SCREENING FOR UNDECLARED DRUGS AND DRUG ANALOGUES
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
- INTRODUCTION
- ADULTERATION CATEGORIES
- BULK INGREDIENTS AND DOSAGE FORMS
- RECOMMENDED ANALYTICAL METHODOLOGIES
- APPENDIX
- APPENDIX A. SCREENING METHODOLOGIES FOR PDE5 INHIBITORS
- HPLC with Photodiode Array Detection
- HPLC with Mass-Spectrometric Detection
- High Performance Thin-Layer Chromatography (HPTLC) with Visual, UV, and/or MS Detection
- Ambient lonization Mass Spectrometry
- NMR Spectroscopy-Low-Field and High-Field
- Bioassay⁸
- USP Reference Standards 〈11〉
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
The illegal addition of undeclared synthetic compounds to products marketed as dietary supplements (DS) is an issue of universal concern. This fraud is practiced to impart therapeutic effects that cannot be achieved by the dietary ingredients alone. Increasingly, synthetic intermediates and structural analogues of the pharmaceuticals and drugs that have been discontinued or withdrawn from the market are being used as adulterants. Multiple adulterating compounds may be added to a single product, frequently in erratic amounts.
The proposed test methodologies facilitate screening for synthetic adulterants. No individual technique is capable of addressing all potential analytes; thus, a combination of orthogonal approaches adds certainty to the analytical outcome. Mass spectrometric techniques provide strong substantiation of the analytical findings. In some cases, e.g., with hormonal drugs, the amounts of physiologically relevant adulterants may be so low that GC-MS or LC-MS may be the only fitting analytical options.
The express purpose of assembling the procedures recommended herein is their suitability for screening. The level of evidence achievable by application of one or several of the recommended procedures is ultimately dictated by the specific requirements of the end-user. It should be noted that structure elucidation and quantitative assessment are beyond the scope of this chapter.
This chapter is meant to be updated regularly, as new concealment methodologies for the adulterants are introduced, or improvements to the methods of analysis are realized.
2 ADULTERATION CATEGORIES
The following major categories of adulterated products are recognized:
- Sexual Enhancement: This category is also referred to as the Erectile Dysfunction (ED) category. It encompasses a functionally coherent group of adulterants, including several approved drugs, their numerous approved and unapproved analogues, and synthetic intermediates. Their functionality is manifested by inhibition of phosphodiesterase type 5 enzyme (PDE5), which hydrolyzes cyclic guanosine 3',5'-monophosphate (cGMP); this group of compounds is frequently identified as PDE5 inhibitors. Screening methods for products adulterated with ED compounds are presented in Appendix A.
- Weight Loss (WL): This category comprises a functionally and chemically diverse collection of compounds that include stimulants, laxatives, diuretics, anorexiants, and psychoactive drugs. Although stimulants constitute an important segment of WL adulterants, the oral anorexiant sibutramine dominates this category, frequently in combination with phenolphthalein, a laxative. Methods for analysis of products adulterated with WL compounds will be addressed in Appendix B (to come).
- Sports Performance Enhancement (SPE): These compounds constitute the third major category of adulteration. Professional and amateur athletes are targeted with designer anabolic steroids and stimulants, which are systematically banned by the World Anti-Doping Agency. Functional and structural diversity, synthetic proclivity of the adulterators, and the generally small amounts of the infringing substances required to elicit a therapeutic effect make this category especially challenging to address. These supplements are customarily formulated in protein- and fat-rich matrices, thereby further complicating detection. For these reasons, GC- and LC-MS" techniques constitute primary analytical methodologies within this category. Analysis of products adulterated with SPE compounds will be addressed in Appendix C (to come).
3 BULK INGREDIENTS AND DOSAGE FORMS
Adulteration may occur either at the level of bulk ingredients or at any subsequent stage of the finished product manufacturing. Analysts should be mindful of the possibility of adulterants physically associated with the finished dosage matrix or excipients, as well as components. In the latter, synthetic compounds have been found embedded into the capsule shell body. This underscores the need for deliberate adjustment to the laboratory procedures that typically focus on the capsule contents alone. Appropriate sampling practices for powders and finished dosage forms should be exercised, particularly when only a limited amount of sample is available.
4 RECOMMENDED ANALYTICAL METHODOLOGIES
Adulteration analysis may be broadly categorized into targeted and nontargeted methods. The distinction between these types may be subtle, and a minor adjustment to the methodology will transform a nontargeted method into a targeted method.
4.1 Targeted
These techniques are warranted when the analytes are known. An example of a targeted approach would be monitoring a chromatographic run at a particular wavelength (or mass), and quantifying the analyte that appears within a predefined retention time window. Targeted analysis is conceptually straightforward, because it relies on pre-existing knowledge of the analyte and allows optimization of test methodology for its reliable detection. The targeted approach also is a rarity in the adulterated products analysis, where the nature of the analyte may be anticipated only tentatively, and variable amounts of multiple adulterants belonging to several functional categories are commonplace.
4.2 Nontargeted
These methods are better suited to a broad-spectrum detection requirement presented by adulterated products. Nontargeted screening trades precise knowledge of the analyte identity, along with specificity and accuracy, for a wider detection scope. Examples of nontargeted chromatographic screening include acquisition of photodiode array data and full mass-spectral scanning following a chromatographic separation. The procedures in this chapter are written with an eye toward applying all techniques in a nontargeted mode, even the ones considered to be inherently targeted, thereby facilitating detection of a suspect adulterant even in the absence of a matching reference compound.
It is generally recommended to apply a broadly nontargeted methodology first, followed by a targeted procedure. It is crucial to clearly define the end-purpose of analysis, and only then decide on the appropriate instrumentation and assemble a logical testing strategy from the procedures provided. Thus, application of a nontargeted screening method may satisfy the requirements of a manufacturer for the purposes of monitoring bulk raw materials. Conversely, a laboratory requiring a higher level of evidence to enact an enforcement action may opt for a two-step procedure: a preliminary screen, followed by confirmatory analysis of suspect samples.
USP Reference Standards recommended for screening are listed at the end of each relevant Appendix. However, considering the rate of propagation of structural analogues and proliferation of newly developed "designer" molecules, establishing and maintaining an all-inclusive catalog of reference materials is both challenging and impractical. Several commercial sources of the compounds of interest exist. Please note that mention of the external reference materials suppliers does not in any way constitute their endorsement, as neither does the listing of reagents, supplies, and instrumentation.
5 APPENDIX
5.1 APPENDIX A. SCREENING METHODOLOGIES FOR PDE5 INHIBITORS
5.1.1 HPLC with Photodiode Array Detection
Solution A: 0.1% Formic acid in water
Solution B: 0.1% Formic acid in acetonitrile
Mobile phase: See Table 1.
Table 1
| Time (min) | Solution A (%) | Solution B (%) |
| 0 | 95 | 5 |
| 15 | 5 | 95 |
| 23 | 5 | 95 |
| 24 | 95 | 5 |
| 31 | 95 | 5 |
Diluent: Acetonitrile and water (50:50)
Standard solution: 100 µg/mL each of USP Sildenafil Citrate RS, USP Tadalafil RS, and USP Vardenafil Hydrochloride RS in Diluent
Sample solution: Combine one-fifth of the dosage unit, 10-20 mg of bulk material, or a small fragment of the capsule shell (3 mm x 3 mm) with 10 mL of Diluent, sonicate for 30 min, and pass through a 0.2-µm PTFE syringe filter.
Chromatographic system
- (See Chromatography (621), System Suitability.)
- Mode: LC
- Detector: Photodiode array, 200-400 nm
- Analytical wavelength: 290 nm
- Column: 2.1-mm x 15-cm; 5-um packing L13
- Column temperature: 40°
- Flow rate: 0.2 mL/min
- Injection volume: 1 µL
System suitability
- Sample: Standard solution
- Suitability requirements
- Column efficiency: NLT 3000 theoretical plates.
- Tailing factor: NMT 1.5
Analysis
- Sample: Sample solution
- Examine the UV spectra of the prominent peaks for similarity to those in the Standard solution or other known PDE5 inhibitor compounds (Figure 1 and Table 5). Typical retention times of several PDE5 inhibitors are provided in Table 5. However, neither retention time match nor the absorbance spectrum similarity should be construed as sufficient confirmation of the chemical identity of an adulterant.
5.1.2 HPLC with Mass-Spectrometric Detection
Preferably, a mass-spectrometric detector is connected in sequence to the UV-Vis detector. The settings below apply to an ion-trap mass spectrometer. Other MS detectors are suitable; however, it is advisable to use spectrometers that possess MS/MS capability.
Solution A: 0.1% Formic acid in water
Solution B: 0.1% Formic acid in acetonitrile
Mobile phase: See Table 2.
Table 2
| Time (min) | Solution A (%) | Solution B (%) |
| 0 | 95 | 5 |
| 15 | 5 | 95 |
| 23 | 5 | 95 |
| 24 | 95 | 5 |
| 31 | 95 | 5 |
Diluent: Acetonitrile and water (50:50)
Standard solution: 5 µg/mL each of USP Sildenafil Citrate RS, USP Tadalafil RS, and USP Vardenafil Hydrochloride RS in Diluent
Sample solution: Combine one-fifth portion of the dosage unit, 10–20 mg of bulk material, or a small fragment of the capsule shell (3 mm × 3 mm) with 10 mL of Diluent, sonicate for 30 min, and pass through the 0.2-µm PTFE syringe filter. Dilute the filtrate 20-fold with Diluent before injection.
Chromatographic system
- (See Chromatography 〈621〉, System Suitability.)
- Mode: LC
- Detector: UV 290 nm
- Column: 2.1-mm × 15-cm; 5-µm packing L14
- Column temperature: 40°
- Flow rate: 0.2 mL/min
- Injection volume: 1 µL
Mass spectrometric system⁵
- (See Mass Spectrometry 〈736〉.)
- Ionization: ESI
- Polarity: Positive or negative
- Sheath gas: 35 mL/min
- Sweep gas: 5 mL/min
- Capillary temperature: 300°
- Source voltage: 5 kV
- Collision: 45 meV
- Scanning: m/z 90–1050 and dependent scan on the most intense ion
System suitability
- Sample: Standard solution
- Suitability requirements
- Resolution: NLT 2.0 between vardenafil and sildenafil peaks
- Tailing factor: NMT 1.5
Analysis
- Sample: Sample solution
- Compare mass-to-charge ratios of the molecular ions [M+H]⁺ or [M−H]⁻ and fragments to those of the Standard solution or other known analytes listed in Table 4. Typical retention times of several common PDE5 inhibitors are provided in Table 5.
5.1.3 High Performance Thin-Layer Chromatography (HPTLC) with Visual, UV, and/or MS Detection
Standard solution: A composite of 0.2-mg/mL each of USP Sildenafil Citrate RS, USP Tadalafil RS, and USP Vardenafil Hydrochloride RS in methanol, with sonication if necessary. Additional reference materials may be available commercially.
Sample solution: Comminute 1 dosage unit, including the capsule shell and tablet coating, or about 500 mg of raw material; combine with 10 mL of methanol, and subject to ultrasonication for 30 min. Centrifuge or filter the solution, and use the supernatant. [NOTE-Upon development, if the chromatographic bands appear too saturated and UV densitometric spectra are distorted, dilute the Sample solution 10-fold with methanol.]
Developing solvent system: tert-Butyl methyl ether, methanol, and 28.0% (w/w) ammonium hydroxide (20:2:1). [NOTE-Strength of ammonium hydroxide was found to be crucial for adequate method performance. It is therefore advisable to establish the titer of higher-concentration ammonia and to adjust the latter to exactly 28.0% immediately before the experiment.]
Chromatographic system
- (See HPTLC for Articles of Botanical Origin (203).)
- Adsorbent: Chromatographic silica gel with an average particle size of 5 µm
- Application volume: 3 µL, as 8-mm bands
- Relative humidity: Condition the plate to a relative humidity of 47% using a suitable device in the presence of a saturated solution of potassium isothiocyanate.
- Temperature: Ambient
- Saturation: 20 min, with paper
- Developing distance: 6 cm
- Derivatization reagent: None
- Drying: 5 min in a current of cold air
- Detection 1: Visual, under illumination with 254-and 365-nm UV light
- Detection 2: UV-Vis spectrometry (scanning densitometer), 190-550 nm
- Detection 3: Mass spectrometry, m/z 90-1050
Mass spectrometric system7
- (See Mass Spectrometry (736).)
- Ionization: ESI
- Polarity: Positive, negative, or rapid switching
- Desolvation gas (N₂): 300 L/h
- Cone gas (N₂): 80 L/h
- Temperatures
- ESI probe: 105°
- Desolvation: 150°
- Capillary voltage: 3.0 kV
- Cone voltage: 50 V
- Scanning: m/z 90-1050
System suitability
- Sample: Standard solution
- Suitability requirements: Under UV light at 254 nm, sildenafil, tadalafil, and vardenafil appear as dark bands against the fluorescent background. Under UV light at 365 nm, sildenafil, tadalafil, and vardenafil appear as blue fluorescent bands.
Analysis: Inspect the plate under short-wave (254 nm) and long-wave (365 nm) UV light. PDE5 inhibitors appear as dark bands against the fluorescent background at 254 nm and typically exhibit different shades of blue fluorescence under 365 nm. Note the similarities in RF values between the bands in the Standard solution and Sample solution, these may be informative, however they do not constitute sufficient proof of identity, Relative intensities of the bands permit approximation of the amounts. Using scanning densitometry, obtain UV spectra of the prominent bands in the Sample solution, and compare them to those of the PDE5 inhibitors in the Standard solution and those provided in Table 5 and Figure 1. Mass-spectrometric interface, if available, may facilitate more definitive assignment of the analyte bands: compare mass-to-charge ratios of the molecular ions [M+H] or [M-H] and fragments to those of the common adulterants listed in Table 4.
5.1.4 Ambient lonization Mass Spectrometry
Diluent: Acetonitrile and water (50:50), with 0.1% formic acid
Standard solution: A composite solution containing 20 µg/mL each of USP Sildenafil Citrate RS, USP Tadalafil RS, and USP Vardenafil
Hydrochloride RS in Diluent. Additional reference materials may be available commercially.
Sample solution: Grind the entire dosage form, including the capsule shell and tablet coating, to a fine powder. Weigh about 50 mg of the resulting powder, or about 50 mg of bulk material, and combine with 5 mL of Diluent. Cap tightly, subject to ultrasonication for 2 min, and vortex thoroughly. Centrifuge or filter the resulting solution, and dilute an aliquot of the supernatant or filtrate 100-fold with Diluent.
Mass spectrometric system
- (See Applications of Mass Spectrometry (1736), Mass Spectrometers, Ionization Procedures, Ambient lonization Procedures.)
- Ionization: Ambient with thermal desorption
- Mode: Thermal profile
- Polarity: Positive, negative, or rapid switching
- Gas temperature: 150°, 250, 350", and 450"
- Scanning: m/z 90-1050
System suitability: Deposit 3-µL aliquots of the Standard solution onto the disposable sample cards. Set the compatible mass spectrometer to a 30-s acquisition of 90-1050 Da. Using one sample card for each temperature setting, acquire mass spectra at each of the following desorption gas temperatures: 150°, 250°, 350°, and 450° in positive ionization mode. Switch polarity, and re-acquire spectra at the same four temperatures in the negative ionization mode. [NOTE-If the mass spectrometer permits rapid polarity switching, both positive and negative spectra may be acquired simultaneously using a single sample.] Confirm that the [M+H] or [M-H] ions of sildenafil, tadalafil, and vardenafil are observed as listed in Table 4.
Analysis: Deposit 3-µL aliquots of the Sample solution onto the disposable sample cards, and follow the procedure outlined above for the Standard solution. In the event that a single ion dominates the mass spectrum at every temperature setting, dilute the Sample solutions 10-fold with Diluent and re-analyze. Compare mass-to-charge ratios of the molecular ions [M+H] or [M-H] and fragments to those of the known analytes listed in Table 4.
5.1.5 NMR Spectroscopy-Low-Field and High-Field
(See Nuclear Magnetic Resonance Spectroscopy (761), Qualitative and Quantitative NMR Analysis.) [NOTE-Deuterated acetonitrile (CD3CN) should be NLT 99.8 atom % D, and should contain 0.05% tetramethylsilane (TMS) as a chemical shift reference. Use of solvents in sealed ampules is recommended. NMR tubes should be suitable for use at the selected magnetic field strength.]
Standard solutions: Dissolve 10 mg of USP Sildenafil Citrate RS, USP Tadalafil RS, or USP Vardenafil Hydrochloride RS in separate 1-mL aliquots of CD3CN, and transfer 700-µL aliquots of the resulting solutions into individual NMR tubes. Additional reference materials are available commercially.
Sample solution: Grind the entire dosage form, including the capsule shell and tablet coating, to a fine powder. Transfer 100-200 mg of the ground material, or an equivalent amount of bulk raw material powder, into a 5-mL sealable glass vial. Add 1 mL of CD3CN, vortex thoroughly, and allow the solids to settle. Transfer about 700 µL of the supernatant to an NMR tube, taking care to minimize transfer of solids.
Instrument performance qualification
- (See Nuclear Magnetic Resonance Spectroscopy (761).)
- Magnetic field strength: NLT 42.5 MHz (H operating frequency)
Data collection: Use the parameters specified in Table 3; perform 90° pulse width calibration before the analysis according to the recommendations of the equipment manufacturer.
Table 3
| Parameter | ¹H-NMR Qualitative Measurement |
| Pulse program | Single pulse ¹H |
| Spectral width | 14 ppm (−1 to 13 ppm) |
| Transmitter offset | Center of spectral width |
| Relaxation delay | 5–10 s |
| Acquisition time | 2–5 s |
| Number of data points per FIDᵃ | NLT 16,000 |
| Temperature | 25° |
ᵃ Free induction decay.
System suitability: Acquire a ¹H spectrum of the Standard solution using the settings outlined in Data collection. Record a sufficient number of scans to ensure that signal-to-noise ratio of the TMS signal is NLT 10.
Analysis: Acquire a ¹H spectrum of the Sample solution under the conditions outlined in Data collection. Record a sufficient number of scans to ensure that the signal-to-noise ratio of the TMS signal is NLT 10. Reference all acquired spectra to the ¹H signal of TMS (0 ppm). Measure and record the chemical shift and multiplicity of the NMR signals in the spectra of the Standard solutions and Sample solution. Compare the ¹H NMR spectrum of the Sample solution to those of the Standard solutions, paying particular attention to the aromatic region (5–9 ppm). Determine whether the chemical shift and multiplicity of the NMR signals in the Sample solution exhibit sufficient similarity to those found in the Standard solutions.
5.1.6 Bioassay⁸
PDE5 enzyme⁹ stock solution: Prepare a concentration of approximately 3000 Units/µL. If necessary, dilute with 40 mM tris(hydroxymethyl)aminomethane hydrochloride (tris-HCl), pH 8.0, 110 mM sodium chloride (NaCl), 2.2 mM potassium chloride (KCl), 3 mM dithiothreitol, and 20% Glycerin. Vortex gently to mix.
PDE5 working solution (100 Units per 6.5 µL): Combine 400 µL of PDE-Glo™ Reaction Buffer 5X, 10 µL of PDE5 enzyme stock solution, and 1590 µL of water. Vortex gently to mix.
cGMP solution: Combine 400 µL of PDE-Glo™ Reaction Buffer 5X, 40 µL of 1-mM cGMP stock solution, and 1560 µL of water. Mix thoroughly by vortexing.
100-mM IBMX stock solution in DMSO: Prepare a 22.2-mg/mL solution of 3-isobutyl-1-methylxanthine (IBMX) in dimethylsulfoxide (DMSO), e.g., dissolve 100 mg of IBMX in 4.5 mL of DMSO. Mix thoroughly by vortexing.
Reaction buffer: Combine 400 µL of PDE-Glo™ Reaction Buffer 5X and 1600 µL of water. Mix thoroughly by vortexing.
Reaction buffer with 4% DMSO: Combine 400 µL of PDE-Glo™ Reaction Buffer 5X, 80 µL of DMSO and 1520 µL of water. Mix thoroughly by vortexing.
Termination buffer: Combine 400 µL of PDE-Glo™ Termination Buffer 5X, 40 µL of 100-mM IBMX stock solution in DMSO, and 1560 µL of water. Mix thoroughly by vortexing.
Detection buffer: Combine 400 µL of PDE-Glo™ Detection Buffer 5X, 16 µL of Protein Kinase A Solution, and 1584 µL of water. Vortex gently to mix.
Kinase-Glo™ reagent: Add 10 mL of Kinase-Glo™ Buffer to the vial of Kinase-Glo™ Substrate, and vortex gently.
Standard solution (400 nM): Dissolve 5 mg of USP Sildenafil Citrate RS in 3.0 mL of DMSO to obtain a 2.5-mM stock solution. Combine a 10-µL aliquot of the resulting solution with 240 µL of DMSO, and mix thoroughly (100 µM). Combine a 10-µL aliquot of the resulting solution with 90 µL of DMSO, and mix thoroughly (10 µM). Combine a 10-µL aliquot of the resulting solution with 240 µL of Reaction buffer, and mix thoroughly (400 nM); the last dilution is the Standard solution.
Control solution: Combine 10 µL of DMSO with 240 µL of Reaction buffer, and mix thoroughly by vortexing.
Sample solution: Grind the entire dosage form, including the capsule shell and tablet coating, to a fine powder. Transfer 100 mg of the ground material into a 5-mL polypropylene vial. Add 3.0 mL of DMSO, and vortex for 60 s. Allow solids to settle, combine 50 µL of the clear supernatant with 200 µL of DMSO, and mix thoroughly by vortexing. Combine a 10-µL aliquot of the resulting solution with 90 µL of DMSO, and mix thoroughly by vortexing. Combine a 10-µL aliquot of the resulting solution with 240 µL of Reaction buffer, and mix thoroughly by vortexing; the last dilution is the Sample solution.
Analysis
- Dispense 12.5-µL aliquots of the Standard solution, Control solution, and Sample solution into microplate wells, in triplicate. Use a white, flat-bottom, opaque polystyrene, nontreated, 96-well microtiter plate.¹⁰ [Note-Do not use treated plates, black plates, or clear plates.]
- Add 6.5 µL of PDE5 working solution to each well. Incubate for 5 min.
- Add 6.0 µL of cGMP solution to effect a 5-µM cGMP concentration in a 25-µL volume. Incubate for 30 min.
- Add 12.5 µL of Termination buffer. Incubate for 5 min.
- Add 12.5 µL of Detection buffer. Incubate for 20 min.
- Add 50 µL of Kinase-Glo™ reagent. Incubate for 10 min.
- Record luminescence at 560 nm with a microplate luminometer at 0.5 s/well.
[Note-Incubate the plate at room temperature, preferably using a plate shaker.]
Calculate average luminescence values for the replicate preparations. Assess the degree of PDE5 inhibition in the Sample solution relative to that observed in the Standard solution and Control solution. Inhibition of PDE5 is manifested as reduction of luminescence: the samples that exhibit suppression of luminescent output comparable to or in excess of that observed with the Standard solution are likely adulterated with synthetic PDE5 inhibitors.
5.2 USP Reference Standards 〈11〉
USP Sildenafil Citrate RS
USP Tadalafil RS
USP Vardenafil Hydrochloride RS
Table 4. Mass Spectral Data for Select PDE5 Inhibitorsa
| # | Name | CAS Number | Chemical Formula | Exact Mass | [M+H]⁺ | [M−H]⁻ | Fragments |
| 1 | Acetaminotadalafil | 1446144-71-3 | C₂₃H₂₀N₄O₅ | 432.1434 | 433 | - | 455 [M+Na]⁺, 391, 311, 269, 250 |
| 2 | Acetil acid | 147676-78-6 | C₁₈H₂₀N₄O₄ | 356.1485 | 357 | - | 329, 300, 285, 268, 256, 242, 166, 131 |
| 3 | Acetildenafil (Hongdenafil) | 831217-01-7 | C₂₅H₃₄N₆O₃ | 466.26924 | 467.28 | - | 449, 439, 420, 404, 396, 381, 355, 353, 339, 325, 324, 311, 297, 285, 166, 127, 111, 99, 97 |
| 4 | Acetylvardenafil | 1261351-28-3 | C₂₅H₃₄N₆O₃ | 466.2692 | 467 | - | 439, 396, 341, 317, 270 |
| 5 | Aildenafil (Dimethylsildenafil, Methisosildenafil) | 496835-35-9 | C₂₃H₃₂N₆O₄S | 488.22057 | 489.23 | - | 432, 377, 313, 311, 283, 113, 99 |
| - | 487.40 | 460, 310, 282 | |||||
| 6 | Aminotadalafil | 385769-84-6 | C₂₁H₁₈N₄O₄ | 390.1328 | 391.14 | - | 269, 262, 241, 239, 224, 197, 169 |
| - | 389.1248 | 362, 298, 262, 234, 233, 232 | |||||
| 7 | (S,R)-Aminotadalafil [(+)-trans-Aminotadalafil] | 1093940-70-5 | C₂₁H₁₈N₄O₄ | 390.1328 | 391 | - | No data |
| 8 | Avanafil | 330784-47-9 | C₂₃H₂₆N₇O₃Cl | 483.1786 | 484.186 | - | 375, 349, 221 |
| 9 | Benzylsildenafil | 1446089-82-2 | C₂₈H₃₄N₆O₄S | 550.2362 | 551 | - | 377, 283 |
| 10 | N-Butylnortadalafil | 171596-31-9 | C₂₅H₂₅N₃O₄ | 431.18451 | 432.25 | - | 310ᵇ, 282ᶜ, 197, 169 |
| 11 | Carbodenafil (Fondenafil) | 944241-52-5 | C₂₄H₃₂N₆O₃ | 452.2536 | 453 | - | 283 |
| 12 | Chlorodenafil | 1058653-74-9 | C₁₉H₂₁ClN₄O₃ | 388.8485 | 390 | - | 360, 311, 291, 254, 183, 136 |
| 13 | Chloropretadalafil | 171489-59-1 | C₂₂H₁₉ClN₂O₅ | 426.0982 | 427 | - | 429, 395, 349, 334, 302, 287, 262, 229, 159, 135 |
| 14 | Cinnamyldenafil | 1446089-83-3 | C₃₂H₃₈N₆O₃ | 554.3005 | 554 | - | 488, 354, 297, 283, 215, 166, 117, 91 |
| 15 | Cyclopentynafil | 1173706-34-7 | C₂₆H₃₆N₆O₄S | 528.2519 | 529 | - | 461, 153 |
| 16 | Depiperazinothiosildenafil | 1353018-10-6 | C₁₇H₂₀N₄O₄S₂ | 408.0926 | 409 | - | 381, 351, 327, 299, 285, 272 |
| - | 407.0845 | 378, 314 | |||||
| 17 | Descarbonsildenafil | 1393816-99-3 | C₂₁H₃₀N₆O₄S | 462.2049 | 463 | - | 418, 377, 360, 311, 299, 283, 255, 151, 87 |
| 18 | N-Desethylvardenafil | 448184-46-1 | C₂₁H₂₈N₆O₄S | 460.18927 | 461.20 | - | 392, 377, 376, 329, 313, 312, 299, 284, 283, 151 |
| 19 | Desmethylcarbodenafil | 147676-79-7 | C₂₃H₃₁N₆O₃ | 438.523 | 439.2451 | - | 339, 311 |
| 20 | N-Desmethylsildenafil | 139755-82-1 | C₂₁H₂₈N₆O₄S | 460.18927 | 461.19 | - | 377, 313, 311, 299, 283 |
| 21 | Dimethylacetildenafil | 1417999-76-8 | C₂₅H₃₄N₆O₃ | 466.2771 | 467 | - | 279, 149, 177 |
| 22 | Dioxo-acetildenafil | 1609405-33-5 | C₂₅H₃₀N₆O₅ | 494.2278 | 495 | - | No data |
| 23 | Dithio-desmethylcarbodenafil | 1333233-46-7 | C₂₃H₃₀N₆O₅S₂ | 470.6572 | 471.1991 | - | 371, 343 |
| 24 | Gendenafil | 147676-66-2 | C₁₉H₂₂N₄O₃ | 354.16919 | 355.31 | - | 327, 298, 285, 283, 256, 242 |
| 25 | Gisadenafil | 334826-98-1 | C₂₃H₃₃N₇O₅S | 519.22639 | 520 | - | No data |
| 26 | Homosildenafil | 642928-07-2 | C₂₃H₃₂N₆O₄S | 488.22057 | 489.23 | - | 467, 461, 377, 313, 311, 283, 127, 111, 113, 99, 97 |
| 27 | Hydroxyacetildenafil (Hydroxyhongdenafil) | 147676-56-0 | C₂₅H₃₄N₆O₄ | 482.26415 | 483.27 | - | 465, 447, 439, 396, 353, 339, 325, 311, 297 |
| 28 | Hydroxychlorodenafil | 1391054-00-4 | C₁₉H₂₃ClN₄O₃ | 390.1459 | 391 | - | No data |
| 29 | Hydroxyhomosildenafil | 139755-85-4 | C₂₃H₃₂N₆O₅S | 504.21549 | 505.22 | - | 487, 461, 423, 377, 312, 311, 283, 284, 225, 166, 129, 112, 99, 97 |
| - | 503.31 | 475, 310, 282 | |||||
| 30 | 2-Hydroxypropylnortadalafil | 1353020-85-5 | C₂₄H₂₃N₃O₅ | 433.16377 | 434.15 | - | 312, 284, 197, 169 |
| 31 | Hydroxythiohomosildenafil (Hydroxyhomosildenafil thione, Sulfohydroxyhomosildenafil) | 479073-82-0 | C₂₃H₃₂N₆O₄S₂ | 520.19264 | 521.20 | - | 503, 477, 461, 419, 393, 355, 354, 327, 325, 291 |
| 32 | Hydroxythiovardenafil | 912576-30-8 | C₂₃H₃₂N₆O₄S₂ | 520.1926 | 521 | - | No data |
| 33 | Hydroxyvardenafil | 224785-98-2 | C₂₃H₃₂N₆O₅S | 504.2155 | 505.26 | - | 344, 312, 253, 169, 99 |
| - | 503 | 475, 310, 282 | |||||
| 34 | Imidazosagatriazinone (Desulfovardenafil) | 139756-21-1 | C₁₇H₂₀N₄O₂ | 312.1586 | 313 | - | 284, 256, 169, 151 |
| 35 | Isopiperazinonafil | 1335201-06-3 | C₂₅H₃₄N₆O₄ | 482.2642 | 483 | - | - |
| - | 481 | 453, 422, 379, 336, 325, 311, 309 | |||||
| 36 | Lodenafil carbonate | 398507-55-6 | C₄₇H₆₂N₁₂O₁₁S₂ | 1034.4102 | 1035 | - | 518, 487, 377, 311 |
| 37 | Mirodenafil | 862189-95-5 | C₂₆H₃₇N₅O₅S | 531.6698 | 532 | - | 488, 404, 362, 296, 268 |
| 38 | Mutaprodenafil | 1387577-30-1 | C₂₇H₃₅N₉O₅S₂ | 629.7635 | 630.2279 | - | 489, 377, 142, 113 |
| 39 | Nitrodenafil | 147676-99-1 | C₁₇H₁₉N₅O₄ | 357.3647 | 358 | - | 307, 289, 261, 217, 176, 154, 136, 107, 89 |
| 40 | Nitroso-prodenafil | 1266755-08-1 | C₂₇H₃₅N₉O₅S₂ | 629.2203 | 630 | - | 142 |
| 41 | Noracetildenafil (Demethylhongdenafil) | 949091-38-7 | C₂₄H₃₂N₆O₃ | 452.25359 | 453.26 | - | 425, 406, 396, 380, 367, 355, 353, 339, 325, 324, 313, 297, 296, 253 |
| 42 | Norneosildenafil (Piperidinosildenafil) | 371959-09-0 | C₂₂H₂₉N₅O₄S | 459.19403 | 460.20 | - | 432, 377, 329, 311, 299, 283 |
| 43 | Norneovardenafil | 358390-39-3 | C₁₈H₂₀N₄O₄ | 356.1485 | 357 | - | 329, 307, 289, 176, 154, 136, 107, 99 |
| 44 | Nortadalafil (Demethyltadalafil) | 171596-36-4 | C₂₁H₁₇N₃O₄ | 375.1219 | 376 | 374.1138 | 262, 234, 233, 232 |
| 45 | N-Octylnortadalafil | 1173706-35-8 | C₂₉H₃₃N₃O₄ | 487.2471 | 488 | - | 366, 227 |
| 46 | Oxohongdenafil | 1446144-70-2 | C₂₅H₃₂N₆O₄ | 480.2485 | 481 | - | 451, 396, 354, 339, 312, 297, 289 |
| 47 | Piperazinonafil (Piperazonifil, Dihydroacetildenafil) | 1335201-04-1 | C₂₅H₃₄N₆O₄ | 482.2642 | 483 | - | - |
| - | 481 | 453, 435, 348, 336, 321, 311, 309, 282, 267 | |||||
| 48 | Piperidinoacetildenafil (Piperiacetildenafil) | 147676-50-4 | C₂₄H₃₁N₅O₃ | 437.2427 | 438 | - | 410, 408, 355, 353, 341, 325, 297, 288 |
| 49 | Piperidinovardenafil (Piperidenafil, Pseudovardenafil) | 224788-34-5 | C₂₂H₂₉N₅O₄S | 459.19403 | 460.20 | - | 432, 403, 391, 377, 349, 329, 312, 311, 301, 299, 284, 283, 270, 256, 169, 151 |
| 50 | Propoxyphenyl aildenafil | 1391053-82-9 | C₂₄H₃₄N₆O₄S | 502.2362 | 503 | - | 252 |
| 51 | Propoxyphenyl hydroxyhomosildenafil (Methylhydroxyhomosildenafil) | 139755-87-6 | C₂₄H₃₄N₆O₅S | 518.2311 | 519 | - | 501, 475, 391, 331, 325, 299, 283, 129, 112, 99 |
| 52 | Propoxyphenyl sildenafil | 877777-10-1 | C₂₃H₃₂N₆O₄S | 488.2205 | 489.2272 | - | 447, 391, 325, 299, 283, 100 |
| 53 | Propoxyphenyl thioaildenafil (Propoxyphenyl thiomethisosildenafil) | 856190-49-3 | C₂₄H₃₄N₆O₃S₂ | 518.2133 | 519 | - | 260 |
| 54 | Propoxyphenyl thiohydroxyhomosildenafil | 479073-90-0 | C₂₄H₃₄N₆O₄S₂ | 534.2083 | 535.2150 | - | 517, 359, 341, 315, 299, 271, 129, 112, 99 |
| 55 | Sildenafil | 139755-83-2 | C₂₂H₃₀N₆O₄S | 474.20492 | 475.21 | - | 447, 418, 391, 377, 374, 346, 329, 311, 297, 283, 255, 163, 160, 100 |
| - | 473.45 | 445, 310, 282 | |||||
| 56 | Tadalafil (Tildenafil) | 171596-29-5 | C₂₂H₁₉N₃O₄ | 389.13756 | 390 | - | 302, 268, 262, 250, 240, 197, 169, 135 |
| - | 388.1288 | 262, 234, 233, 232 | |||||
| 57 | (−)-trans-Tadalafil (ent-Tadalafil) | 629652-72-8 | C₂₂H₁₉N₃O₄ | 389.1376 | 390 | - | 779 [2M+H]⁺, 262, 250, 135 |
| 58 | Thioaildenafil (Sulfoaildenafil, Thiomethisosildenafil) | 856190-47-1 | C₂₃H₃₂N₆O₃S₂ | 504.19773 | 505.21 | - | 448, 393, 327, 299, 113, 99 |
| 59 | Thiohomosildenafil | 479073-80-8 | C₂₃H₃₂N₆O₃S₂ | 504.19773 | 505.21 | - | 477, 421, 393, 357, 355, 343, 327, 315, 299, 271, 113, 99 |
| 60 | Thioquinapiperifil (KF31327) | 220060-39-9 | C₂₄H₂₈N₆OS | 448.2045 | 449 | - | 363, 246, 225, 204, 121 |
| 61 | Thiosildenafil (Sulfosildenafil, Sildenafil thione) | 479073-79-5 | C₂₂H₃₀N₆O₃S₂ | 490.18208 | 491.19 | - | 407, 393, 343, 341, 327, 315, 313, 299, 283, 271, 163, 99 |
| 62 | Udenafil | 268203-93-6 | C₂₅H₃₆N₆O₄S | 516.2519 | 517.260 | - | 474, 418, 347, 325, 299, 283 |
| 63 | Vardenafil | 224785-90-4 | C₂₃H₃₂N₆O₄S | 488.22057 | 489.2274 | - | 461, 420, 377, 376, 375, 346, 339, 329, 312, 299, 284, 283 |
| - | 487.33 | 459, 310, 282 | |||||
| 64 | Xanthoanthrafil (Benzamidenafil) | 1020251-53-9 | C₁₉H₂₃N₃O₆ | 389.15869 | 390.31 | - | 344, 252, 223, 151, 107, 91 |
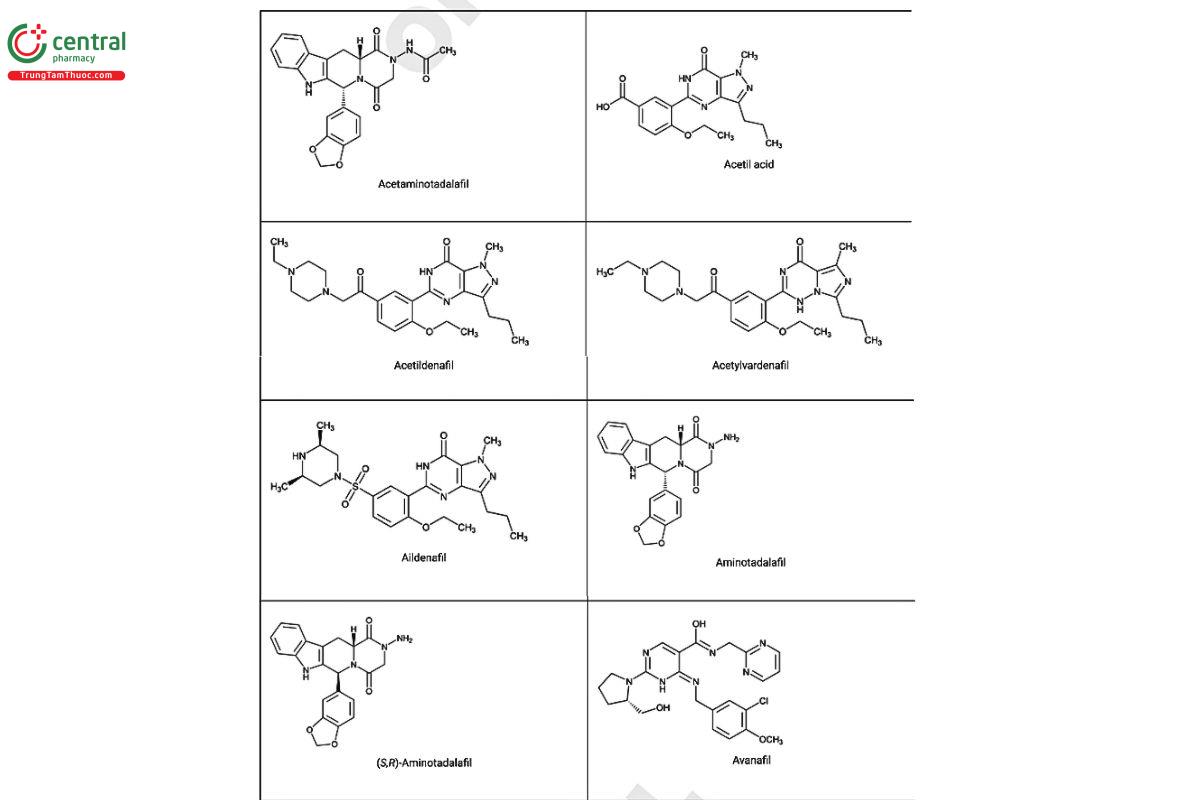
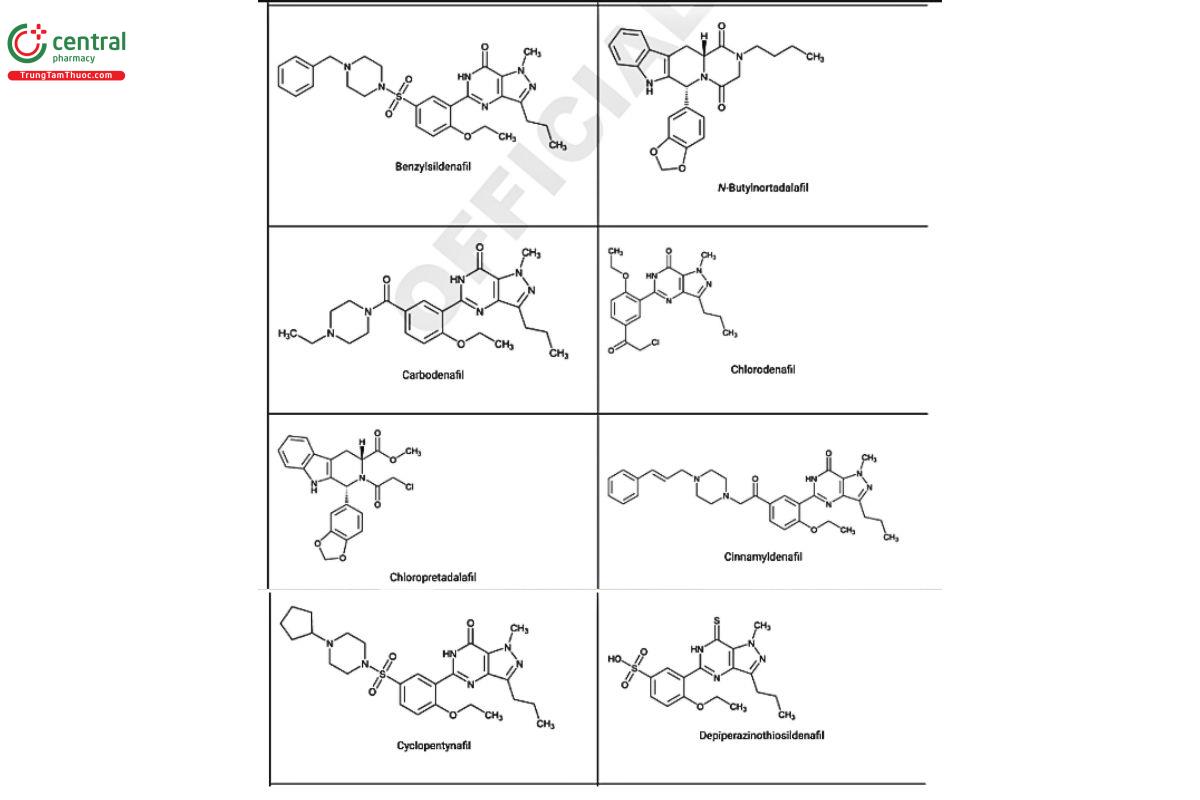
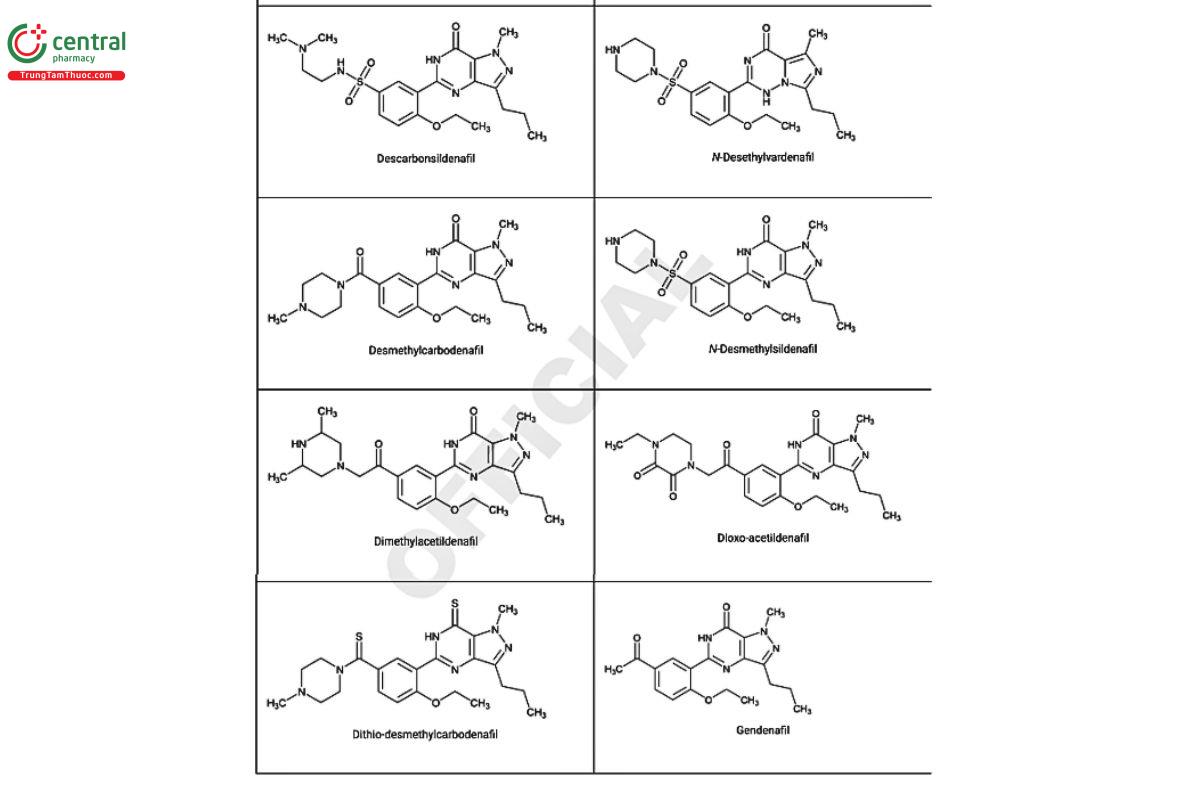
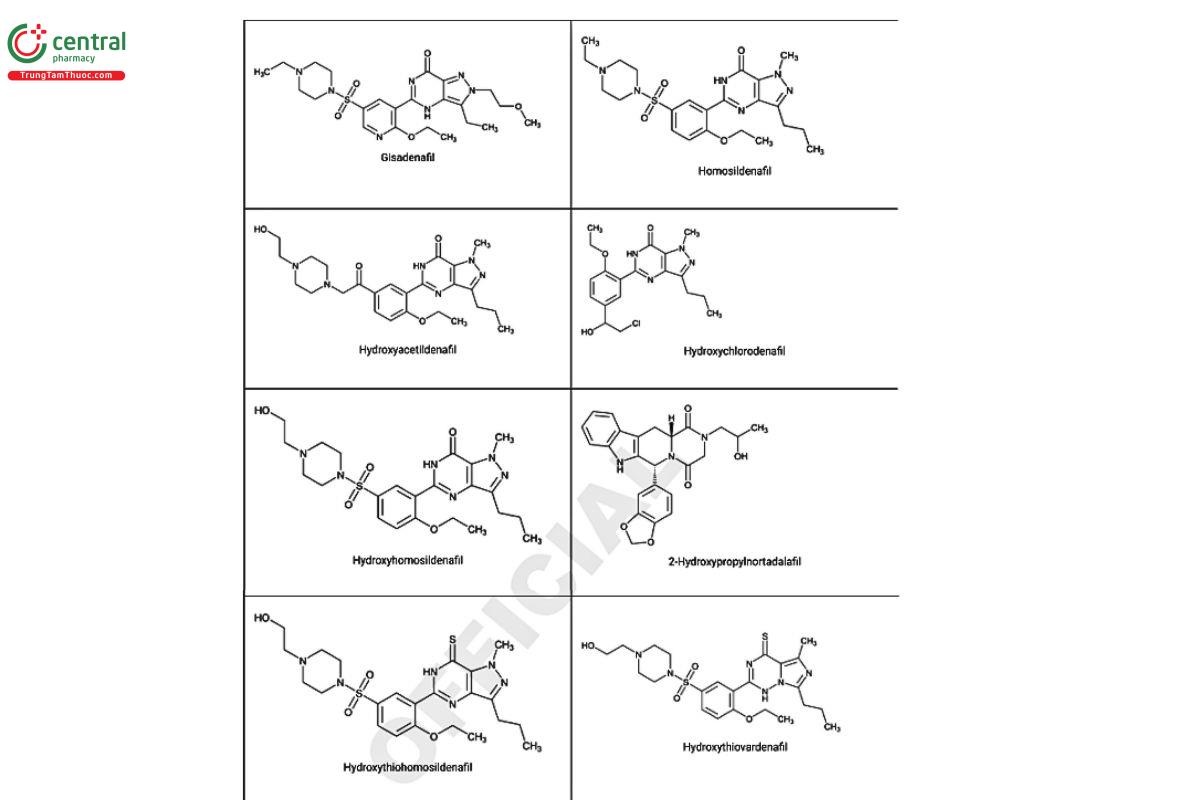
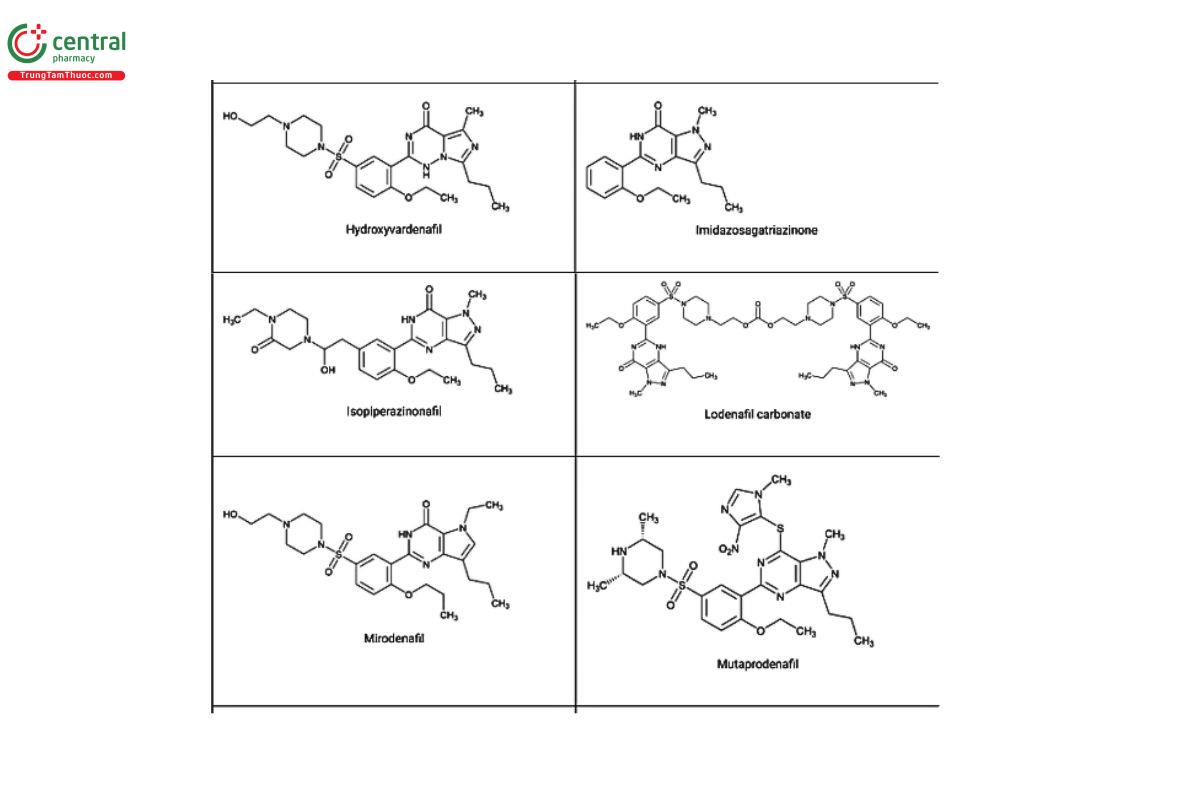
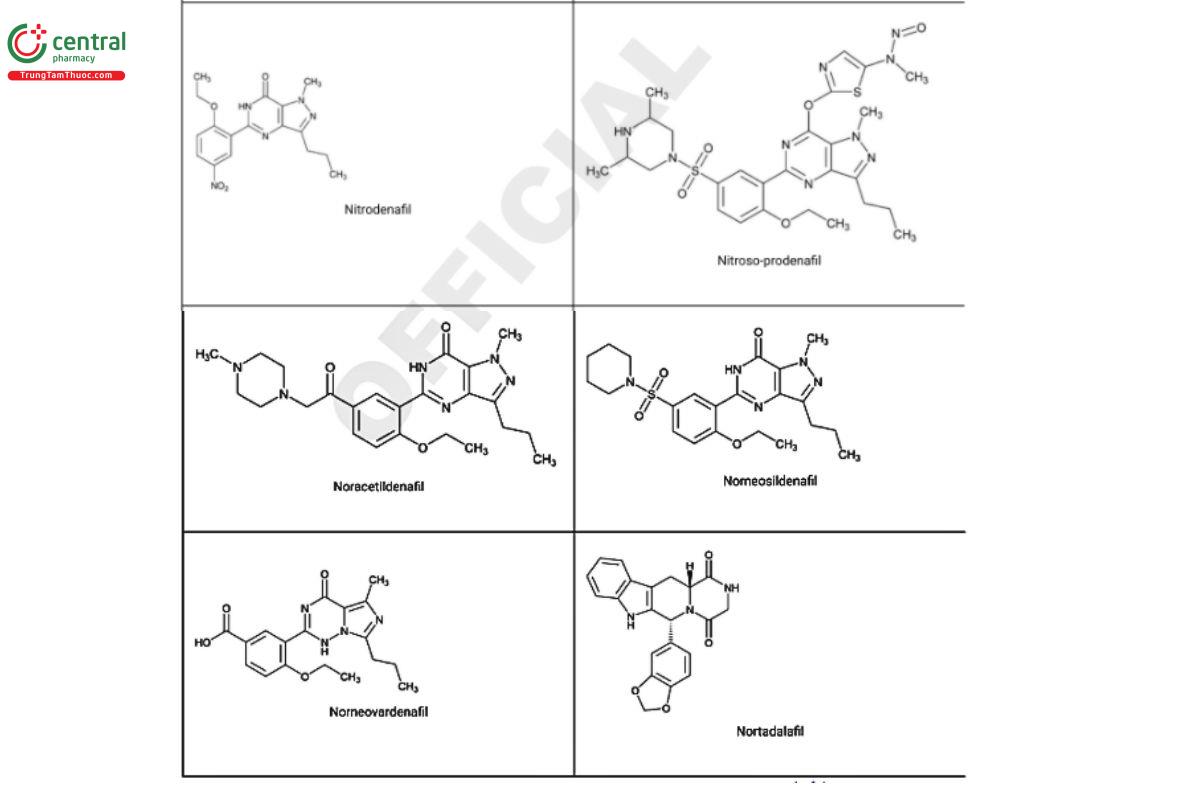
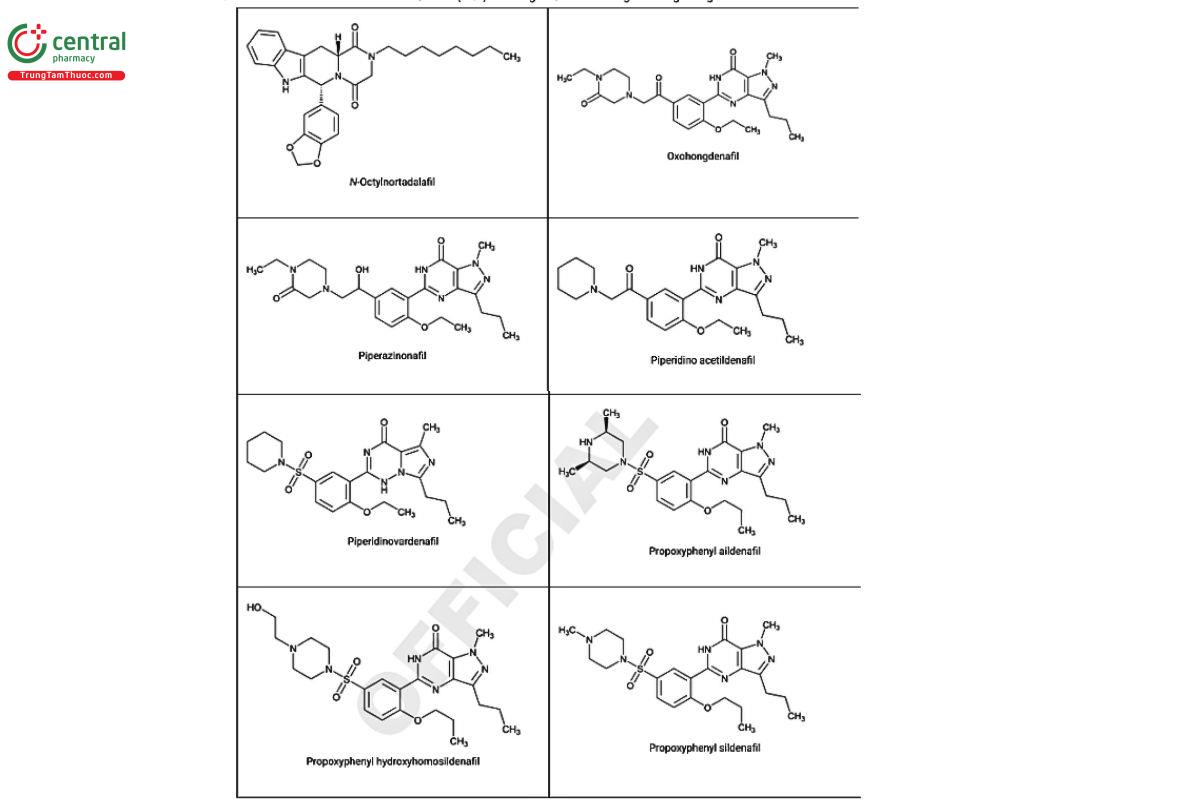
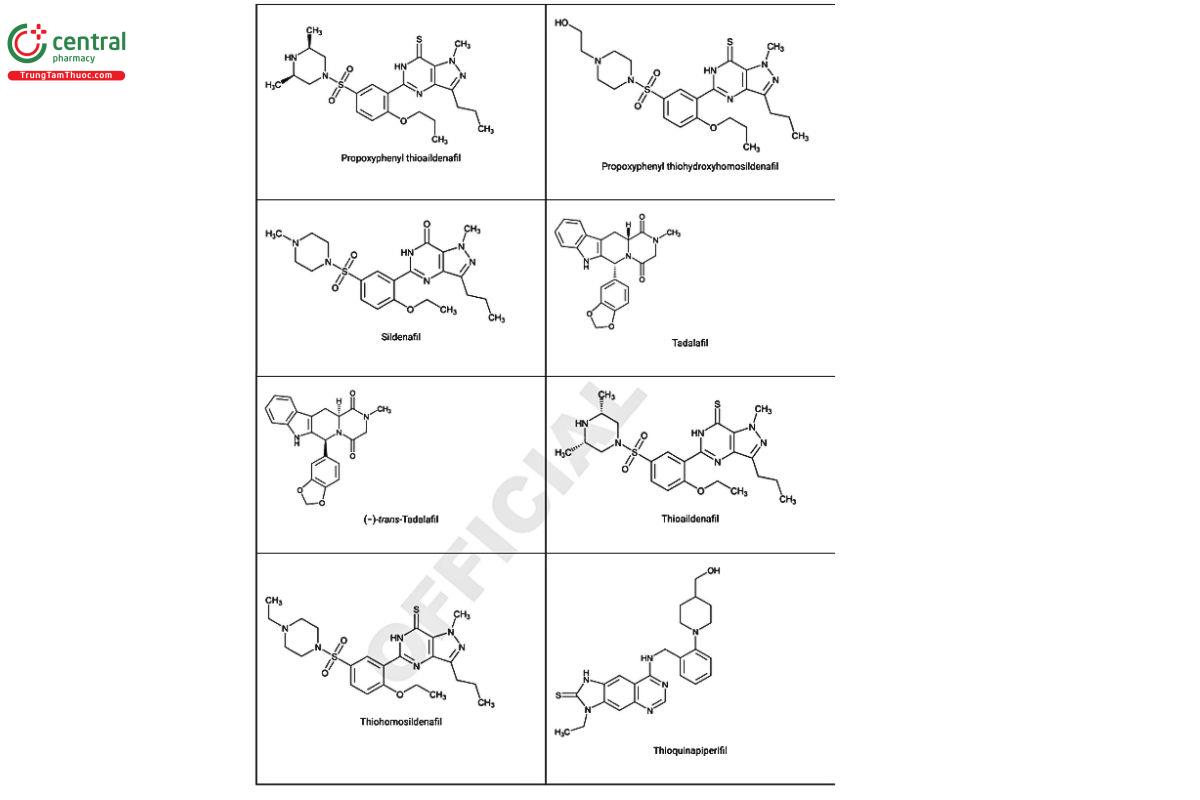
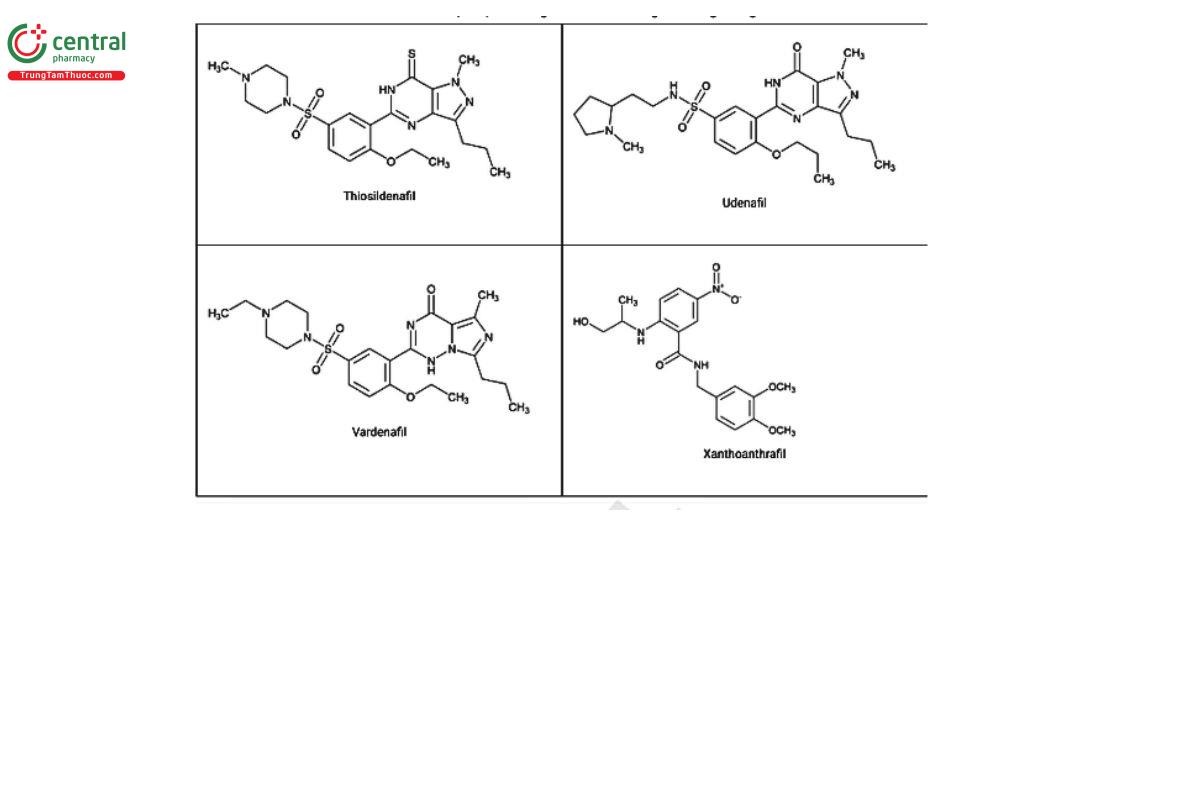
a Compiled from peer-reviewed literature, and communications with USP collaborators. See corresponding chemical structures in Figure 2.
b Bold, fragment subjected to MS3 fragmentation.
c Italics, MS3 fragments derived from the parent fragment (bold).
Table 5. UV Absorbance Maxima and Retention Time Data for Select PDE5 Inhibitora
| # | Name | CAS Number | Chemical Formula | UV Absorbance Maxima (nm) | UV Absorbance Spectrum Type (Figure 1) | Retention Time (min)ᵇ | Relative Retention Time with Respect to Sildenafil |
| 1 | Acetaminotadalafil | 1446144-71-3 | C₂₃H₂₀N₄O₅ | 202, 222, 282 | b | 14.3 | 1.1 |
| 2 | Acetil acid | 147676-78-6 | C₁₈H₂₀N₄O₄ | 230, 260, 285 | - | - | - |
| 3 | Acetildenafil (Hongdenafil) | 831217-01-7 | C₂₅H₃₄N₆O₃ | 234, 282 | e | 12.5 | 1.0 |
| 4 | Acetylvardenafil | 1261351-28-3 | C₂₅H₃₄N₆O₃ | 218, 246, 268(s)ᶜ | d | 11.4 | 0.9 |
| 5 | Aildenafil (Dimethylsildenafil, Methisosildenafil) | 496835-35-9 | C₂₃H₃₂N₆O₄S | 226, 294 | a | 13.3 | 1.0 |
| 6 | Aminotadalafil | 385769-84-6 | C₂₁H₁₈N₄O₄ | 200, 220, 284, 290(s) | b | 14.2 | 1.1 |
| 7 | (S,R)-Aminotadalafil [(+)-trans-Aminotadalafil] | 1093940-70-5 | C₂₁H₁₈N₄O₄ | 225, 283 | - | - | - |
| 8 | Avanafil | 330784-47-9 | C₂₃H₂₆N₇O₃Cl | 198, 244 | f | 13.0 | 1.0 |
| 9 | Benzylsildenafil | 1446089-82-2 | C₂₈H₃₄N₆O₄S | 291 | - | - | - |
| 10 | N-Butylnortadalafil | 171596-31-9 | C₂₅H₂₅N₃O₄ | 222, 284 | - | - | - |
| 11 | Carbodenafil (Fondenafil) | 944241-52-5 | C₂₄H₃₂N₆O₃ | 295 | - | - | - |
| 12 | Chlorodenafil | 1058653-74-9 | C₁₉H₂₁ClN₄O₃ | 211, 235, 279 | - | - | - |
| 13 | Chloropretadalafil | 171489-59-1 | C₂₂H₁₉ClN₂O₅ | 204, 222, 284 | b | 17.4 | 1.3 |
| 14 | Cinnamyldenafil | 1446089-83-3 | C₃₂H₃₈N₆O₃ | 239 | - | - | - |
| 15 | Cyclopentynafil | 1173706-34-7 | C₂₆H₃₆N₆O₄S | 218, 290 | - | - | - |
| 16 | Depiperazinothiosildenafil | 1353018-10-6 | C₁₇H₂₀N₄O₄S₂ | 295, 354 | - | - | - |
| 17 | Descarbonsildenafil | 1393816-99-3 | C₂₁H₃₀N₆O₄S | 225, 295 | - | - | - |
| 18 | N-Desethylvardenafil | 448184-46-1 | C₂₁H₂₈N₆O₄S | 226, 246(s) | d | 11.9 | 0.9 |
| 19 | Desmethylcarbodenafil | 147676-79-7 | C₂₃H₃₁N₆O₃ | 226, 296 | a | 11.9 | 0.9 |
| 20 | N-Desmethylsildenafil | 139755-82-1 | C₂₁H₂₈N₆O₄S | 224, 294 | a | 12.9 | 1.0 |
| 21 | Dimethylacetildenafil | 1417999-76-8 | C₂₅H₃₄O₃N₆ | 233, 276 | - | - | - |
| 22 | Dioxo-acetildenafil | 1609405-33-5 | C₂₅H₃₀N₆O₅ | No data | - | - | - |
| 23 | Dithio-desmethylcarbodenafil | 1333233-46-7 | C₂₃H₃₀N₆O₅S₂ | 258, 285, 356 | - | - | - |
| 24 | Gendenafil | 147676-66-2 | C₁₉H₂₂N₄O₃ | 232, 274 | f | 16.8 | 1.3 |
| 25 | Gisadenafil | 334826-98-1 | C₂₃H₃₃N₇O₅S | No data | - | - | - |
| 26 | Homosildenafil | 642928-07-2 | C₂₃H₃₂N₆O₄S | 226, 292 | a | 13.3 | 1.0 |
| 27 | Hydroxyacetildenafil (Hydroxyhongdenafil) | 147676-56-0 | C₂₅H₃₄N₆O₄ | 234, 280 | e | 12.2 | 0.9 |
| 28 | Hydroxychlorodenafil | 1391054-00-4 | C₁₉H₂₃ClN₄O₃ | 212, 303 | - | - | - |
| 29 | Hydroxyhomosildenafil | 139755-85-4 | C₂₃H₃₂N₆O₅S | 226, 296 | a | 12.9 | 1.0 |
| 30 | 2-Hydroxypropylnortadalafil | 1353020-85-5 | C₂₄H₂₃N₃O₅ | 222, 284 | - | - | - |
| 31 | Hydroxythiohomosildenafil | 479073-82-0 | C₂₃H₃₂N₆O₄S₂ | 228, 296, 352 | c | 15.0 | 1.2 |
| 32 | Hydroxythiovardenafil | 912576-30-8 | C₂₃H₃₂N₆O₄S₂ | 203, 235, 316 | - | - | - |
| 33 | Hydroxyvardenafil | 224785-98-2 | C₂₃H₃₂N₆O₅S | 216 | - | - | - |
| 34 | Imidazosagatriazinone (Desulfovardenafil) | 139756-21-1 | C₁₇H₂₀N₄O₂ | 212, 253 | - | - | - |
| 35 | Isopiperazinonafil | 1335201-06-3 | C₂₅H₃₄N₆O₄ | 221, 290 | - | - | - |
| 36 | Lodenafil carbonate | 398507-55-6 | C₄₇H₆₂N₁₂O₁₁S₂ | 226, 296 | a | 15.4 | 1.2 |
| 37 | Mirodenafil | 862189-95-5 | C₂₆H₃₇N₅O₅S | 216, 248 | f | 14.5 | 1.1 |
| 38 | Mutaprodenafil | 1387577-30-1 | C₂₇H₃₅N₉O₅S₂ | 218, 240, 283, 297, 335 | - | - | - |
| 39 | Nitrodenafil | 147676-99-1 | C₁₇H₁₉N₅O₄ | 212, 298 | - | - | - |
| 40 | Nitroso-prodenafil | 1266755-08-1 | C₂₇H₃₅N₉O₅S₂ | 241, 301 | - | - | - |
| 41 | Noracetildenafil (Demethylhongdenafil) | 949091-38-7 | C₂₄H₃₂N₆O₃ | 234, 280 | e | 12.3 | 0.9 |
| 42 | Norneosildenafil (Piperidinosildenafil) | 371959-09-0 | C₂₂H₂₉N₅O₄S | 226, 300 | a | 18.7 | 1.4 |
| 43 | Norneovardenafil | 358390-39-3 | C₁₈H₂₀N₄O₄ | 215, 241 | - | - | - |
| 44 | Nortadalafil (Demethyltadalafil) | 171596-36-4 | C₂₁H₁₇N₃O₄ | No data | - | - | - |
| 45 | N-Octylnortadalafil (Octylnortadalafil) | 1173706-35-8 | C₂₉H₃₃N₃O₄ | 281 | - | - | - |
| 46 | Oxohongdenafil | 1446144-70-2 | C₂₅H₃₂N₆O₄ | 481 | - | - | - |
| 47 | Piperazinonafil (Piperazonifil, Dihydroacetildenafil) | 1335201-04-1 | C₂₅H₃₄N₆O₄ | 221, 290 | - | - | - |
| 48 | Piperidinoacetildenafil (Piperiacetildenafil) | 147676-50-4 | C₂₄H₃₁N₅O₃ | 234, 284 | e | 13.1 | 1.0 |
| 49 | Piperidinovardenafil (Piperidenafil, Pseudovardenafil) | 224788-34-5 | C₂₂H₂₉N₅O₄S | 224, 246(s) | d | 16.5 | 1.3 |
| 50 | Propoxyphenyl aildenafil | 1391053-82-9 | C₂₄H₃₄N₆O₄S | 215, 225, 295 | - | - | - |
| 51 | Propoxyphenyl hydroxyhomosildenafil (Methylhydroxyhomosildenafil) | 139755-87-6 | C₂₄H₃₄N₆O₅S | 226, 294 | a | 13.4 | 1.0 |
| 52 | Propoxyphenyl sildenafil | 877777-10-1 | C₂₃H₃₂N₆O₄S | 226, 292 | a | 13.6 | 1.0 |
| 53 | Propoxyphenyl thioaildenafil (Propoxyphenyl thiomethisosildenafil) | 856190-49-3 | C₂₄H₃₄N₆O₃S₂ | 227, 295, 355 | - | - | - |
| 54 | Propoxyphenyl thiohydroxyhomosildenafil | 479073-90-0 | C₂₄H₃₄N₆O₄S₂ | 227, 295, 353 | - | - | - |
| 55 | Sildenafil | 139755-83-2 | C₂₂H₃₀N₆O₄S | 224, 294 | a | 13.0 | 1.0 |
| 56 | Tadalafil (Tildenafil) | 171596-29-5 | C₂₂H₁₉N₃O₄ | 200, 222, 284, 292(s) | b | 15.0 | 1.2 |
| 57 | (−)-trans-Tadalafil (ent-Tadalafil) | 629652-72-8 | C₂₂H₁₉N₃O₄ | 231, 282, 289 | - | - | - |
| 58 | Thioaildenafil (Sulfoaildenafil, Thiomethisosildenafil, Sulfodimethylsildenafil, Dimethylthiosildenafil) | 856190-47-1 | C₂₃H₃₂N₆O₃S₂ | 228, 250(s), 296, 352, 366(s) | c | 15.5 | 1.2 |
| 59 | Thiohomosildenafil (Sulfohomosildenafil, Homosildenafil thione) | 479073-80-8 | C₂₃H₃₂N₆O₃S₂ | 228, 248(s), 296, 354, 370(s) | c | 14.4 | 1.1 |
| 60 | Thioquinapiperil (KF31327) | 220060-39-9 | C₂₄H₂₈N₆OS | 211, 268, 363 | - | - | - |
| 61 | Thiosildenafil (Sulfosildenafil, Sildenafil thione) | 479073-79-5 | C₂₂H₃₀N₆O₃S₂ | 228, 250(s), 296, 356, 368(s) | c | 15.2 | 1.2 |
| 62 | Udenafil | 268203-93-6 | C₂₅H₃₆N₆O₄S | 228, 298 | f | 13.6 | 1.0 |
| 63 | Vardenafil | 224785-90-4 | C₂₃H₃₂N₆O₄S | 226, 252(s) | d | 12.1 | 0.9 |
| 64 | Xanthoanthrafil (Benzamidenafil) | 1020251-53-9 | C₁₉H₂₃N₃O₆ | 202, 228, 278, 390 | f | 15.3 | 1.2 |
a Compiled from peer-reviewed literature, and contributed by the USP collaborators. See corresponding chemical structures in Figure 2.
b Retention times derived from the experiments conducted as described in HPLC with Photodiode Array Detection and in HPLC with Mass-Spectrometric Detection methods.
c (s) denotes a shoulder.
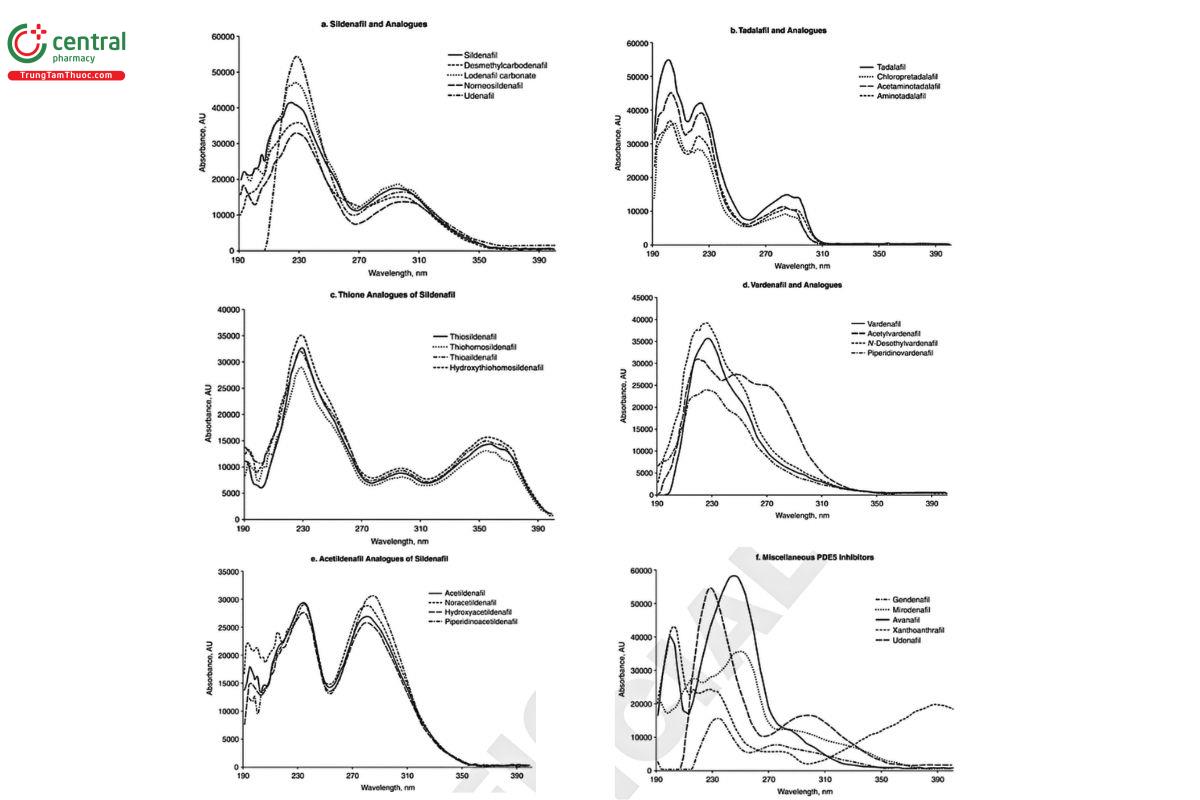
Figure 2. Chemical structures of select PDE5 inhibitors.12
1 In the United States, dietary supplements are defined as substances that are ingested, in agreement with 21 U.S. Code §321 (ff)(2)(A)(1) Definitions of dietary supplements, nutritional supplements, functional foods, and bioactive food additives may vary extensively, depending on local or national legislation. In the marketplace, there is a trend toward expanding the mode of delivery of the adulterating compounds to routes not covered by the regulatory definition for dietary supplements, Le, topical oils, creams, lotions, e-cigarettes, chewing gums, sprays, and others. Such novel delivery systems present unique challenges, particularly from the standpoint of sample preparation, and are not considered for the purposes of this chapter to be dietary supplements. However, recognizing the emerging threat, USP chooses to highlight the existence of these products. In no way should mention of these products be interpreted as a comment on their legal status or be perceived as an expansion of the definition of DS.
2 CacheSyn (http://www.cachesvn.com/); Santa Cruz Biotechnology, Inc. (http://www.scbt.com); TLC Pharmachem (http://www.tlcpharmachem.com/); and Toronto Research Chemicals (http://www.tro-canada.com/) are some of the potential sources of rare and hard-to-find adulterant reference materials.
3 The procedure was developed on the Agilent Technologies Zorbax SB-C18 column.
4 The procedure was developed on the Agilent Technologies Zorbax SB-C18 column.
5 The settings were found appropriate for Thermo Electron LTQ XL Linear lon Trap Mass Spectrometer. Users will need to optimize their respective instrumentation according to the manufacturer's recommendations.
6 32% Ammonia solution is available from EMD Millipore.
7 Procedure was developed using Expression CMS mass spectrometer from Advion, equipped with a TLC-MS interface available from CAMAG. If other mass spectrometers are used, relevant settings will have to be optimized. The bands were directly eluted with a mixture of water and acetonitrile (80:20) containing 0.1% formic acid.
8 The procedures were developed using commercial Promega PDE-Glo™ Phosphodiesterase Assay Kit, Catalog # V1361. It includes the following reagents: PDE-Glo™ Reaction Buffer 5X (Catalog # V133A); PDE-Glo™ Detection Buffer 5X (Catalog # V134A), Protein Kinase A Solution (Catalog# V135A); PDE-Glo™ Termination Buffer 5X (Catalog# V136A); cGMP Stock Solution, 1 mM (Catalog# V641A); CAMP, 1 mM (Catalog # V642B); Kinase-Glo™ Substrate (Catalog#V672A); and Kinase-Glo™ Buffer (Catalog# V673A). Kits from alternative suppliers may also be used, e.g., BPS Science, Catalog #60350, although re-optimization of test procedures will be required.
9 The procedures were developed using human phosphodiesterase 5A from BPS Bioscience Catalog# 60050. The enzyme is available from numerous suppliers, e.g., Sigma-Aldrich Catalog #E9034.
10 Conforming plates are available from Corning (Costar 3912 or Costar 3963), Thermo Scientific (Nunc Catalog #236105), and other vendors.
11 Data contributed by USP collaborators.
12 Data compiled from published sources.


