Spacers and Valved Holding Chambers Used with Inhalation Aerosols—Characterization Tests
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
Change to read:
1 INTRODUCTION
1.1 Background
Spacers and valved holding chambers (VHCs) are widely used in conjunction with inhalation aerosols [commonly known as pressurized metered-dose inhalers (MDIs)]. When used correctly, VHCs assist with the administration of inhalation aerosols to patients who have poor coordination of MDI actuation and inhalation, and VHCs reduce oro-pharyngeal deposition of the drug. Patients using these spacers/VHCs in general breathe tidally when inhaling their medication, regardless of any delay, and this is particularly true for young and elderly users. Also, spacers and VHCs often come with a facemask instead of the mouthpiece normally supplied for inhalation aerosols. These add-on devices (spacers/VHCs) interface with the actuator/mouthpiece of the MDI and provide additional volume for the aerosol plume to develop. In this chapter, these aspects are addressed by providing patient-use appropriate scenarios that have been evaluated extensively.
Although existing methods for evaluating inhalation aerosols rely on sampling at a constant flow rate, spacers and VHCs may be used by patients who have poor inhalation technique (i.e., they delay inhalation after actuating the MDI). This chapter describes tests that are intended to provide information on how the spacer/VHC modifies the aerosol emitted by the MDI.
1.2 Purpose
The purpose of this chapter is to define potential standardized methods for characterizing the in vitro performance of a given MDI drug product with a specific spacer and VHC. It is recognized that spacer/VHC configurations will deliver a modified particle-size distribution and dose compared with the dose indicated on the label by the manufacturer of the MDI product, chiefly by collecting almost all of the portion of the dose contained in particles too large to be available for inhalation by the patient and potentially reach the lungs. This chapter does not advise on the outcome of such changes, rather the interpretation of data obtained using the methods described should be developed by the user after discussion with the appropriate receiving organization.
1.3 Rationale
This chapter includes separate testing by a multi-stage cascade impactor to determine aerosol aerodynamic particle size distribution (APSD) and tests mimicking fully coordinated and fully uncoordinated use, with tidal breathing simulated to establish emitted mass (EM) with the MDI-spacer/VHC coupled to a breathing simulator. (USP 1-Dec-2019) It is now possible to combine both APSD and EM measurements by means of a mixing inlet. (USP 1-Dec-2019) However, the more conservative approach adopted herein reflects the absence of method validation data for multiple marketed drug products delivered by MDI. Spacers and VHCs modify the APSD substantially from what is emitted by the MDI when used alone. In particular, almost all of the mass fraction of drug associated with particles having ballistic trajectories, by virtue of propellant expansion upon actuation, is contained within the add-on device. The user should refer to the Canadian Group standard for further interpretation of changes in APSD brought about by the presence of a spacer or VHC.
1.4 Recommendations
The use of the induction port identified in Inhalation and Nasal Drug Products: Aerosols, Sprays, and Powders—Performance Quality Tests 〈601〉 is recommended for compliance with the pharmacopeial standard. However, it is acknowledged that alternative designs of the induction port, as yet to be included as pharmacopeial standards, offer unique testing opportunities. These are either anatomically correct or idealized to have aerosol transport properties similar to an anatomically correct induction port. Given the widespread use of spacers/VHCs for infants and small children, it is important to note that many of these anatomically appropriate induction ports are scaled in terms of such potential users (e.g., the “Alberta” idealized throats, (USP 1-Dec-2019) developed at the University of Edmonton, Canada). The user is free to adopt such an induction port but should specify the induction port design and age group with which the induction port will be used.
The evaluation of facemask performance requires a model face of the appropriate age range specified in the labeling for the add-on device (i.e., infant, small child, or adult). This arrangement is necessary because there is no other way to accurately simulate the magnitude of the dead space or test for the possibility of leak pathways between facemask and face, both of which are known to influence efficiency of the drug delivery. Some face models are available commercially. (USP 1-Dec-2019) Therefore, because the use of a face model is an appropriate way to test these add-on devices, the user will need to either acquire a model from a source where age-appropriate face models have been developed and validated, or develop and validate his or her own design. Whichever pathway is chosen, a description of the model, in particular the dimensions of the face where the facemask comes into contact, should be provided in the data report to the recipient.
1.5 Definitions of Key Terms Relating to This Chapter
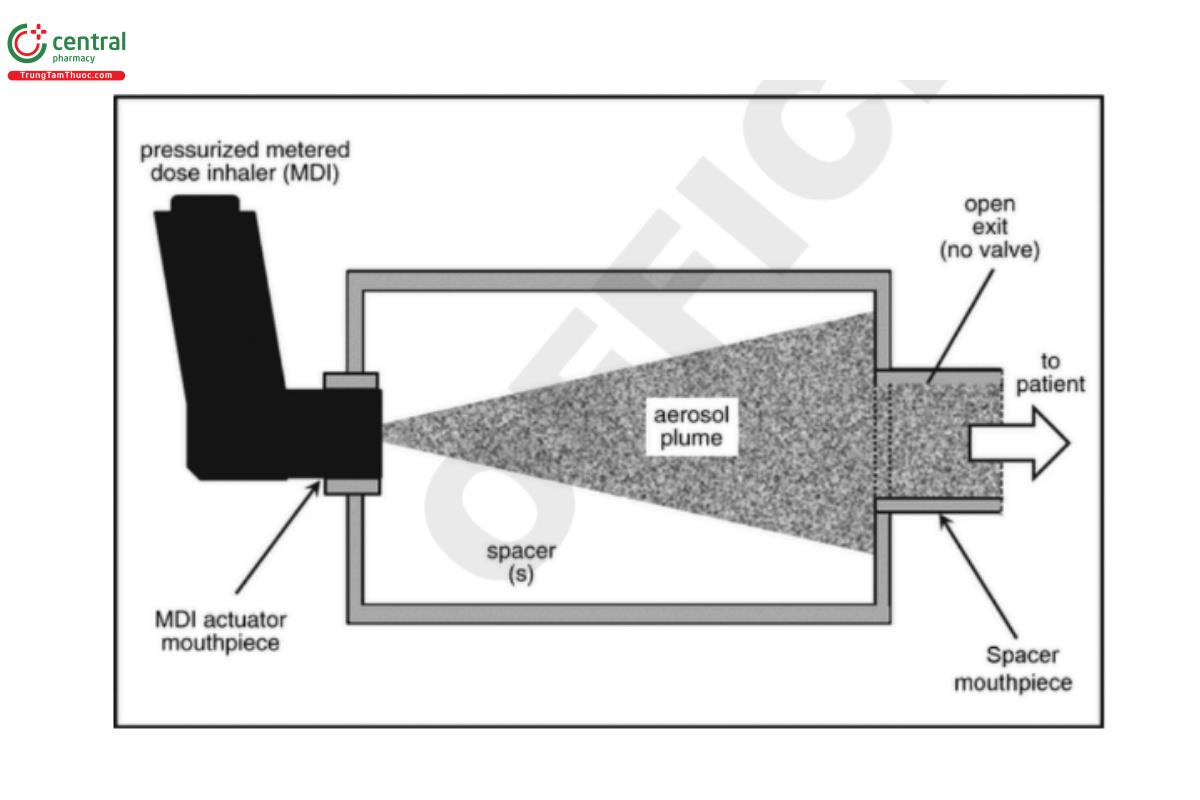
Spacers: Open tubes that do not have a valve at the exit nearest the patient interface (either the mouthpiece or facemask) to retain the aerosol before the user can inhale (see Figure 1). Spacers simply increase the distance between the MDI mouthpiece and the mouth of the patient. If the patient exhales instead of inhaling, the medication will be blown out of the spacer and lost.
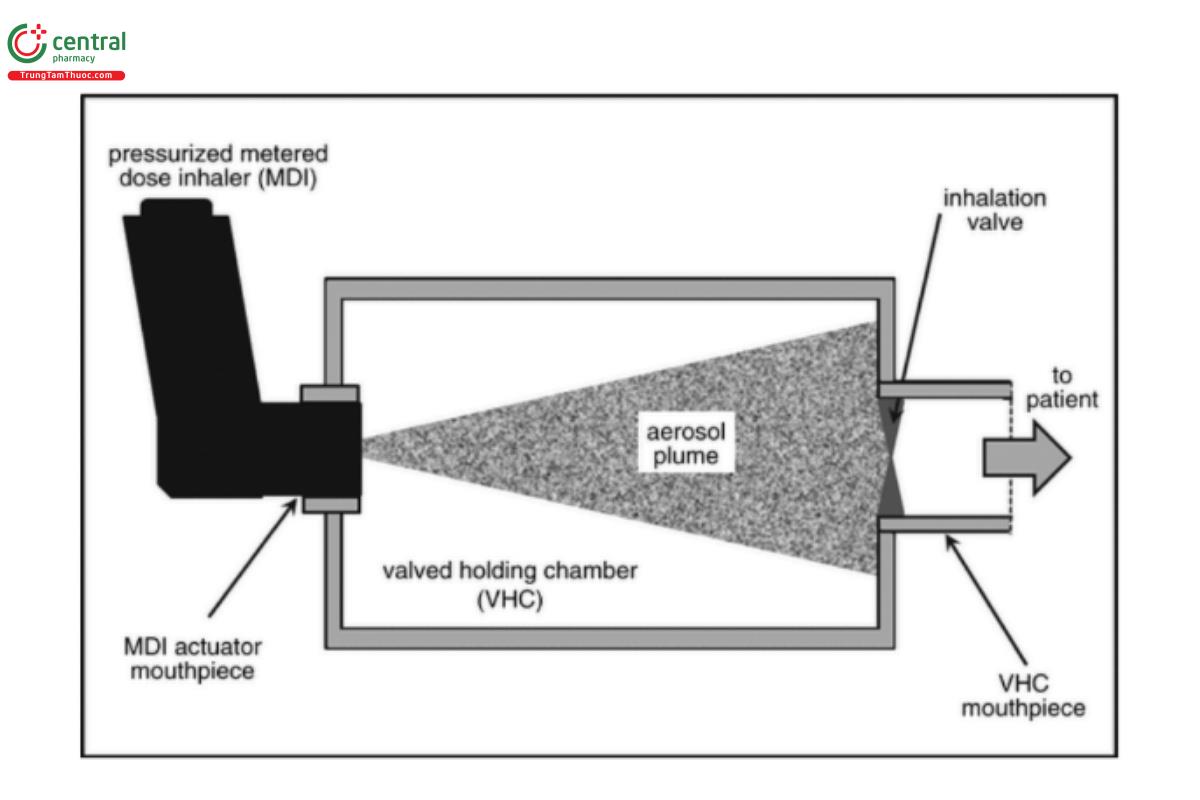
VHCs: Contain at least one valve that opens to allow the patient to inhale aerosol on inspiration. This inhalation valve remains closed at other times during each breathing cycle (see Figure 2). Some VHCs may also contain an exhalation valve that is open only during exhalation to direct the exhaled flow away from the inhalation valve.
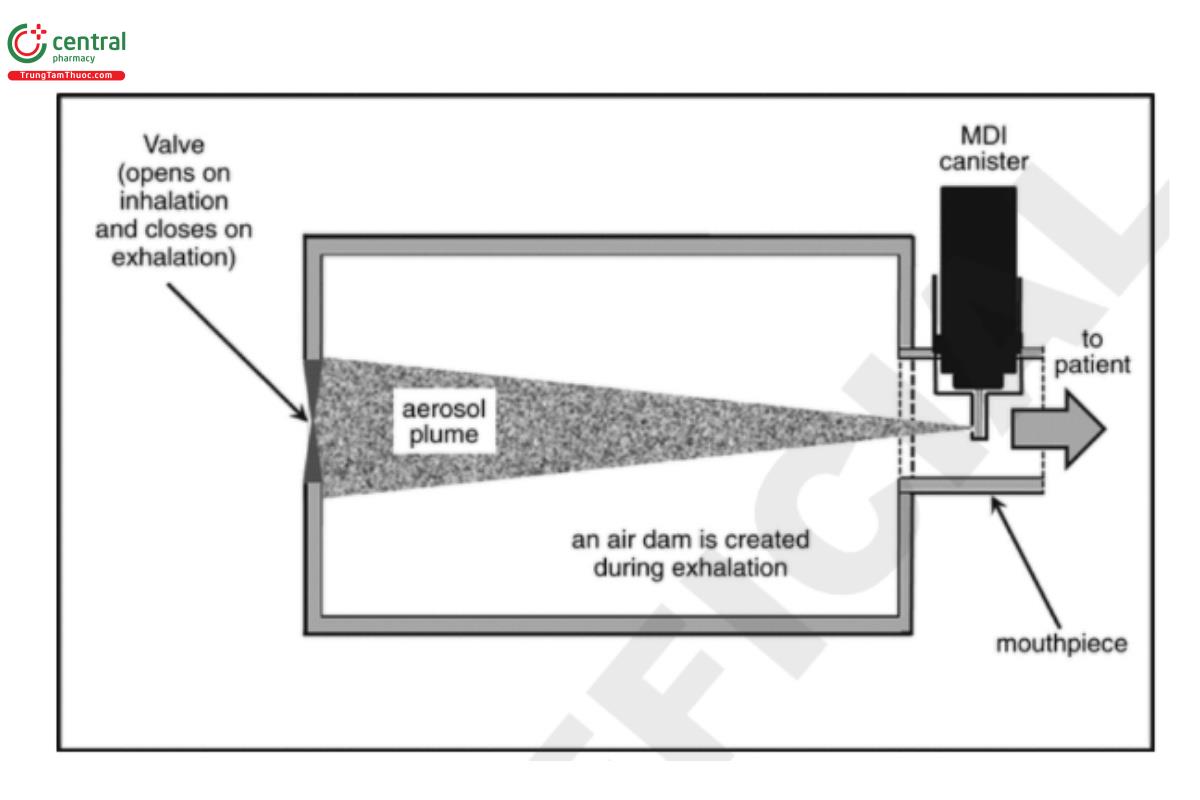
Integral actuator: A companion piece with some spacers and VHCs that accepts a MDI canister removed from its actuator-mouthpiece. A “reverse firing” feature may enable the design to be modified such that the built-in actuator faces away from rather than toward the user. In the example illustrated in Figure 3, the spacer operates in this way. It has some of the characteristics of a VHC, in that an air dam is created by closure of the valve located distally from the user on exhalation into the chamber. This type of spacer can therefore be evaluated as if it were a VHC, because the aerosol released on MDI actuation is conserved during exhalation.
1.6 Choice of Drug Product with which to Test
It is understood that testing will take place with each single active component within the drug product or with each strength of multi-strength drug products as required by the recipient of the data.
Change to read:
2 TEST METHOD SELECTION
2.1 Spacer/VHC Configurations
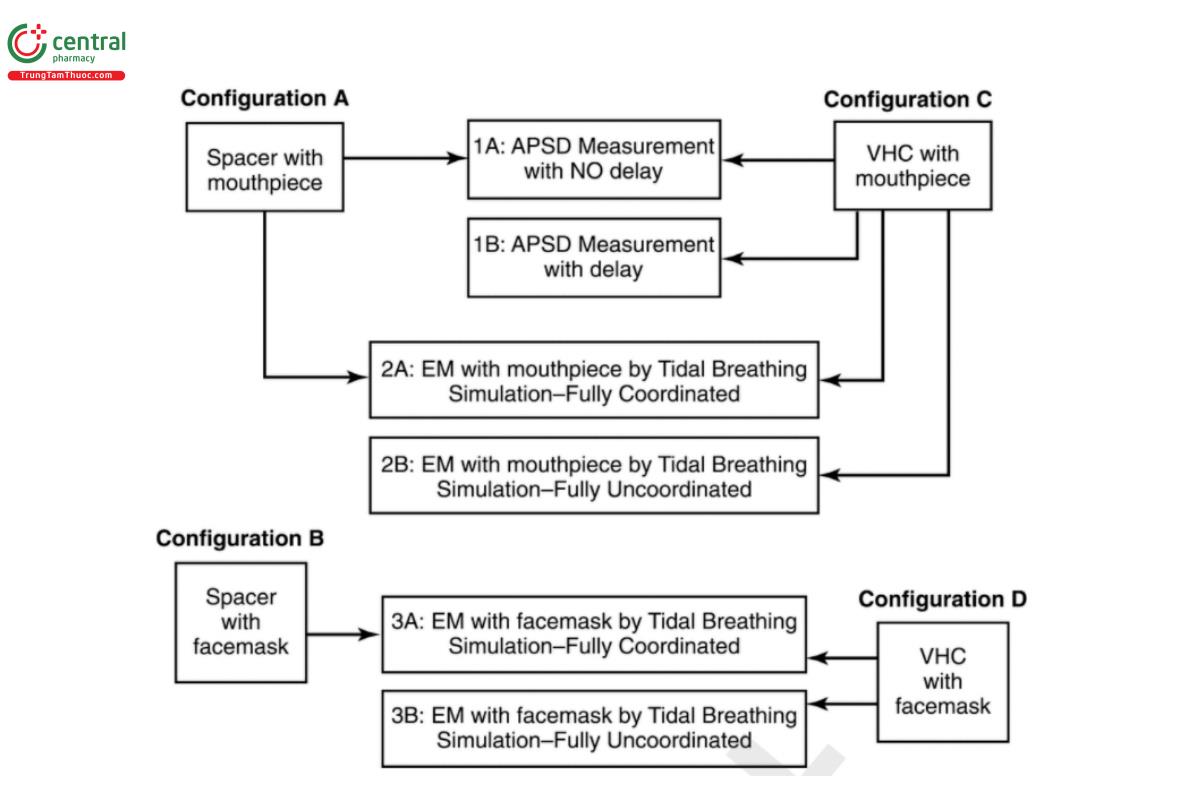
Four spacer/VHC configurations have been identified that require different test methods to complete the evaluation process (see Table 1).
Table 1. Identification of Test Configurations of the Spacer/VHC
| Configuration | Spacer | VHC | Mouthpiece | Facemask |
| A | +ᵃ | −ᵇ | + | − |
| B | + | − | − | + |
| C | − | + | + | − |
| D | − | + | − | + |
ᵃ + denotes that this item was part of the condition.
ᵇ − denotes that this item was not part of the condition.
In Configuration A, the device being tested is a spacer with a mouthpiece. Likewise, in Configuration B, the device under test is a spacer with a facemask. In Configuration C, the device is a VHC with a mouthpiece, and in Configuration D the device is a VHC with a facemask. The evaluation of devices without patient interface (i.e., those intended for use by patients on mechanical ventilation) is outside the scope of this chapter.
Tests for configuration A
Configuration A, spacer with mouthpiece (see Figure 4), comprises two tests: 1) APSD measurement with no delay (see Part 1A); and 2) EM by breathing simulation-fully coordinated (see Part 2A). Testing with a delay would be inappropriate for this configuration because the medication cannot be retained within the spacer because it has no valving mechanism.
Tests for configuration B
Configuration B, spacer with facemask (see Figure 4), comprises one test with the facemask in place: 1) EM by breathing simulation-fully coordinated (see Part 3A).[NOTE-The facemask should NOT be removed for this test, because it is important to be able to evaluate the effect of dead space between facemask and face, as well as the correct function of an exhalation valve, if provided.] (USP 1-Dec-2019) It is recommended that this class of device be evaluated for APSD measurement with no delay (see Part 1A) by removing the facemask and coupling the spacer on-axis to the induction port entry with a suitable connector. Testing with delay would be inappropriate for this configuration, because the medication cannot be retained within the spacer because it has no valving mechanism. [NOTE-Removal of the facemask is acceptable for APSD determination, because the entire bolus of medication leaving the spacer is sampled in the continuous vacuum that is required to operate the cascade impactor.] (USP 1-Dec-2019)
Tests for configuration C
Configuration C, VHC with mouthpiece (see Figure 4), comprises four tests: 1) APSD measurement with no delay (see Part 1A); 2) APSD measurement with delay (see Part 1B); 3) EM by breathing simulation-fully coordinated (see Part 2A); and 4) EM by breathing simulation-fully uncoordinated (see Part 2B).
Tests for configuration D
CConfiguration D, VHC with facemask (see Figure 4), comprises two tests with the facemask in place: 1) EM by breathing simulation-fully coordinated (see Part 3A); and 2) EM by breathing simulation-fully uncoordinated (see Part 3B). [NOTE-The facemask should NOT be removed for this test, because it is important to be able to evaluate the effect of dead space between facemask and face, as well as the correct function of an exhalation valve, if provided.] (USP 1-Dec-2019) It is recommended that this class of device be evaluated for APSD measurement with and without delay (see Parts 1A and 1B) by removing the facemask and coupling the VHC on-axis to the induction port entry with a suitable connector. [NOTE-Removal of the facemask is acceptable for APSD determination, because the entire bolus of medication leaving the spacer is sampled in the continuous vacuum that is required to operate the cascade impactor.] (USP 1-Dec-2019)
2.2 Comments on Test Methods
The tests in Parts 1A and 1B (see Table 2) are an extension of the procedures described in 〈601〉. However, sampling at a constant flow rate via a cascade impactor does not do more than enable the assessment of the APSD of the emitted aerosol. In the later parts of Products for Nebulization—Characterization Tests 〈1601〉, tests are described in which tidal breathing is simulated to assess spacer/VHC performance in terms of the delivered dose (equivalent to EM) in situations that are more representative of patient use. In Part 1A (see Table 2), the purpose is to enable the determination of metrics for which a direct comparison can be made with and without the add-on device. This methodology is the same as that described using either the Andersen Cascade Impactor without pre-separator or the Next Generation Impactor without pre-separator (see 〈601〉) (USP 1-Dec-2019) and is therefore not repeated here. In Part 1B (see Table 2), delayed sampling is introduced because add-on devices, especially VHCs, are widely prescribed for individuals who, for whatever reason, are unable to coordinate MDI actuation with the onset of inhalation as described for the MDI alone in “patient information” and “instructions for use”. All of the tests in Part 1 are undertaken with the facemask removed from the spacer/VHC equipped with this type of patient interface. This modification is allowed so that the device can be readily interfaced by fitting the facemask adapter directly to the entry of the induction port without the complication of a direct facemask-to-induction port connection, where internal dead space would likely be both ill-defined and unrepresentative of the “in-use” condition.
Table 2. In Vitro Tests for Spacers and VHCs
| Test | Delay at Constant Flow Rate | Breathing Simulation | Suitability | Relevance to Patient Use Scenarios |
| Part 1A: Measurement of APSD with 4-L sample volume | None | Not applicable | Spacers and VHCs | Baseline performance compared with MDI alone |
| Part 1B: Measurement of APSD with 4-L sample volume | For example, 2, 5, or 10 sᵃ | Not applicable | VHCs only | Simulation of a delay by a poorly coordinated user |
| Part 2A: (USP 1-Dec-2019) Measurement of EM | Not applicable | Filter collection of aerosol simulating tidal breathing in coordinated (USP 1-Dec-2019) use | Spacers and VHCs | Simulation of optimum use (fully coordinated) with pMDI (USP 1-Dec-2019) actuation |
| Part 2B: Measurement of EM with sufficient number of inhalations from the VHC following guidance in the patient instructions to ensure that the drug is delivered from the VHC as intendedᵇ | Not applicable | Filter collection of aerosol simulating tidal breathing in uncoordinated use | VHCs only | Simulation of fully uncoordinated use with pMDI actuation (USP 1-Dec-2019) |
| Part 3A: (USP 1-Dec-2019) Measurement of EM delivered by facemask with sufficient number of inhalations from the VHC following guidance in the patient instructions to ensure that the drug is delivered from the VHC as intendedᵇ | Not applicable | Filter collection of aerosol behind lips or at nares of model, simulating tidal breathing in coordinated (USP 1-Dec-2019) use | Spacers and VHCs | Simulation of optimum use (fully coordinated) with pMDI (USP 1-Dec-2019) actuation. (USP 1-Dec-2019) Simulation of use with facemask applied with a 1.6-kg forceᶜ |
| Part 3B: Measurement of EM delivered by facemask with sufficient number of inhalations from the VHC following guidance in the patient instructions to ensure that the drug is delivered from the VHC as intendedᵇ | Not applicable | Filter collection of aerosol behind lips or at nares of model, simulating tidal breathing in uncoordinated use | VHCs only | Simulation of fully uncoordinated use with pMDI actuation. Simulation of use with facemask applied with a 1.6-kg forceᶜ (USP 1-Dec-2019) |
ᵃ Other delay intervals may also be used as required, if justified.
ᵇ Representative breathing patterns for different patient ages are listed in Table 3; other patterns may be used as required, if justified.
ᶜ 1.6-kg applied force has been shown to be clinically appropriate, but the force used may differ from this value if justified.
The tests in Parts 2 and 3, depending on whether a mouthpiece or facemask is present as the patient interface, respectively (see Table 2), evaluate the performance of the inhalation valve (and exhalation valve, if equipped) of the add-on device. This evaluation is accomplished by simulating tidal breathing, with parameters that are appropriate for the intended user group (e.g., infant, small child, or adult). This testing approach creates the continually varying flow conditions that are expected in use. Note that in the determination of EM, a sufficient number of inhalations should be taken in accordance with the instructions for use of the VHC, to ensure that the intended amount of drug is delivered.
Spacers/VHCs using a facemask as the patient interface should be evaluated ideally with the facemask in place (see 1.3 Rationale). The desirable approach is such that in vitro testing makes use of a model face that is representative of the age range for which the spacer/VHC is intended. There are currently limited commercially available model faces; however, the peer-reviewed literature contains several articles providing the necessary technical information to enable suitable age-appropriate model(s) to be created for the purpose of testing spacer/VHCs. It is also important that the surfaces of the face coming into contact with the facemask have mechanical characteristics (e.g., deformability) that are representative of the skin and underlying soft tissues when the facemask is applied to the face with an appropriate force. In Part 3 (see Table 2), the spacer/VHC is evaluated by a breathing simulation with the facemask fitted, mimicking similar fully coordinated use as defined in Part 2A and also fully uncoordinated use as defined in Part 2B (see Table 2). By comparing the EM of the drug with and without the use of a facemask, it is possible to assess the influence of the facemask.
[Note—Not all of the tests described in this chapter are applicable to open-tube spacers because such devices require fully coordinated use; otherwise, drug delivery will be significantly reduced. Figure 4 shows the decision tree to be followed so that the tests chosen are appropriate to the type of add-on device being evaluated.]
Table 2 further defines the test, method, applicable device, and relevance to patient use scenarios of the procedures that are described in this chapter. Example delay intervals of 2, 5, and 10 s are noted in the table, although other delay intervals may be substituted or added as required and justified. For the testing in Parts 3A and B (USP 1-Dec-2019) (see Figure 4), it is recognized that currently there are limited commercially available face models representing the most widely recognized age categories, which may be required (i.e., infant, small child, and adult). The user is advised to select an appropriate commercial model or develop his or her own, providing justification for the selection as required. Additionally, Table 3 shows representative tidal-breathing patterns for the various patient age categories from neonate to adult.
Table 3. Representative Tidal Breathing Patternsᵃ
| Pediatric | Adult | ||||
| Parameter | Neonate | Infant | Small Child | Normal 1ᵇ | Normal 2ᵇ |
| Tidal volume (mL) | 25 | 50 | 155 | 770 | 500 |
| Frequency (min⁻¹) | 40 | 30 | 25 | 12 | 13 |
| I/E ratioᶜ | 1:3 | 1:3 | 1:2 | 1:2 | 1:2 |
| Minute volume (mL) | 1000 | 1500 | 3875 | 9240 | 6500 |
ᵃ With the permission of the Canadian Standards Association (operating as the CSA Group), material is reproduced from CSA Group standard CAN/CSA-Z264.1-02 (R2011), “Spacers and Holding Chambers for Use with Metered-Dose Inhalers”, which is copyrighted by CSA Group, 5060 Spectrum Way, Suite 100, Mississauga ON, L4W 5N6. This material is not the complete and official position of the CSA Group on the referenced subject, which is represented solely by the standard in its entirety. Although use of the material has been authorized, CSA is not responsible for the manner in which the data are presented, nor for any interpretations thereof.
For more information or to purchase standards from the CSA Group, please visit shop.csa.ca/ or call 1-800-463-6727.
ᵇ Normal 1 represents a resting adult of large build; Normal 2 represents an active adult of normal build.
ᶜ I/E ratio = inspiratory time/expiratory time.
Testing is undertaken (see 3.1 No Delay between the MDI Actuation and Sampling Onset) to ensure that there are no leakages of ambient air into the measurement apparatus. The purpose is not to evaluate leakages associated with the spacer/VHC. Such testing would be impractical because of the variety of adapters that would be needed to interface the flow meter with the different designs associated with these add-on devices. (USP 1-Dec-2019)
Change to read:
3 MEASUREMENT OF APSD
3.1 No Delay between the MDI Actuation and Sampling Onset
Background
In Part 1A (see Figure 4), the measurement of APSD from the spacer or VHC with no delay following actuation of the inhaler is defined as an important test of the optimum performance of the device. This test also provides information with which to compare the in vitro performance of the MDI with and without the add-on device present.
Test protocols
If the spacer/VHC is intended for adult use, and comparison is being made with the MDI alone, follow one of (USP 1-Dec-2019) the procedures given for inhalation aerosols and sprays, (USP 1-Dec-2019) as required. Alternatively, the pediatric version of (USP 1-Dec-2019) the Marple-Miller Cascade Impactor (Model 150P) (USP 1-Dec-2019) may be used.
If the spacer/VHC is intended for neonates, infants, or small children, it may be appropriate to use an alternative apparatus that operates at the reduced flow rate(s) more appropriate for these classes of patients. Examples are the Next Generation Impactor (without pre-separator) (USP 1-Dec-2019) operated at 15.0 L/min or the pediatric version of (USP 1-Dec-2019) the Marple-Miller Cascade Impactor (Model 150P) that functions at either 4.9 or 12.0 L/min.
Set up the cascade impactor as described in the manufacturer's literature. To ensure efficient particle capture, coat appropriately the particle collection surface of each stage with Glycerol, silicone oil, or other suitable liquid unless this has been demonstrated to be unnecessary. Connect an induction port with internal dimensions as defined in 〈601〉 to the impactor inlet. Connect the impactor to a vacuum pump via a suitable flow control valve.
Attach a flowmeter capable of providing volumetric flow rate, within the range of operation leaving the meter, to the induction port, ensuring an airtight seal. Verify that the flow rate for the test is within ±5% of the specified flow rate. This is achieved by applying a vacuum to the test apparatus by means of the flow control valve located between the cascade impactor and vacuum source. Turn off the source of vacuum to this apparatus after this check has been completed and remove the flow meter.
Attach a suitable coupling for the spacer/VHC to the induction port. (USP 1-Dec-2019) Conduct the remainder of the (USP 1-Dec-2019) evaluation with the mouthpiece of the spacer or VHC, if so equipped, connected to the test apparatus. If the spacer or VHC is equipped with a facemask, remove the facemask and perform the evaluation with the add-on device connected to the test apparatus by means of the facemask adapter.
Prepare the spacer/VHC (USP 1-Dec-2019) for the test by washing in accordance with the manufacturer's instructions, if so indicated. If no instructions for preparation are provided, test the device out of the package without prewashing, and note this information in the test report.
Connect the spacer or VHC via the mouthpiece or facemask adapter to the entry to the induction port described in 〈601〉. This connection can be made either with a short piece of flexible tubing or with a purpose-built coupling that ensures on-axis alignment with the entry to the induction port. If flexible tubing is used for this connection, ensure that the add-on device is supported in such a way that its long axis aligns with the axis of the induction port entry. Care should be taken to ensure that the coupling, however constructed, does not result in a significant loss of the drug and/or change in measured APSD.
(USP 1-Dec-2019)
The practice of actuating the MDI with the spacer or VHC initially disconnected from the induction port and then rapidly connecting the add-on device after MDI actuation is not recommended, because there is always a small delay interval and medication will be lost in an uncontrolled manner from an open-tube spacer if this procedure is followed.
Follow the directions in the patient information and instructions for use for the MDI to prime it before inserting into the MDI adapter of the spacer/VHC. In the case of an add-on device equipped with an integral actuator, after priming the MDI canister in its mouthpiece adapter as supplied, remove the canister from the adapter and clean the external valve stem with a suitable wipe. Insert the canister valve stem into the receptacle following the instructions for use for the particular add-on device, taking care to avoid premature actuation.
In cases where more than one actuation of the MDI is needed to collect sufficient mass of drug product, follow the directions in the patient information and instructions for use; if no directions are provided, allow a minimum of 30 s between actuations. Turn on the vacuum to the cascade impactor before delivering any actuations. It is important to deliver the minimum number of actuations to obtain an adequate sample of the size-fractionated drug mass for measurements to be made to the same degree of precision as would be the case if the MDI was evaluated alone.
After the MDI actuation and aerosol sampling are completed, recover the drug from each component of the cascade impactor, including the induction port, and also from within the spacer/VHC by using a validated method appropriate to the product being measured. Carefully remove the backup filter of the cascade impactor, whose purpose is to capture the drug mass passing the last impaction stage, from its location and place it in a suitable container. For each sample, add a predetermined volume of a suitable solvent to the container and agitate to dissolve the collected drug. Transfer a sample to a cuvette or vial for drug assay using a syringe equipped with an in-line filter to retain any suspended material. Care must be taken with the recovery of a drug retained by the add-on device, because a greater volume of solvent may be needed, compared with volumes needed for recovery from each impaction plate/cup of the impactor. Perform separate assays to measure the mass of drug recovered from each component, using a validated procedure.
Repeat the measurement with the required number of devices and replicates per device. Typically, five separate spacers or VHCs are each tested once, but other testing protocols may be adopted as required, if justified. These protocols may include replicate measurements per device to obtain information about intra- as well as inter-device performance.
Measurements
Total mass of drug collected: This procedure is not a test of the product or add-on device but serves as a system suitability test to ensure that the results are valid. The total mass of drug collected comprises the sum of the drug mass in all of the components, including the MDI mouthpiece and from within the spacer/VHC [material balance (MB)] divided by the number of actuations of the MDI. Note that typically up to 5% of the target-delivered label claim may be retained by the cascade impaction apparatus as inter-stage wall losses.
Total mass of drug emitted: The total mass of drug emitted from the spacer/VHC with zero seconds delay (delivered dose, equivalent to EM) is determined from the sum of the values for the mass of drug collected in all of the components of the test apparatus divided by the number of actuations of the MDI. Spacers and VHCs are intended to reduce the mass of coarse particles inhaled by the patient; therefore, an important part of data interpretation of the APSD should involve separate assessments of the pertinent subfractions relevant to the inhalation aerosol product. The precise size limits for these subfractions will agree with the recipient of the test data. However, to provide initial guidance on this matter, it is suggested that the following be considered as a minimum:
- Determination of the mass of drug retained by the spacer/VHC together with the mass recovered from the induction port
- Stage-by-stage profile of the remaining EM
Further interpretation of these data is at the discretion of the user. The performance of the spacer/VHC is compared with that of the MDI alone, following the procedure described in 〈601〉.
3.2 Delay between the MDI Actuation and Sampling Onset
[Note—This test is inappropriate for spacers.]
Background
This test requires the adaptation of the Andersen Cascade Impactor without pre-separator and the Next Generation Impactor without pre-separator (see 〈601〉) (USP 1-Dec-2019) to accept a means by which the delay interval can be realized. The actuation of the MDI into the VHC on test with its mouthpiece or facemask adapter disconnected from the entry to the induction port, followed by connection to the port with the vacuum applied to the apparatus immediately after the delay interval has elapsed, is difficult to perform accurately and may result in leakage or an inaccurate realization of the delay interval. For these reasons, an apparatus of the type described in Figure 5 is recommended, because it enables the VHC mouthpiece/facemask adapter to remain in position throughout the measurement. The methodology for delay testing is based on the apparatus illustrated, but other equipment offering similar capability may be used if justified.
Test protocols
The vertically mounted shutter plate comprises a circular opening in its upper half and is mounted at setup such that the gap between the VHC mouthpiece/facemask adapter and induction port entry is closed, as illustrated (see Figure 5). The induction port and VHC adapter fit tightly into the mounting block. In this position, when vacuum is applied to the test apparatus, air is sampled by the apparatus at the desired flow rate via a bypass channel on the side of the adapter facing the induction port. This arrangement avoids the need to start the flow through the apparatus after the delay interval has elapsed, so that the cascade impactor is always operated at a constant flow rate.
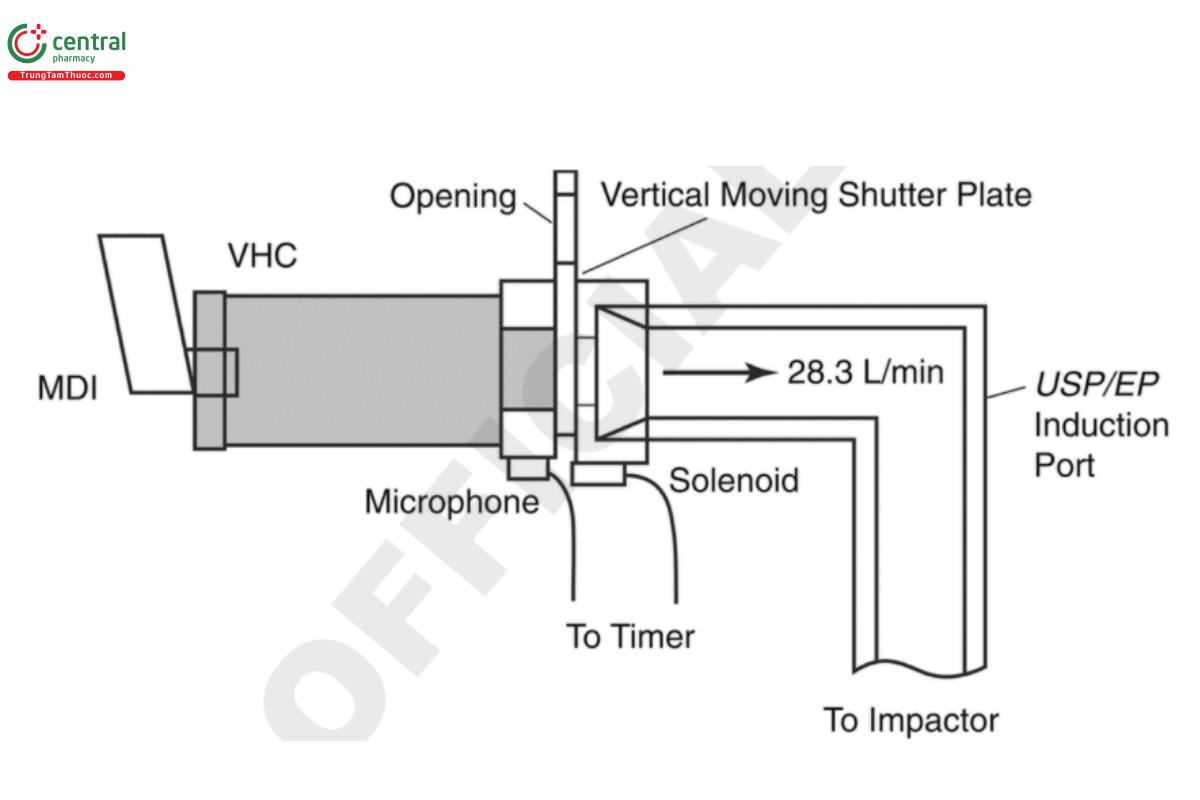
In the configuration described in Figure 5, when the MDI is actuated, a microphone located on the adapter block detects the sound emitted at actuation of the MDI, starting a timer that operates a solenoid valve that retracts a pin immediately after the preset delay interval has expired. This process permits gravity to operate on the shutter, which drops to the lower position in which the aperture is aligned with the VHC mouthpiece/facemask adapter and induction port entrance. The aerosol retained within the VHC is sampled as soon as the shutter moves to the “open” position. This procedure avoids the risk of capturing any “blow-by” aerosol that might escape the VHC as the propellant expands immediately following MDI actuation but would not be inhaled by a user. If blow-by is observed, it should be noted in the test report. In the configuration shown in Figure 5, the adapter introduces <5 mL of additional volume to the aerosol pathway from the VHC to the filter, and the minimum delay interval achievable is 1 s.
Example delay intervals of 2, 5, and 10 s have been noted earlier in this chapter, although other delay interval(s) may be substituted or added if required and justified.
Connect the impactor to a vacuum pump via a suitable flow control valve.
The delay apparatus, if constructed in accordance with Figure 5 will introduce leakage into the apparatus, once attached to the induction port because the bypass flow passage is open in order that in-flow comes in via this passage rather than through the VHC before the shutter drops after the prescribed delay period. The flow rate verification test is therefore conducted before the delay apparatus is attached. Connect an induction port with internal dimensions as defined in (601) to the impactor inlet. Attach a flowmeter capable of providing volumetric flow rate, within the range of operation leaving the meter, to the coupling, ensuring an airtight seal. Verify that the flow rate for the test is within ±5% of the specified flow rate. This is achieved by applying a vacuum to the test apparatus by means of the flow control valve located between the cascade impactor and vacuum source. Turn off the source of vacuum to this apparatus after this check has been completed and remove the flow meter. (USP 1-Dec-2019)
Securely attach the exit port from the delay apparatus to the entry of the induction port. Conduct the remainder of the evaluation with the mouthpiece of the spacer or VHC, if so equipped, connected to the test apparatus. If the spacer or VHC is equipped with a facemask, remove the facemask and perform the evaluation with the add-on device connected to the test apparatus by means of the facemask adapter. If this is the first testing of the spacer/VHC or the intention is to evaluate the add-on as new for each delay condition, prepare it for the test by washing in accordance with the manufacturer's instructions, if so indicated. If no instructions for preparation are provided, test the device out of the package without prewashing, and note this information in the test report. (USP 1-Dec-2019) Connect the VHC via the (USP 1-Dec-2019) patient interface (mouthpiece or facemask adapter) to the entry port of the delay apparatus.
Before actuating the MDI for the first time into the VHC being tested, ensure that the delay apparatus is set such that the shutter is in the "up" or "closed" position. Select the desired delay interval for the timer.
Follow the directions in the patient information and instructions for use before inserting the actuator/mouthpiece of the MDI into the MDI adapter of the VHC before the first actuation into the test apparatus. If the spacer/VHC is designed with an integral actuator, after priming the MDI canister in its mouthpiece adapter as supplied, remove the canister from the adapter and clean the external valve stem with a suitable wipe. Insert the canister valve stem into the receptacle following the instructions for the particular add-on device, taking care to avoid premature actuation.
In cases where more than one actuation of the MDI is needed to collect sufficient mass of drug product, follow the directions in the patient instructions, or if no direction is provided, allow a minimum of 30 s between actuations. Deliver the minimum number of actuations needed to obtain an adequate sample of the size-fractionated drug mass so that measurements can be made to the same degree of precision as if the MDI was evaluated alone. Reposition the shutter of the delay apparatus in the up or closed position, and check that the timer is set for the desired delay after each MDI actuation. Maintain the vacuum through the entire sequence until all actuations are completed.
After the sampling part of the measurement is completed, recover the drug from each component of the cascade impactor, including the induction port, and from within the spacer/VHC by using a validated method appropriate to the specific product. In the configuration shown in Eigure 5, the surface area of the shutter that is exposed to aerosol particles is minimal. Therefore, recovery of drug from this component needs to be done only for the most accurate work; in which case, the inner surfaces of the aperture in the shutter plate and the exit port are washed with recovery solvent. Care should be taken with the recovery of drug retained by the VHC, because a greater volume of solvent than is needed for recovery from each impaction plate/cup of the impactor may be needed to be sure that drug is quantitatively recovered. Assay for the mass of drug recovered from each component separately by using a validated procedure.
Repeat the measurement with the required number of devices and replicates per device. Typically, five separate spacers or VHCs are each tested once, but other designs, including replicate measurements per device, may be adopted as required.
Measurements
Total mass of drug collected: The total mass of drug collected in all of the components, including the MDI mouthpiece and from within the spacer/VHC (MB) is divided by the number of actuations of the MDI. The component of the dose that may escape past the inhalation valve of the VHC on MDI actuation (due to momentary pressurization of the interior of the chamber) is termed "blow-by", and it may not be captured for the assay. If this phenomenon is observed, it must be noted. [NOTE-Quantification of blow-by is not a practical proposition because the act of collecting the aerosol will inevitably apply back-pressure to the valve, reducing or eliminating the phenomenon altogether.] The performance of the spacer/VHC tested with delay (see 3.2 Delay between the MDI Actuation and Sampling Onset) is compared with that of the spacer/VHC tested with no delay (see 3.1 No Delay between the MDI Actuation and Sampling Onset). VHCs are intended to reduce the mass of coarse particles inhaled by the patient, so an important part of data interpretation of the APSD should involve separate assessments of the pertinent subfractions relevant to the inhalation aerosol product. The precise size limits for these subfractions will agree with the recipient of the test data. However, to provide initial guidance on this matter, it is suggested that the following be considered as a minimum:
- Determination of the mass of drug retained by the spacer/VHC together with the mass recovered from the induction port
- Stage-by-stage profile of the remaining EM
Further interpretation of these data is at the discretion of the user.
Change to read:
4 MASS OF DRUG DELIVERED FROM A SPACER/VHC WHILE SIMULATING PATIENT TIDAL BREATHING
4.1 Without Facemask
Cascade impactors used in the assessment of APSD are designed to operate at fixed flow rates. However well the add-on device may perform during an in vitro test of function using such an apparatus, the operation of critical moving components (i.e., inhalation and exhalation valves of VHCs) is not evaluated in the way that these components would perform when the VHC is used by the patient.
additional test that simulates tidal breathing is therefore included, because this type of respiratory pattern is most commonly encountered with patients using an add-on device, particularly neonates, infants, and children who are incapable of a forced inhalation maneuver. Several representative, age-related breathing patterns are listed in Table 3.
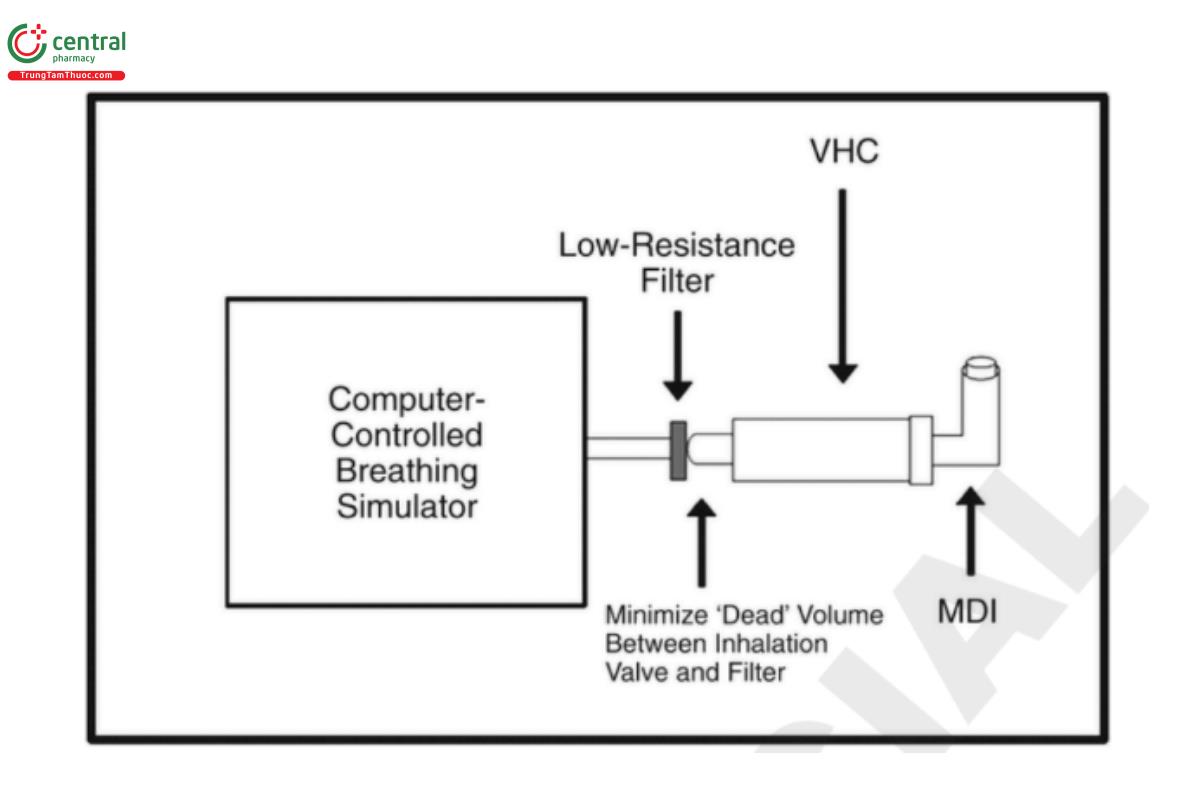
There is no breathing simulator recognized in 〈601〉. However, 〈1601〉 provides for the use of a commercially available breathing simulator that is able to generate the same breathing profiles as those specified in Table 3. The methodology provided assumes that equipment meeting this standard is available. Figure 6 illustrates schematically a setup that has been effective for these measurements, but other configurations may be used as required.
The filter system used to collect the aerosol at the mouthpiece/facemask adapter of the spacer/VHC should be a suitably validated, low-resistance filter capable of quantitatively collecting the aerosol and enabling recovery of the drug substance with use of an appropriate solvent. If the filter is contained in its own housing, the dead volume of the filter casing must not exceed 10% of the tidal volume used in the breath simulation. This restriction will likely make it necessary to use filters without a separate housing when performing tests that simulate neonatal or infant use.
The first part of the test is suitable for both spacers and VHCs, because it simulates the delivery of medication mimicking a fully coordinated user actuating the MDI at the onset of inhalation. However, the second part of the test is only suitable for VHCs because the MDI is actuated at the onset of exhalation to simulate a fully uncoordinated user.
Test, part 1
Set the breathing simulator to the required breathing pattern (identified from Table 3) in accordance with the manufacturer's instructions. Alternative breathing patterns may be used if required. Ensure that the simulator has been calibrated before use so that the actual volumes and frequency are within ±5% of the indicated value during each breathing cycle. Calibration syringes are available from various suppliers of mechanical ventilation equipment to verify volumes. To verify frequency, the method varies among breathing simulator manufacturers, and therefore the advice of the particular supplier/manufacturer should be sought if the method is not explicitly stated in the operating instructions for the apparatus.
Ensure that the selected flow waveform from the computer-controlled breathing simulator (Figure 6) is stable before proceeding. Prepare each device in the group of spacers or VHCs to be tested by washing in accordance with the manufacturer's instructions, if indicated. If no instructions for preparation are provided, test the devices out of package without prewashing and note this fact in the test report.
Connect the mouthpiece or facemask adapter of the spacer/VHC to the breathing simulator by means of a short length of flexible hose. Locate the aerosol filter as close as possible to the mouthpiece of the spacer/VHC using an adapter or other means that will enable a leak-tight seal to be formed with the minimum amount of space (dead volume) between the device and the filter. Ensure that the adapter does not restrict the path of the aerosol.
Filtrete is a suitable electret filter medium (USP 1-Dec-2019) that readily releases collected drug quantitatively upon addition of a suitable solvent during the assay procedure. However, other suitable filter media may be used provided that the tester is satisfied that the filter is capable of collecting the entire emitted dose and that drug recovery for assay is quantitative.
Follow the directions in the patient information and instructions for use for the MDI to prime it before inserting into the MDI adapter of the spacer/VHC before the first actuation into the test apparatus. In the case of an add-on device equipped with an integral actuator, after priming the MDI canister in its mouthpiece adapter as supplied, remove the canister from the adapter and clean the external valve stem with a suitable wipe. Insert the canister valve stem into the receptacle by following the instructions for use for the particular add-on device, taking care to avoid premature actuation. This test typically should require only one actuation of the MDI into the spacer/VHC per determination. However, for certain highly potent products delivering low unit mass of drug per actuation, more than one actuation of the MDI may be needed to collect a sufficient mass of drug product. Under such circumstances, follow the directions in the patient information and instructions for use for the MDI, or allow a minimum of 30 s between actuations if no direction is provided.
For the first part of the test, perform a single actuation timed to coincide with the beginning of an inhalation. Allow sampling to occur for five additional breathing cycles for the actuation. If additional actuations are required to improve analytical sensitivity, ensure that the sampling time is of sufficient length for the spacer/VHC to be emptied of remaining aerosol before the next actuation is delivered, and do not disconnect the spacer/VHC from the breathing simulator between actuations.
Remove the filter carefully from its location and place it in a suitable container. Add a predetermined volume of a suitable solvent to the container and agitate to dissolve the collected drug. Transfer a sample to a cuvette or vial for drug assay using a syringe equipped with an in-line filter to retain any suspended material. Recover the mass of drug from the filter by using a validated procedure appropriate to the specific product, and assay for the mass of drug collected by using a validated procedure. Calculate the total mass of drug per actuation; this is the emitted mass in the fully coordinated condition (EMc). Repeat the measurement with the required number of devices and replicates per device.
Test, part 2
[Note—This part of the test is inappropriate for spacers.] For the second part of the test, either clean the VHC between measurements or evaluate a new VHC out of its packaging, as required and justified. Report whether cleaning or replacement of the VHC was carried out. Repeat the procedure above in Test, Part 1 with the same VHC, this time actuating the MDI timed to coincide with the onset of exhalation. Calculate the total mass of drug per actuation; this is the EM in the fully uncoordinated condition (EMuc). Repeat the measurement with the required number of devices and replicates per device. Calculate the ratio of EMc with the EMuc.
4.2 With Facemask
Test, part 1
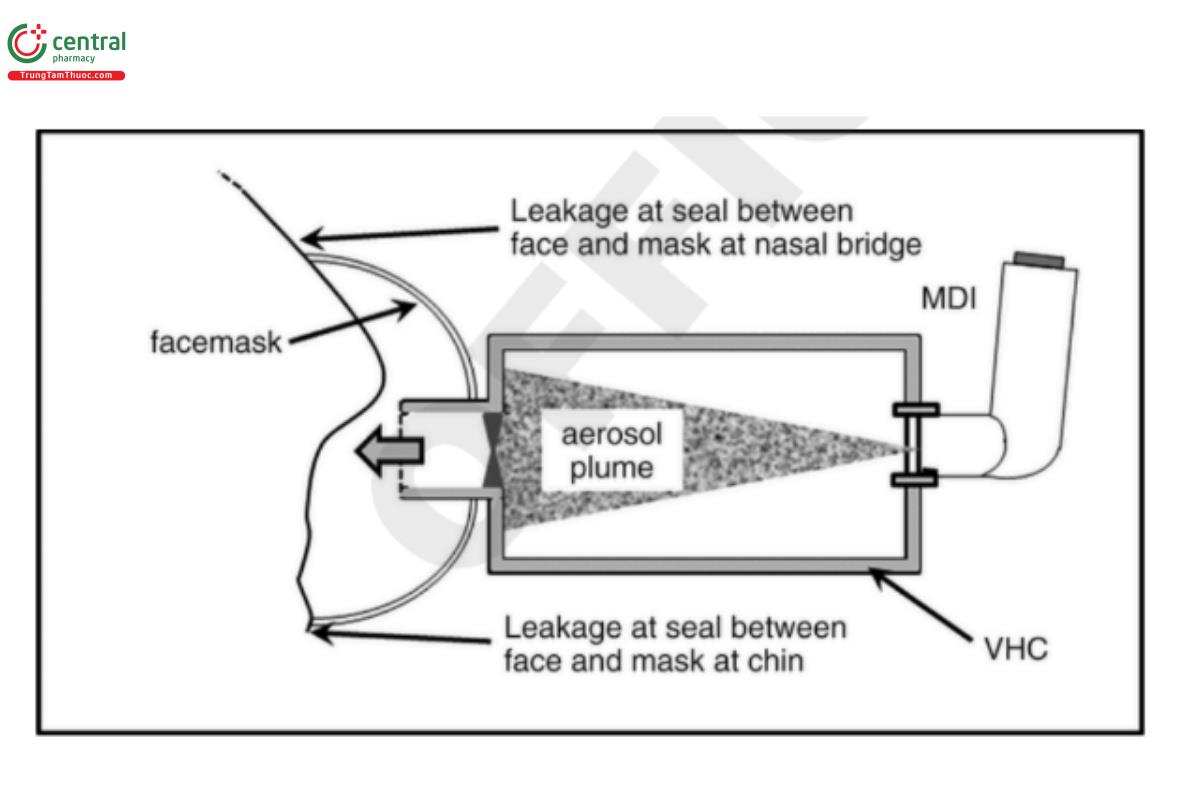
The purpose of this test is to compare the EM from a spacer/VHC equipped with a facemask to that obtained in the fully coordinated simulation with the facemask removed. Facemasks are widely prescribed for infants, small children, and adults who lack coordination to use a mouthpiece-based product. The evaluation of spacers/VHCs supplied with a facemask, rather than a mouthpiece, requires additional attention beyond the methods described previously, because the facemask itself has a major part to play in the transport of the drug-containing aerosol particles from the add-on device to the patient (Figure 7). It is therefore important that the facemask be tested in situ as part of the add-on device, rather than separately. A critical component of the test apparatus is the representation of a human face appropriate to the age range for which the add-on device is intended (e.g., neonate, infant, small child, or adult). There are few models that can be defined as an apparatus in a way that is similar to what is done for cascade impactors in the aerodynamic particle size analysis of MDI-generated aerosols, although some face models are currently available commercially. Ideally, standardized, age-appropriate models would be preferred. (USP 1-Dec-2019)
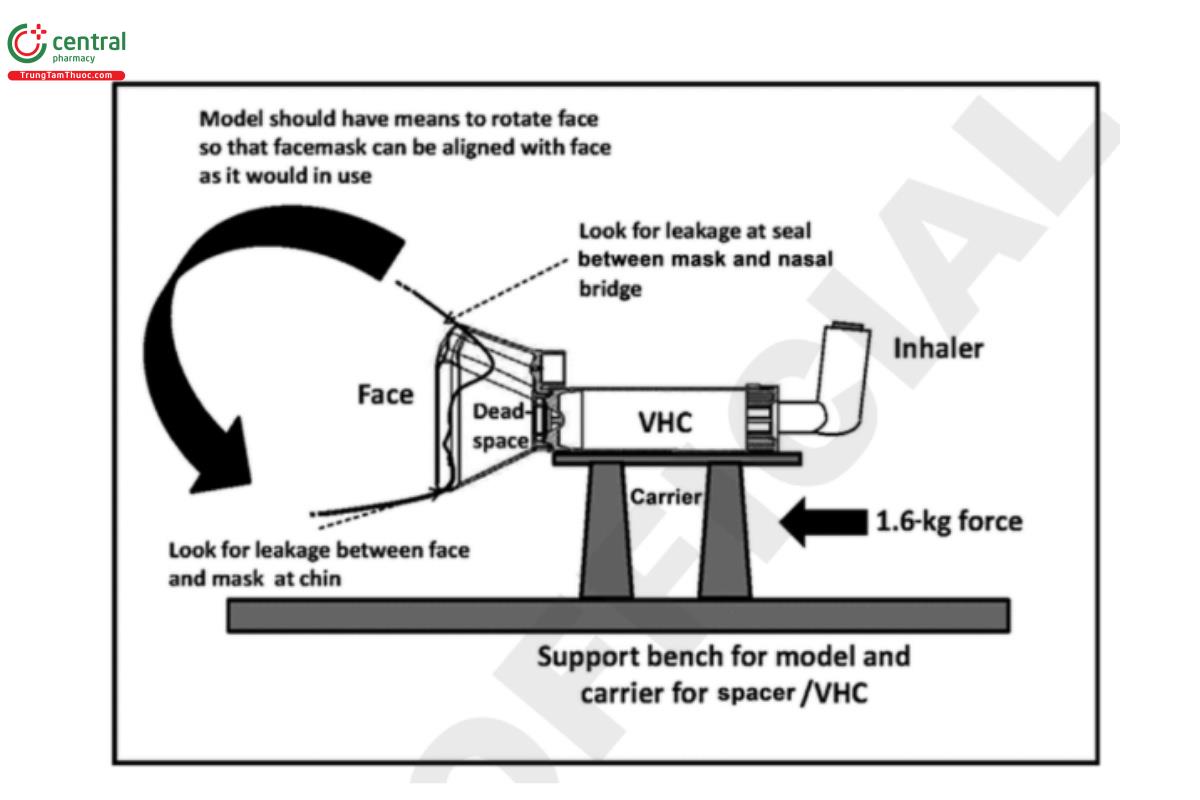
Ideal attributes for face models are the following:
- Appropriate facial dimensions for the intended user age range
- Ability to apply the facemask with the predicted amount of dead space when it is applied with a clinically appropriate force to the model
- Physiologically accurate soft facial tissue modeling around the chin, cheeks, and nose where the facemask makes contact
- Means of correctly mounting the spacer/VHC so that the facemask is oriented with the correct alignment to the face, as would occur when in use by a patient
[Note—Some models may include anatomically accurate realization of the upper airway (naso- or oropharynx), with the aerosol collection filter located at the distal port of the model representing the entry to the lungs.]
For the sake of simplicity and to realize a measure of spacer/VHC performance comparable to the measure obtained with facemask removed in Parts 2A and B (USP 1-Dec-2019) (see Eigure 4), the default assumption is that the model face simulates open-mouth breathing without an anatomically accurate upper airway, and that the aerosol collection filter is located in a cavity immediately behind the lips of the model. Alternatively, if an obligate nasal breathing infant model is being used, then the filter can be placed behind the nares. The mass of drug is reported as the emitted mass (EM facemask).
Select a face model appropriate to the intended age range for the spacer/VHC facemask, and mount the face model in an appropriate fixture. The fixture should enable the facemask to be located at an appropriate angle to the face model, such that an effective seal between the facemask and face model is created with a clinically appropriate force. This is typically a loading force of 1.6 kg but may differ from this value if justified by the design of the facemask. The arrangement shown schematically in Figure 8 is one way of achieving the desired result, but other approaches may be adopted as required, if justified.
Locate and secure the filter media in the cavity behind the lips of the face model. Filtrete is a suitable electret filter medium (USP 1-Dec-2019) that readily releases collected drug quantitatively upon addition of a suitable solvent during the assay procedure. However, another suitable filter medium may be used, provided that the tester is satisfied that it is capable of collecting the entire emitted dose, and that drug recovery for the assay is quantitative.
Verify that a seal has been obtained between the facemask and face model. This is conveniently done by connecting the outlet (distal) port of the face model, using a short length of flexible hose, to an in-line calibrated gas flow meter whose distal port is in turn connected to the vacuum source via a regulating valve capable of setting the flow rate to a suitable value, typically 30 L/min. Connect a similar flow meter to the port for the MDI (MDI adapter) on the spacer/VHC, and ensure that the mask is positioned in order to maximize flow rate. (USP 1-Dec-2019)
[NOTE-Leakage from the facemask to face seal, as detected by lower flow rate, is known to reduce drug delivery.]
Turn off the vacuum source after this check has been completed, and disconnect the flexible hose from the in-line flow meter. Remove the upstream flow meter from the MDI adapter of the spacer/VHC.
Connect the outlet (distal) port of the face model to the appropriate port of the breathing simulator using a short length of flexible hose. Set the breathing simulator to the required breathing pattern (identified from Table 3) in accordance with the manufacturer's instructions. Alternative breathing patterns may be used if required. Ensure that the simulator has been calibrated before use so that the actual volumes and frequency are within ±5% of the indicated value during each breathing cycle. Calibration syringes are available from various suppliers of mechanical ventilation equipment to verify volumes. To verify frequency, the method varies among breathing simulator manufacturers, and therefore the advice of the particular supplier/manufacturer should be sought if the method is not explicitly stated in the operating instructions for the apparatus. Ensure that the selected flow waveform from the breathing simulator is stable before proceeding.
Follow the directions in the patient information and instructions for use for the MDI to prime it before inserting it into the MDI adapter of the spacer/VHC before the first actuation into the test apparatus. In the case of an add-on device equipped with an integral actuator, after priming the MDI canister in its mouthpiece adapter as supplied, remove the canister from the adapter, and clean the external valve stem with a suitable wipe. Insert the canister valve stem into the receptacle according to the instructions for use for the particular add-on device, taking care to avoid premature actuation. Typically, it should require only one actuation of the MDI into the spacer/VHC per determination. However, for certain highly potent products delivering a low-unit mass of drug per actuation, more than one actuation of the MDI may be needed to collect a sufficient mass of drug product. Under such circumstances, follow the directions in the patient instructions or allow a minimum of 30 s between actuations if no direction is provided. Deliver the minimum number of actuations needed to obtain an adequate sample of the drug so that mass measurements can be made with acceptable precision.
Perform a single actuation in the spacer/VHC, timed to coincide with the beginning of an inhalation (see Figure 4, Part 3A). Allow sampling to occur for five additional breathing cycles for the actuation. If additional actuations are required to improve analytical sensitivity, ensure that the sampling time is of sufficient duration for the spacer/VHC to be emptied of remaining aerosol before the next actuation is delivered, and do not disconnect the spacer/VHC from the breathing simulator between actuations.
Following the tests, remove the filter carefully from its location and place it in a suitable container. Add a predetermined volume of a suitable solvent to the container and agitate to dissolve the collected drug. Transfer a sample to a cuvette or vial for drug assay using a syringe equipped with an in-line filter to retain any suspended material. Recover the mass of drug from the filter by using a validated method appropriate to the specific product, and assay for the mass of drug collected by using a validated procedure. Calculate the total mass of drug per actuation; this is the emitted mass in the fully coordinated condition with facemask fitted to the spacer/VHC (EMc-fm). Repeat the measurement with the required number of devices and replicates per device.
The values of EMc from the test undertaken in Part 2A (USP 1-Dec-2019) (see Figure 4) in which the facemask adapter was removed for comparison of EM, simulating fully coordinated use by a breathing simulator can be compared to the EMc-fm in order to understand the impact of the facemask on drug delivery.
Test, part 2
For a VHC only, the same process can be repeated, but with the MDI actuation(s) timed to coincide with the beginning of exhalation (rather than inhalation) (see Figure 4, Part 3B). In this instance, the mass of drug collected per actuation is the emitted mass in the uncoordinated condition (EMuc–fm). The ratio of EMc–fm from Part 1 of this test (USP 1-Dec-2019) and the EMuc–fm can now be calculated.
(USP 1-Dec-2019)


