Saccharin Calcium
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
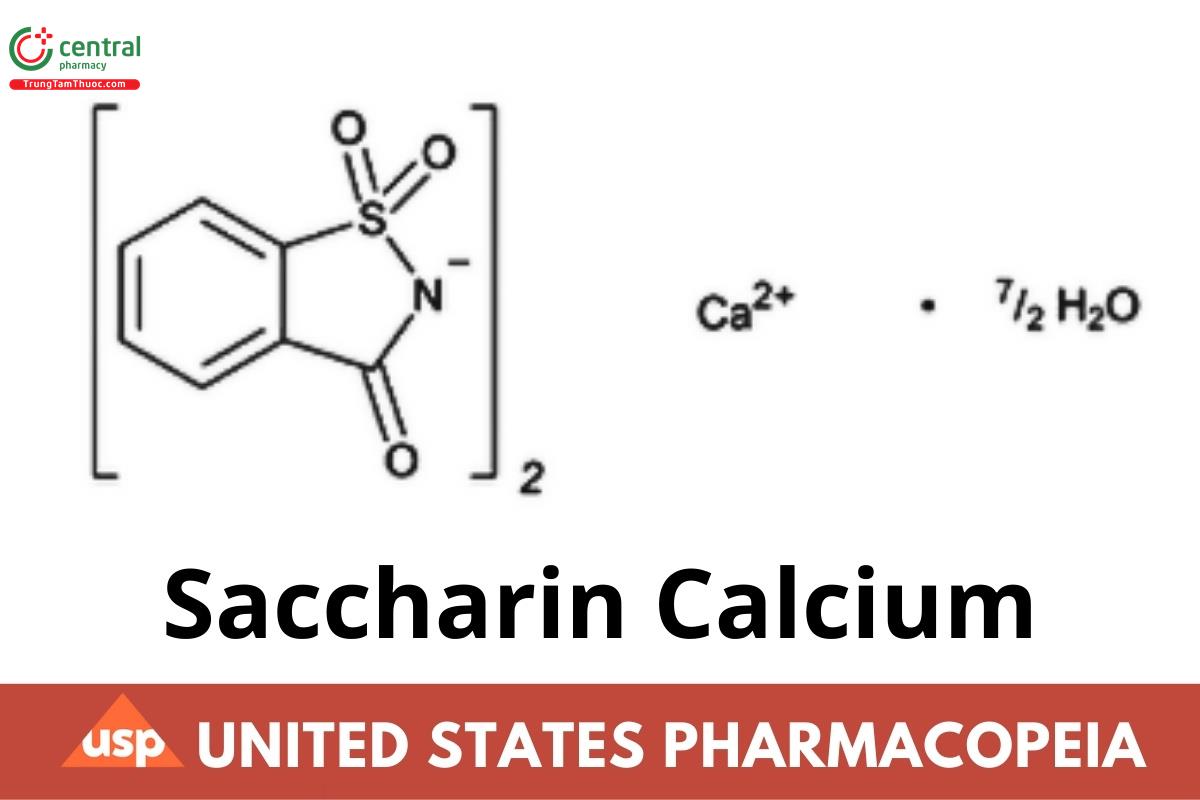
C14H8CaN2O6S · 3½H2O 467.48
C14H8CaN2O6S 404.44
1,2-Benzisothiazol-3(2H)-one, 1,1-dioxide, calcium salt, hydrate (2:7);
1,2-Benzisothiazolin-3-one 1,1-dioxide calcium salt hydrate (2:7) CAS RN®: 6381-91-5.
Anhydrous CAS RN®: 6485-34-3.
1 DEFINITION
Change to read:
Saccharin Calcium contains NLT 98.0% and NMT 102.0% (USP 1-May-2020) of saccharin calcium (C14H8CaN2O6S), calculated on the anhydrous basis.
2 IDENTIFICATION
Change to read:
A. Spectroscopic Identification Tests 〈197〉, Infrared Spectroscopy: 197K (CN 1-May-2020)
Sample: Dry at 105° to constant weight.
Acceptance criteria: Meets the requirements
B.
Sample solution: 100 mg/mL
Analysis: To the Sample solution add 2 drops of methyl red TS, and neutralize with 6 N ammonium hydroxide. Add 3 N hydrochloric acid, dropwise, until the solution is acid to the indicator. Add ammonium oxalate TS.
Acceptance criteria: A white precipitate is formed when the ammonium oxalate is added. This precipitate is insoluble in 6 N acetic acid but dissolves in hydrochloric acid.
C. Calcium salts moistened with hydrochloric acid impart a transient yellowish-red color to a nonluminous flame.
3 ASSAY
Change to read:
3.1 Procedure
Solution A: 50 mM dibasic potassium phosphate (K₂HPO₄) buffer in 0.1% (v/v) phosphoric acid solution
Solution B: Methanol
Mobile phase: See Table 1.
Table 1
| Time (min) | Solution A (%) | Solution B (%) |
| 0 | 90 | 10 |
| 7.0 | 90 | 10 |
| 8.0 | 5 | 95 |
| 10.0 | 5 | 95 |
| 10.1 | 90 | 10 |
| 15.0 | 90 | 10 |
Diluent: Methanol and water (50:50 v/v)
System suitability solution: 0.1 mg/mL of phthalic anhydride and 0.1 mg/mL of USP Saccharin Calcium RS in Diluent
Standard solution: 0.1 mg/mL of USP Saccharin Calcium RS in Diluent
Sample solution: 0.1 mg/mL of Saccharin Calcium in Diluent
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 230 nm
Column: 4.6-mm × 15-cm; 3.5-µm packing L1
Column temperature: 20 ± 5°
Flow rate: 1.0 mL/min
Injection volume: 10 µL
Run time: 15 min
System suitability
Samples: System suitability solution and Standard solution
[Note—The retention times for phthalic anhydride and saccharin calcium are about 6.3 and 7.3 min, respectively. Phthalic anhydride is a potential impurity.]
Suitability requirements
Resolution: NLT 1.5 between the phthalic anhydride and saccharin calcium peaks, System suitability solution
Tailing factor: NMT 1.5, Standard solution
Relative standard deviation: NMT 0.73% for five replicate injections, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of saccharin calcium in the portion of Saccharin Calcium taken:
Result = (rU/rS) × (CS/CU) × 100
rU = peak area of saccharin calcium from the Sample solution
rS = peak area of saccharin calcium from the Standard solution
CS = concentration of USP Saccharin Calcium RS in the Standard solution (mg/mL)
CU = concentration of Saccharin Calcium in the Sample solution (mg/mL)
Acceptance criteria: 98.0%–102.0% on the anhydrous basis (USP 1-May-2020)
4 IMPURITIES
4.1 Limit of Toluenesulfonamides
Internal standard solution: 0.25 mg/mL of caffeine in methylene chloride
Standard stock solution: 20.0 µg/mL of USP o-Toluenesulfonamide RS and 20.0 µg/mL of USP p-Toluenesulfonamide RS in methylene chloride
Standard solution: Evaporate 5.0 mL of Standard stock solution to dryness in a stream of nitrogen. Dissolve the residue in 1.0 mL of the Internal standard solution.
Sample stock solution: 200 mg/mL in water. If necessary, adjust with 1 N sodium hydroxide or 1 N hydrochloric acid to a pH of 7–8 before final dilution.
Sample solution: Shake 50 mL of the Sample stock solution with four quantities each of 50 mL of methylene chloride. Combine the lower layers, dry over anhydrous sodium sulfate, and filter. Wash the filter and the sodium sulfate with 10 mL of methylene chloride. Combine the solution and the washings, and evaporate almost to dryness in a water bath at a temperature not exceeding 40°. Using a small quantity of methylene chloride, quantitatively transfer the residue into a suitable 10-mL tube, evaporate to dryness in a stream of nitrogen, and dissolve the residue in 1.0 mL of the Internal standard solution.
Blank solution: Evaporate 200 mL of methylene chloride to dryness in a water bath at a temperature not exceeding 40°. Dissolve the residue in 1 mL of methylene chloride.
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: GC
Detector: Flame ionization
Column: 0.53-mm × 10-m fused silica; coated with a 2-µm film of phase G3
Temperatures
Injector: 250°
Column: 180°
Detector: 250°
Carrier gas: Nitrogen
Flow rate: 10 mL/min
Injection volume: 1 µL
Injection type: Split ratio, 2:1
System suitability
Samples: Standard solution and Blank solution
[Note—The substances are eluted in the following order: o-toluenesulfonamide, p-toluenesulfonamide, and caffeine.]
Suitability requirements: There are no peaks present in the Blank solution at the retention times of the Internal standard solution, o-toluenesulfonamide, and p-toluenesulfonamide; Blank solution
Resolution: NLT 1.5 between o-toluenesulfonamide and p-toluenesulfonamide, Standard solution
Analysis
Samples: Standard solution and Sample solution
Acceptance criteria: See Table 2. If any peaks due to o-toluenesulfonamide and p-toluenesulfonamide appear in the chromatogram obtained with the Sample solution, the ratio of their areas to that of the Internal standard solution is NMT the corresponding ratio in the chromatogram obtained with the Standard solution.
Table 2
| Name | Acceptance Criteria, NMT (ppm) |
| o-Toluenesulfonamide | 10 |
| p-Toluenesulfonamide | 10 |
4.2 Limit of Benzoate and Salicylate
Sample solution: 50 mg/mL
Analysis: To 10 mL of the Sample solution add 5 drops of 6 N acetic acid and 3 drops of ferric chloride TS.
Acceptance criteria: No precipitate or violet color appears.
5 SPECIFIC TESTS
5.1 Water Determination, Method I 〈921〉: NMT 15.0%
Change to read:
5.2 Readily Carbonizable Substances Test 〈271〉
Matching fluid A: Cobaltous chloride CS, ferric chloride CS, cupric sulfate CS, and water (0.1:0.4:0.1:4.4) (USP 1-May-2020)
Sample solution: 40 mg/mL in sulfuric acid maintained at (USP 1-May-2020) 48°–50° for 10 min
Acceptance criteria: The Sample solution has no more color than Matching fluid A, when viewed against a white background.
5.3 Clarity of Solution
[Note—The Sample solution is to be compared to Reference suspension A and to water in diffused daylight 5 min after preparation of Reference suspension A.]
Diluent: 200-g/L solution of sodium acetate
Hydrazine solution: 10.0 mg/mL of hydrazine sulfate. [Note—Allow to stand for 4–6 h.]
Methenamine solution: Transfer 2.5 g of methenamine to a 100-mL glass-stoppered flask, add 25.0 mL of water, insert the glass stopper, and mix to dissolve.
Primary opalescent suspension: Transfer 25.0 mL of Hydrazine solution to the Methenamine solution in the 100-mL glass-stoppered flask. Mix, and allow to stand for 24 h. [Note—This suspension is stable for 2 months, provided it is stored in a glass container free from surface defects. The suspension must not adhere to the glass and must be well mixed before use.]
Opalescence standard: Transfer 15.0 mL of the Primary opalescent suspension and dilute to 1000 mL. [Note—This suspension should not be used beyond 24 h after preparation.]
Reference suspension A: Opalescence standard and water (1 in 20)
Reference suspension B: Opalescence standard and water (1 in 10)
Sample solution: 200 mg/mL in Diluent
Analysis
Samples: Diluent, Reference suspension A, Reference suspension B, Sample solution, and water
Transfer a sufficient portion of the Sample solution to a test tube of colorless, transparent, neutral glass with a flat base and an internal diameter of 15–25 mm to obtain a depth of 40 mm. Similarly transfer portions of Reference suspension A, Reference suspension B, water, and Diluent to separate matching test tubes. Compare solutions in diffused daylight, viewing vertically against a black background (see Visual Comparison 〈630〉).
Acceptance criteria: The Sample solution shows the same clarity as that of water, or Diluent, or its opalescence is NMT that of Reference suspension A.
5.4 Color of Solution
Diluent A: 200-g/L solution of sodium acetate
Diluent B: 10-g/L solution of hydrochloric acid
Standard stock solution: Ferric chloride CS, Cobaltous chloride CS, cupric sulfate CS, and Diluent B (3.0:3.0:2.4:1.6)
Standard solution: Standard stock solution and Diluent B (1:99). [Note—Prepare the Standard stock solution and (USP 1-May-2020) Standard solution immediately before use.]
Sample solution: Use the Sample solution from the test for Clarity of Solution.
Analysis
Samples: Diluent A, Standard solution, Sample solution, and water
Transfer a sufficient portion of the Sample solution to a test tube of colorless, transparent, neutral glass with a flat base and an internal diameter of 15–25 mm to obtain a depth of 40 mm. Similarly transfer portions of the Standard solution, Diluent A, and water to separate matching test tubes. Compare solutions in diffused daylight, viewing vertically against a white background (see Visual Comparison 〈630〉).
Acceptance criteria: The Sample solution has the appearance of water or Diluent A, or is not more intensely colored than the Standard solution.
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in well-closed containers. Store at room temperature.
Labeling: Where the quantity of saccharin calcium is indicated in the labeling of any preparation containing Saccharin Calcium, this shall be expressed in terms of saccharin (C7H5NO3S).
Change to read:
USP Reference Standards 〈11〉
USP Saccharin Calcium RS
USP o-Toluenesulfonamide RS (USP 1-May-2020)
USP p-Toluenesulfonamide RS (USP 1-May-2020)


