
Global challenges include security, language, culture, tariff codes/taxes, hiring, training, customs, holidays, country-specific import and export requirements, time differences, airline schedules, and documentation. Guidance for such issues can be found in regulatory guidance documents [e.g., FDA Drug Supply Chain Security Act (DSCSA) or International Council for Harmonisation (ICH) Q Series].
4 IDP ENVIRONMENTAL CONDITIONS
The environmental controls for the distribution of IDPs may be provided for on container labeling, product certificate of analysis, safety data sheet, or other records available for shipping purposes; however, these environmental conditions are not always known at the time of transportation to the trial sites, such as the clinical setting or the patient's home (for home administration). There must be confirmation that the proper steps are taken to ensure IDP supply chain integrity throughout the distribution process. At the point that the IDP is dispensed to the research participant (subject or patient), and is in their custody, the proper handling and storage of the IDP should be explained (in written form or verbally) at the clinical site, in the hospital, or in the participant's home. IDPs that have been dispensed to a patient or subject may not be returned to research stock at the investigator site to be dispensed again. In the case where the IDP is provided to a member of the research team to be dispensed to the participant, but it is not dispensed to the subject or patient, and the chain of custody can be verified to have remained within the study team, the IDP may be received back to the investigational drug service to be dispensed again only if there are standard operating procedures (SOPs) and documentation in place to ensure control of the IDP. Please note that this redispensing guidance may be overruled if the specific protocol or sponsor SOPs or instructions forbids redispensing in any case.
4.1 Unblinding
Blinding of IDPs is performed to reduce or eliminate bias in clinical trials and assists in providing randomization across patient populations. In certain circumstances, in order to maintain the safety of research participants, emergency unblinding is necessary. This may be done for an individual dose or a batch/lot. In either case, the unblinded IDP would be separated from the supply of the IDP waiting to be dispensed. Depending upon the reason for the unblinding, the sponsor shall provide instruction regarding return of the IDP or destruction. The impact of unblinding a product can create a need for distribution of a new IDP, either from the manufacturer, depot, or from another clinical trial site. The supply chain partners should have a written procedure establishing responsibilities, procedures, precautions, records, and final destination regarding the unblinding of IDPs to ensure proper handling of this material and avoid the redistribution of IDPs with a broken randomized code.
4.2 Comparators
Most IDPs in late Phase 2 and all of Phase 3 clinical trials will be compared to the current standard of care (i.e., commercially available product). This process is generally done using a comparator product that may or may not be currently in the market place in countries where the clinical trial is being conducted. The distribution process for the comparator should follow the shipping requirements on the IDP labeling for that product or from the manufacturer of the product. The IDP provider using comparators should understand and follow the distribution requirements of the comparator to ensure a safe and efficacious product for the clinical trial patient.
4.3 Returned IDPs
There should be the same concern for environmental monitoring and supply chain integrity for returned IDPs, as some sponsors may choose to redistribute any returned IDPs that are unopened and undispensed to other clinical trial sites. Returned IDPs intended for redistribution must meet all requirements (i.e., initial release criteria) of the product and associated local regulations. The decision to reuse returned IDPs should be supported by a risk-based assessment.
5 ASSESSING RISK EARLY IN THE DISTRIBUTION PROCESS
When shipping IDPs to depots and clinical trial sites, using common shipping lanes (routes between the point of origin and the destination point) and within appropriate temperature zones, a risk assessment can identify the probability and the impact missteps in the supply chain. ICH Q9 presents some risk assessment tools that can be used to help organizations to identify their risks and how to address them. Factors such as product category (e.g., narcotics, biologics, radiopharmaceuticals), storage conditions specification (e.g., temperature, light, humidity, vibration), environmental conditions that products will face according to the transportation route (e.g., origin, destiny, and lanes), duration of journey, shipping container (e.g., if temperature-controlled, if active or passive), and type of vehicle should be taken into consideration for risk assessment. Organizations should also evaluate if delays along the transportation route or due to custom holds can increase risk rates and should propose strategies to mitigate these risks.
6 GLOSSARY
Blinding:
The process through which one or more parties to a clinical trial are unaware of the treatment assignments. Blinded studies are conducted to prevent the unintentional biases that can affect subject data when treatment assignments are known.
Comparator:
An investigational or marketed product (i.e., active control), or placebo, used as a reference in a clinical trial.
Distribution:
Refers to elements such as shipping and transportation activities that are associated with the movement and supply of drug products.
Investigational drug product (IDP):
Any pharmaceutical product (new product or reference product) or placebo being tested or used as a reference in a clinical trial.
Protocol:
The study plan on which a clinical trial is based.
Rescue medication:
Medicines identified in the protocol as those that may be administered to the patients when the efficacy of the investigational drug product is not satisfactory, or the effect of the IDP is too great and is likely to cause a hazard to the patient, or to manage an emergency situation.
Standard operating procedure (SOP):
Detailed written instructions to achieve uniformity of performance of a specific function. ICH/ISO 9001 required for repeated application of processes that require standardization. All SOPs require quality assurance approval.
Trial site:
The location where trial-related activities are actually conducted.


