RESIDUAL SOLVENTS
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
- INTRODUCTION
- CLASSIFICATION OF RESIDUAL SOLVENTS BY RISK ASSESSMENT
- CONTROL STRATEGY
- LIMITS OF RESIDUAL SOLVENTS
- OPTIONS FOR DESCRIBING LIMITS OF CLASS 2 AND CLASS 3 RESIDUAL SOLVENTS
- REPORTING LEVELS OF RESIDUAL SOLVENTS
- IDENTIFICATION, CONTROL, AND QUANTIFICATION OF RESIDUAL SOLVENTS
- ANALYTICAL PROCEDURES FOR CLASS 1 AND CLASS 2 RESIDUAL SOLVENTS
- ANALYTICAL PROCEDURES FOR CLASS 3 RESIDUAL SOLVENTS
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
For pharmacopeial purposes, residual solvents in pharmaceuticals are defined as organic volatile chemicals that are used or produced in the manufacturing of drug substances, excipients, or dietary ingredients, or in the preparation of drug products or dietary supplement products. Appropriate selection of the solvent for the synthesis of a drug substance or an excipient may enhance the yield or determine characteristics such as crystal form, purity, and solubility. Therefore, the solvent may sometimes be a critical element in the synthetic process and may not be completely removed by the manufacturing process. Because residual solvents do not provide therapeutic benefit, they should be removed, to the extent possible, to meet safety-based limits, ingredient and product specifications, good manufacturing practices, or other quality-based requirements.
The objective of this general chapter is to define acceptable amounts of residual solvents in pharmaceutical drug products and dietary supplement products for the safety of the patient. Tests for residual solvents are not generally mentioned in specific monographs because the solvents used may vary from one manufacturer to another, however, the limits to be applied must comply with those specified in this general chapter.
This chapter provides procedures for the analysis of residual solvents, although alternative validated methodologies may also be used to demonstrate compliance with the defined limits. For guidance on verification of USP procedures or validation of alternative methods for residual solvents, see Residual Solvents-Verification of Compendial Procedures and Validation of Alternative Procedures (1467). This chapter does not address solvents deliberately used as excipients, nor does it address solvates. The limits specified in this chapter do not apply directly to excipients, drug substances, or dietary ingredients except where specified in the individual monographs. However, residual solvent levels present in drug substances, excipients, and dietary ingredients may be used to demonstrate compliance as an integral part of the control strategy, thereby reducing or eliminating the need for analysis in the product (see 3. Control Strategy).
Information on residual solvents in coating materials, colorants, flavors, capsules, and imprinting inks is generally not needed unless Class 1 solvents are used in the manufacture of these components.
Throughout this chapter, the term "likely to be present" (LTBP) refers to 1) solvents used or produced in the final manufacturing step; 2) solvents used or produced in earlier manufacturing steps that are not consistently removed by a validated process; and 3) solvents properly declared by a validated supplier of a drug substance, excipient, or dietary ingredient.
2 CLASSIFICATION OF RESIDUAL SOLVENTS BY RISK ASSESSMENT
Residual solvents assessed in this general chapter are listed in Appendix 1 by common names and structures. USP is aligned with the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Harmonised Tripartite Guideline for Residual Solvents Q3C (R5) approach for the classification of residual solvents (see Appendix 2 for additional details). These solvents were evaluated for their possible risk to human health and were placed into one of three classes based on their toxicity data and their environmental impact as shown in Table 1.
Table 1. Classification of Residual Solvents and Their Assessments
| Residual Solvent Classes | Assessment |
| Class 1 (solvents to be avoided) | Known human carcinogens |
| Strongly suspected human carcinogens | |
| Solvents particularly known to have ozone-depleting properties | |
| Class 2 (solvents to be limited) | Nongenotoxic animal carcinogens or possible causative agents of other irreversible toxicity, such as neurotoxicity or teratogenicity |
| Solvents suspected of other significant but reversible toxicities | |
| Class 3 (solvents with low toxic potential) | Solvents with low toxic potential to humans; no health-based exposure limit is needed |
The limits for Class 2 solvents are based on the toxicological permitted daily exposure (PDE), calculated as defined in Appendix 3, whereas Class 3 solvents are considered less toxic, and control to 50 mg/day or less for each of these solvents is acceptable without justification. The lists in this chapter are not exhaustive and are subject to revision. For solvents not listed, Appendix 3 may be used to define PDE limits if sufficient toxicological data are available. A flow diagram for potential sources of residual solvents in pharmaceutical drug products and dietary supplements is shown in Figure 1.
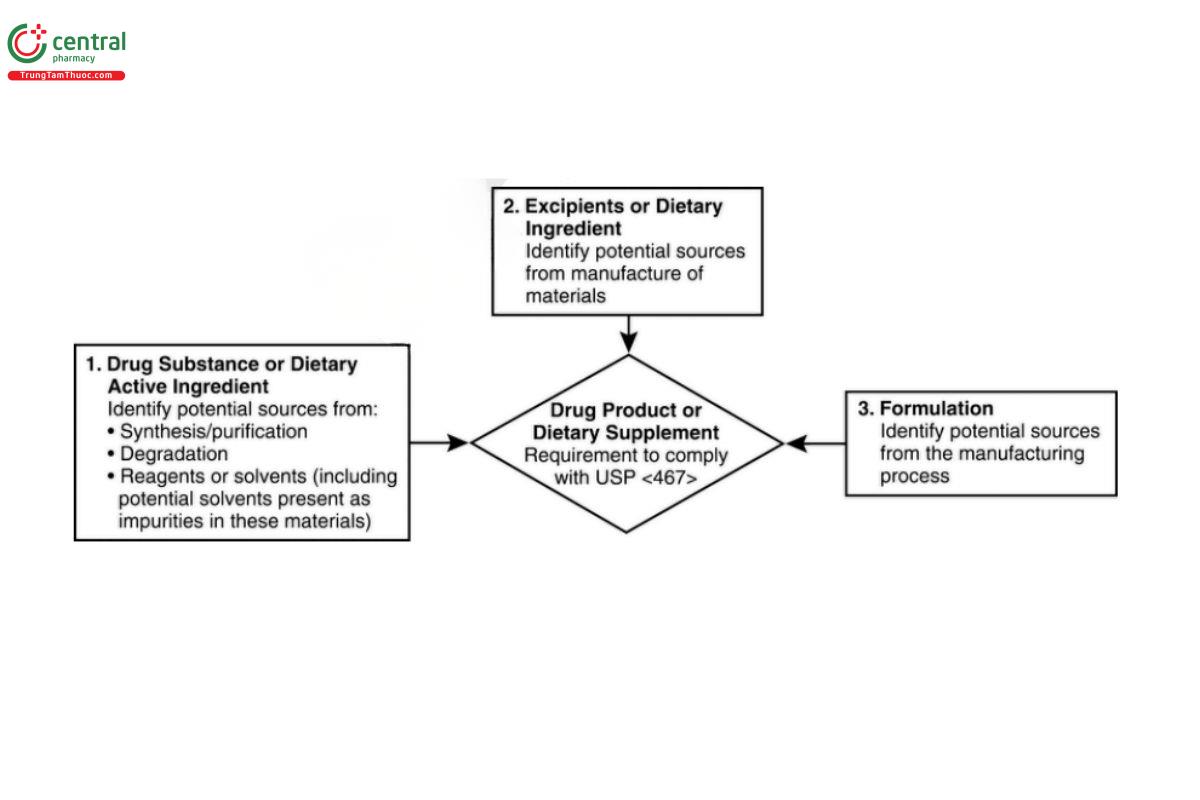
There are three potential sources of residual solvents in the pharmaceutical drug products and dietary supplement products that should be considered (see Figure 1):
1. Drug substance or dietary active ingredient
- Potential solvent sources include their use or formation during the synthesis or purication of the drug substance; their presence in raw materials or reagents used in their synthesis; or degradation of the drug substance.
2. Excipients and/or dietary ingredient
Potential solvent sources include their use or formation during the manufacture or purication of excipients.
3. Formulation
- Potential solvent sources are associated with their use or formation during the official product manufacturing process.
The potential presence of other solvents as impurities in the solvents used must be taken into consideration in the assessment of solvents LTBP. Because of the high toxicity of benzene and other Class 1 solvents, all likely sources of these solvents must be considered. For example, potential sources of benzene may include its presence as an impurity in a solvent used in the manufacturing process, its use in the manufacture of starting material; or its production as a reaction by-product. In the event that the user has insufficient information to complete a thorough assessment of the potential sources of residual solvents (or as an alternative to performing this assessment), solvent screening may be used (see 8. Analytical Procedures for Class 1 and Class 2 Residual Solvents).
Pharmaceutical drug products and dietary supplement products should contain no higher levels of residual solvents than can be supported by safety data. All solvents included in this general chapter are listed in Appendix 1. Those solvents that show toxicity of special concern or carcinogenicity, and/or atmospheric ozone-depletion effects (Class 1, see Table 2), should be avoided in the production of drug substances, dietary supplement ingredients, excipients, pharmaceutical drug products, and dietary supplement products unless their use can be strongly justified in a risk-benefit assessment. Those solvents associated with less severe but still significant toxicity (Class 2, see Table 2) should be limited to protect patients from potential adverse effects. Whenever it is practicable, less toxic solvents (Class 3, see Table 4) should be used. For the purposes of this pharmacopeia, when a manufacturer has received approval from a regulatory authority for the use of a solvent not currently listed in this chapter, or for a level of a listed solvent higher than the limit currently given in this chapter, it is the responsibility of that manufacturer to notify USP of the identity and level of the solvent, and the appropriate test procedure. USP will then address the information in the individual monograph. A new solvent or revised limit that has been approved through the ICH process will be added to the appropriate list in this general chapter. See Appendix 2 for additional background information related to residual solvents.
3 CONTROL STRATEGY
The user may choose to demonstrate compliance with this chapter by analysis for residual solvents in the ocial product or by analysis of the ocial substances. These control strategy options are presented in Figure 2.
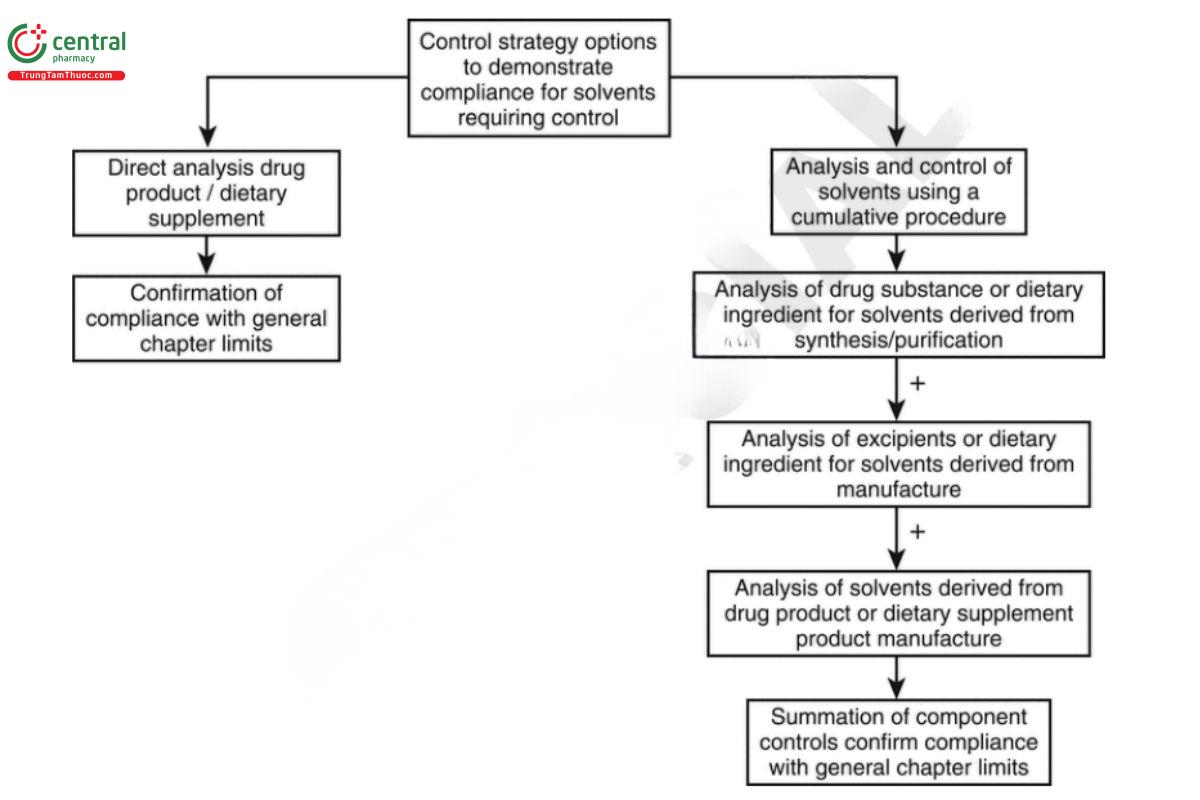
Compliance with the chapter requires that all solvents LTBP comply with the control limits.
The following options are to be considered when designing the control strategy.
1. Testing the official product: This approach is acceptable in all cases.
2. Analysis of the official substances and using a cumulative approach to determine the solvent content in the official product:
A. If no solvent(s) are used during the manufacture of the official product, use the cumulative approach to determine the solvent content in the active pharmaceutical ingredient (API) and excipients or dietary ingredients to calculate solvent levels in the official product.
B. If solvent(s) are used during the manufacture of the official product, use the cumulative approach as described in (a) above. Then, for each solvent used in the manufacture, determine the content in the finished product or after the manufacturing step where the solvent(s) were used.
In all cases, the solvent levels in the final official product must not exceed the limits defined in the chapter.
4 LIMITS OF RESIDUAL SOLVENTS
4.1 Class 1: Solvents to Be Avoided
Class 1 residual solvents (Table 2) should not be used in the manufacture of drug substances, excipients, dietary ingredients, or official products because of their unacceptable toxicities or deleterious environmental effects. However, if their use in order to produce an official product with a significant therapeutic advance is unavoidable, their levels should be restricted as shown in Table 2, unless otherwise stated in the individual monograph. The solvent 1,1,1-trichloroethane is included in Table 2 because it is a severe environmental hazard. The stated limit of 1500 ppm is based on a review of safety data.
The procedures described in 7. Identification, Control, and Quantification of Residual Solvents in this general chapter are to be applied wherever possible. Otherwise an appropriate validated procedure is to be used.
Table 2. Control Limits for Class 1 Residual Solvents in Ocial Products: Solvents to Be Avoided
| Solvent | Concentration Limit (ppm) | Concern |
| Benzene | 2 | Carcinogen |
| Carbon tetrachloride | 4 | Toxic and environmental hazard |
| 1,2-Dichloroethane | 5 | Toxic |
| 1,1-Dichloroethene | 8 | Toxic |
| 1,1,1-Trichloroethane | 1500 | Environmental hazard |
4.2 Class 2: Solvents to Be Limited
Class 2 residual solvents (Table 3) should be limited in drug substances, excipients, dietary ingredients, and ocial products because of the inherent toxicities of these residual solvents. PDEs are given to the nearest 0.1 mg/day, and concentrations are given to the nearest 10 ppm. The method used to establish PDEs for residual solvents is presented in Appendix 3.
Table 3. Class 2 Residual Solvents in Ocial Products
| Solvent | PDE (mg/day) | Concentration Limit (ppm) |
| Acetonitrile | 4.1 | 410 |
| Chlorobenzene | 3.6 | 360 |
| Chloroform | 0.6 | 60 |
| Cumene | 0.7 | 70 |
| Cyclohexane | 38.8 | 3880 |
| 1,2-Dichloroethene | 18.7 | 1870 |
| 1,2-Dimethoxyethane | 1.0 | 100 |
| N,N-Dimethylacetamide | 10.9 | 1090 |
| N,N-Dimethylformamide | 8.8 | 880 |
| 1,4-Dioxane | 3.8 | 380 |
| 2-Ethoxyethanol | 1.6 | 160 |
| Ethylene glycol | 6.2 | 620 |
| Formamide | 2.2 | 220 |
| Hexane | 2.9 | 290 |
| Methanol | 30.0 | 3000 |
| 2-Methoxyethanol | 0.5 | 50 |
| Methylbutylketone | 0.5 | 50 |
| Methylcyclohexane | 11.8 | 1180 |
| Methylene chloride | 6.0 | 600 |
| Methylisobutylketone | 45 | 4500 |
| N-Methylpyrrolidone | 5.3 | 530 |
| Nitromethane | 0.5 | 50 |
| Pyridine | 2.0 | 200 |
| Sulfolane | 1.6 | 160 |
| Tetrahydrofuran | 7.2 | 720 |
| Tetralin | 1.0 | 100 |
| Toluene | 8.9 | 890 |
| Trichloroethylene | 0.8 | 80 |
| Xylenea | 21.7 | 2170 |
a Usually 60% m-xylene, 14% p-xylene, and 9% o-xylene with 17% ethyl benzene.
4.3 Class 3: Solvents with Low Toxic Potential
Class 3 solvents (Table 4) are regarded as less toxic and of lower risk to human health than Class 1 and Class 2 residual solvents. Class 3 includes no solvent known to be a human health hazard at levels normally accepted in pharmaceuticals. However, there are no long-term toxicity or carcinogenicity studies for many of the residual solvents in Class 3. Available data indicate that they are less toxic in acute or short-term studies and negative in genotoxicity studies.
It is considered that amounts of these residual solvents of 50 mg/day or less of each solvent (corresponding to 5000 ppm or 0.5% * w / w Option 1, which is described below) would be acceptable for each solvent without justification. Higher amounts may also be acceptable, provided that they are realistic in relation to manufacturing capability and good manufacturing practice. If a Class 3 solvent limit in an individual monograph is greater than 0.5%, that residual solvent should be identified and quantified. The procedures described in 7. Identification, Control, and Quantification of Residual Solvents, with appropriate modifications to the standard solutions, are to be applied wherever possible. Otherwise, an appropriate validated procedure is to be used.
Table 4. Class 3 Residual Solvents in Official Products
| Acetic acid | Isobutyl acetate |
| Acetone | Isopropyl acetate |
| Anisole | Methyl acetate |
| 1-Butanol | 3-Methyl-1-butanol |
| 2-Butanol | Methylethylketone |
| Butyl acetate | |
| tert-Butylmethyl ether | 2-Methyl-1-propanol |
| Dimethyl sulfoxide | Pentane |
| Ethanol | 1-Pentanol |
| Ethyl acetate | 1-Propanol |
| Ethyl ether | 2-Propanol |
| Ethyl formate | Propyl acetate |
| Formic acid | Triethylamine |
| Heptane |
5 OPTIONS FOR DESCRIBING LIMITS OF CLASS 2 AND CLASS 3 RESIDUAL SOLVENTS
This chapter provides two options for establishing compliance. Both Option 1 and Option 2 are based on the PDE.
5.1 Option 1-Concentration Limit
The concentration limits in ppm stated in Table 3 for Class 2 solvents and the general requirement for Class 3 (5000 ppm, equivalent to 0.5% w/w) are used. The values for Class 2 solvents were calculated using the equation below by assuming a product weight of 10 g administered daily.
Concentration (ppm) = (1000 µg/mg × PDE)/dose
Here, PDE is given in terms of milligrams per day (mg/day), and dose is given in grams per day (g/day). These limits are considered acceptable for all drug substances, excipients, dietary ingredients, and ocial products. Therefore, Option 1 may be applied if the daily amount is not known or does not exceed 10 g. If all ocial substances (drug substances, excipients, and/or dietary ingredients) in a formulation or dietary supplement meet the limits given in Option 1, these components may be used in any proportions. No further calculation is necessary, provided that the daily amount does not exceed 10 g. Products that are administered in doses (daily intake for dietary supplements) greater than 10 g/day are to be considered under Option 2.
5.2 Option 2-Summation of Components Content
Option 2 uses the PDE and the actual maximum daily amount of the product to calculate solvent exposure and assess compliance.
Option 2 must be used to demonstrate compliance with this chapter where the maximum daily dose of the official product exceeds 10 g/day or where at least one component in the formulation exceeds the Option 1 limits.
These limits are applied by summing the amounts of a residual solvent present in each of the components of the official product. The contribution of each solvent per day should result in a total amount that does not exceed the PDE.
Consider the example of the application of Option 1 and Option 2 limits to acetonitrile concentration in an official product. The PDE for acetonitrile is 4.1 mg/day, thus the Option 1 limit is 410 ppm. The maximum administered daily mass of an official product is 5.0 g, and the official product contains two excipients. Assuming that there is no other source of acetonitrile in the manufacturing process, the calculated official product content of acetonitrile and the daily exposure are given in Table 5.
Table 5. Example of Meeting the Requirement for Acetonitrile as per Option 2
| Component | Amount in Formulation (g) | Acetonitrile Content (ppm) | Daily Exposure (mg) |
| Drug substance | 0.3 | 800 | 0.24 |
| Excipient 1 | 0.9 | 400 | 0.36 |
| Excipient 2 | 3.8 | 800 | 3.04 |
| Ocial product | 5.0 | 728 | 3.64 |
Excipient 1 meets the Option 7 limit, but the official substance, Excipient 2, and Official product do not meet the Option 1 limit. Nevertheless, the product meets the Option 2 limit of 4.1 mg/day and thus conforms to the recommendations in this chapter.
Consider another example using acetonitrile as a residual solvent. The maximum administered daily mass of an official product is 5.0 g, and the official product contains two excipients. Assuming no other source of acetonitrile in the manufacturing process, the Official product's content of acetonitrile and the maximum content of residual acetonitrile are given in Table 6.
Table 6. Example of Failing the Requirement for Acetonitrile as per Option 2
| Component | Amount in Formulation (g) | Acetonitrile Content (ppm) | Daily Exposure (mg) |
| Drug substance | 0.3 | 800 | 0.24 |
| Excipient 1 | 0.9 | 2000 | 1.8 |
| Excipient 2 | 3.8 | 800 | 3.04 |
| Ocial product | 5.0 | 1016 | 5.08 |
In this example, the product meets neither the Option 1 nor the Option 2 limit according to the summation. Compliance with the chapter may still be demonstrated via analysis of the Ocial product as it is possible that the formulation process may have reduced the level of acetonitrile below the calculated content. If the level of acetonitrile was not reduced during formulation to the allowed limit, then the manufacturer of the Ocial product should take other steps to reduce the amount of acetonitrile in the ocial product. If the manufacturer receives approval from the regulatory authority for such a higher level of residual solvent, it is the responsibility of that manufacturer to notify USP regarding the identity of this solvent and the approved residual solvent limit in the article. USP will then address this topic in the individual monograph.
6 REPORTING LEVELS OF RESIDUAL SOLVENTS
Manufacturers of pharmaceutical products need certain information about the content of residual solvents in drug substances and excipients to meet the criteria of this chapter. The following statements are given as acceptable examples of the information that could be provided from a supplier of drug substances or excipients to a pharmaceutical manufacturer. The supplier might choose one of the following as appropriate:
- If Class 1 solvents are present, they should be identied and quantied.
- Only Class 2 solvents X, Y,… are LTBP. All are below the Option 1 limit. (Here the supplier would name the Class 2 solvents represented by X, Y,…)
- Only Class 2 solvents X, Y,…, and Class 3 solvents are LTBP. Residual Class 2 solvents are below the Option 1 limit and residual Class 3 solvents are below 0.5% each.
- Only Class 3 solvents are LTBP. Loss on drying (LOD) is not greater than 0.5%.
- If only Class 3 solvents are LTBP and LOD is more than 0.5%, they should be identied and quantied.
- If solvents of Class 2 or Class 3 are present at greater than their Option 1 limits or 0.5%, respectively, they should be identied and quantied to allow compliance with Option 2.
The term LTBP as used in the above examples refers to the solvent used or produced in the nal manufacturing step and to solvents used or produced in earlier manufacturing steps and not removed consistently by a validated process.
7 IDENTIFICATION, CONTROL, AND QUANTIFICATION OF RESIDUAL SOLVENTS
Whenever possible, the substance under test needs to be dissolved to release the residual solvent. The control strategy may involve analysis of official products, active ingredients, excipients, and dietary ingredients. In some cases the sample does not dissolve completely. In such cases it may be acceptable to pulverize the sample into a fine powder carefully, avoiding the generation of frictional heat and in a manner that minimizes the loss of volatile solvents. This technique ensures that any residual solvent that may be present can be released. It should be performed as fast as possible immediately prior to dispersion in the sample solvent, to minimize the loss of volatile solvents during the procedure,
[ NOTE-The organic-free water specified in the following procedures produces no significantly interfering peaks when chromatographed.]
7.1 Class 1 and Class 2 Residual Solvents
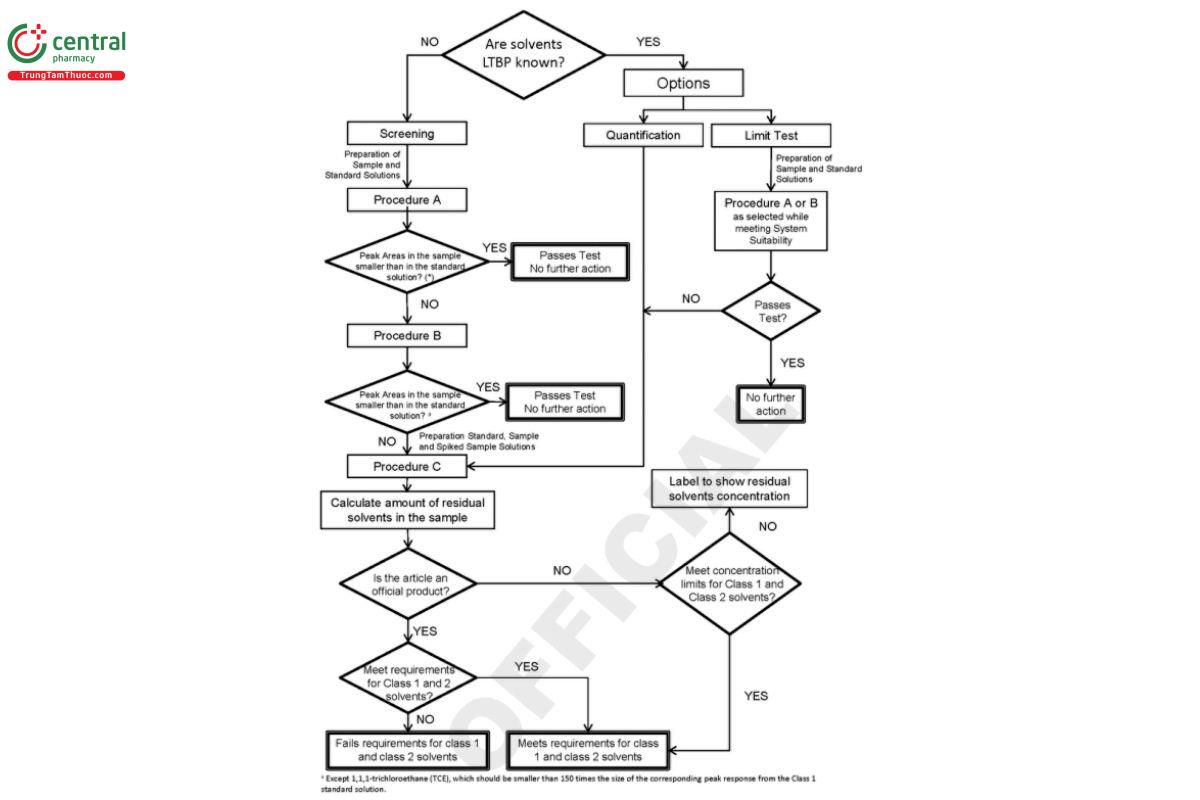
This chapter describes analytical procedures (Procedures A, B, and C) for evaluating the levels of all Class 1 and the majority of Class 2 residual solvents. For each test matrix, verification is needed to demonstrate reliability of the compendial procedure, as described in (1467) When the solvents that are present or potentially present in the sample are known, they can be determined using a limit test such as Procedure A or Procedure B, or by a quantitative test, such as Procedure C.
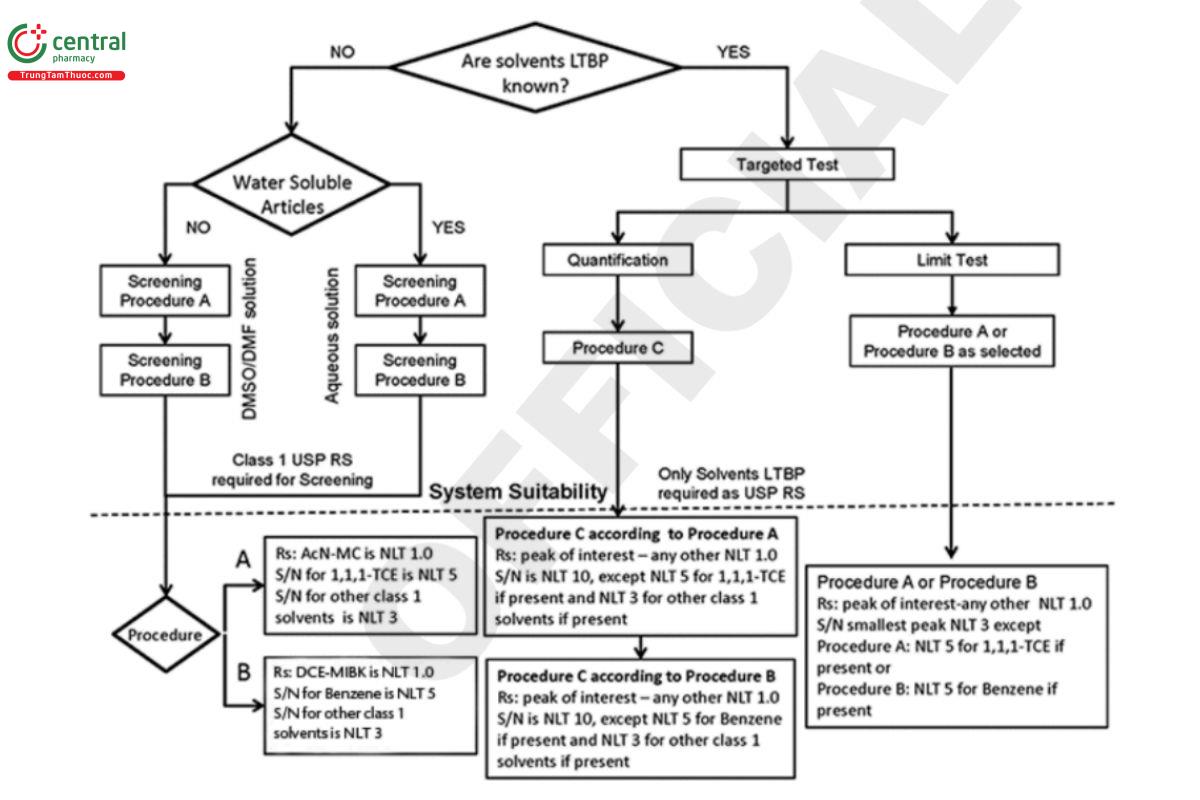
When the solvents LTBP are not known, use the screening tests of Procedure A and Procedure B as required. Procedure A and Procedure B can separate most of the solvents listed in this chapter. Additionally, Eigure 3 can be used as a decision tree to demonstrate compliance.
In a screening test, if the article does not meet the acceptance criteria of Procedure A, then Procedure B is to be used to demonstrate compliance. If the article does not meet the criteria using Procedure A and Procedure B, then Procedure C must be used to quantify the residual solvent(s) present in the article.
When the information about solvents LTBP in the material is known, the system suitability requirements only need to be demonstrated for the solvents expected to be present. Several residual solvents listed in this chapter are not detected at the limit concentration using the analytical procedure described below. Those solvents include formamide, 2-ethoxyethanol, 2-methoxyethanol, ethylene glycol, N methylpyrrolidone, sulfolane, N,N-dimethylacetamide, and N,N-dimethylformamide. Those solvents should be determined using an alternative method that has been appropriately validated to demonstrate that the method is suitable for its intended purpose, l.e., quantitative determination of residual solvent or limit tests for them (see Category II in Validation of Compendial Procedures 〈1225〉, Table 2, which provides denitions and general guidance on analytical procedures validation without indicating specic validation criteria for each characteristic). Chapter 〈1467〉 provides additional guidance on the specic validation criteria for alternative methods used for identication and quantication of residual solvents.
Class 3 Residual Solvents
Procedures A and B can separate most solvents in Classes 1, 2, and 3. However, the procedures have not been validated for Class 3 solvents, and appropriate validation will be required (see the entry for alternative methods under 7.1 Class 1 and Class 2 Residual Solvents).
8 ANALYTICAL PROCEDURES FOR CLASS 1 AND CLASS 2 RESIDUAL SOLVENTS
8.1 Chromatographic Systems
(See Chromatography 〈621〉, System Suitability and Figure 4.)
Procedure A
Mode: GC
Injector: Headspace (see Table 9 for headspace settings)
Injection type: Split ratio, 1:5. [Note—Split ratio can be modied to optimize sensitivity.]
Detector: Flame ionization
Column: 0.32-mm × 30-m capillary fused silica coated with 1.8-µm layer of phase G43 or 0.53-mm × 30-m wide-bore coated with a 3.0-µm layer of phase G43
Column temperature: See Table 7.
Table 7. Procedure A Column Temperature
| Initial Temperature (°) | Temperature Ramp (°/min) | Final Temperature (°) | Hold Time at Final Temperature (min) |
| 40 | 0 | 40 | 20 |
| 40 | 10 | 240 | - |
| 240 | 0 | 240 | 20 |
Temperatures
Injection port: 140°
Detector: 250°
Carrier gas: Helium, nitrogen, or hydrogen
Linear velocity: About 35 cm/s for helium (adjustment and verication are necessary for other carrier gases)
Procedure B
Mode: GC
Injector: Headspace (see Table 9 for headspace settings)
Injection type: Split ratio, 1:5. [Note—Split ratio can be modied to optimize sensitivity.]
Detector: Flame ionization
Column: 0.32-mm × 30-m fused-silica coated with a 0.25-µm layer of phase G16 or a 0.53-mm × 30-m wide-bore coated with a 0.25-µm layer of phase G16
Column temperature: See Table 8.
Table 8. Procedure B Column Temperature
| Initial Temperature (°) | Temperature Ramp (°/min) | Final Temperature (°) | Hold Time at Final Temperature (min) |
| 50 | 0 | 50 | 20 |
| 50 | 6 | 165 | - |
| 165 | 0 | 165 | 20 |
Temperatures
Injection port: 140°
Detector: 250°
Carrier gas: Helium, nitrogen, or hydrogen
Linear velocity: About 35 cm/s for helium (adjustment and verication are necessary for other carrier gases)
procedure c
Use the chromatographic conditions, from either Procedure A or Procedure B, that provide the best performance for the solvent(s) being tested.
headspace parameter settings
There are three general sets of headspace operating parameters that are used for Procedures A and B (Table 9).
Table 9. Headspace Operating Parameters
| Procedure Parameters | Headspace Instrumental Setting | ||
| Set 1 | Set 2 | Set 3 | |
| Equilibration temperature (°) | 80 | 105 | 80 |
| Equilibration time (min) | 60 | 45 | 45 |
| Transfer-line temperature (°) [if appropriate]a | 85 | 110 | 105 |
| Syringe temperature (°) [if appropriate] | 80–90 | 105–115 | 80–90 |
| Carrier gas: helium, nitrogen,b or hydrogenb at an appropriate pressureb | - | - | - |
| Pressurization time(s) (sec) [if appropriate] | ≥60 | ≥60 | ≥60 |
| Injection volume (mL)c | 1 | 1 | 1 |
a It is recommended to increase the temperature of the transfer line between runs to eliminate any potential condensation of solvents.
b The use of nitrogen or hydrogen as the carrier gas requires adjustment and verication.
c Injection volume may be expressed in terms of the injection time.
8.2 Screening of Water-Soluble Articles
STOCK SOLUTIONS
Class 1 standard stock solution: [NOTE-When transferring solutions, place the tip of the pipet just below the surface of the liquid and mix.]
Transfer 1.0 mL of USP Residual Solvents Mixture-Class 1 RS to a 100-mL volumetric flask containing about 9 mL of dimethylsulfoxide, dilute with water to volume, and mix. Transfer 1.0 mL of this solution to a 100-mL volumetric flask containing about 50 mL of water, dilute with water to volume, and mix. Transfer 10 mL of this solution to a 100-mL volumetric flask containing about 50 mL of water, dilute with water to volume, and mix.
Class 2 mixture A standard stock solution: Transfer 1.0 mL of USP Residual Solvents Class 2-Mixture A RS to a 100-mL volumetric flask,
dilute with water to volume, and mix.
Class 2 mixture B standard stock solution: Transfer 1.0 mL of USP Residual Solvents Class 2-Mixture B RS to a 100-mL volumetric flask, dilute with water to volume, and mix..
Sample stock solution: Transfer about 250 mg of the article under test, accurately weighed, to a 25-mL volumetric flask, dissolve in and dilute with water to volume, and mix.
SYSTEM SUITABILITY SOLUTION
Class 1 system suitability solution: Transfer 1.0 mL of Class 1 standard stock solution to an appropriate headspace vial, add 5.0 mL of Sample stock solution, apply the stopper, cap, and mix.
STANDARD SOLUTIONS
Class 1 standard solution: [NOTE-When transferring solutions, place the tip of the pipet just below the surface of the liquid and mix.]
Transfer 1.0 mL of Class 1 standard stock solution to an appropriate headspace vial containing 5.0 mL of water. Apply the stopper, cap, and mix.
Class 2 mixture A standard solution: Transfer 1.0 mL of Class 2 mixture A standard stock solution to an appropriate headspace vial containing 5.0 mL of water, apply the stopper, cap, and mix.
Class 2 mixture B standard solution: Transfer 5.0 mL of Class 2 mixture B standard stock solution to an appropriate headspace vial, add 1.0 mL of water, apply the stopper, cap, and mix.
SAMPLE SOLUTION
Sample solution: Transfer 5.0 mL of Sample stock solution to an appropriate headspace vial, add 1.0 mL of water, apply the stopper, cap, and mix.
PROCEDURE A
Analysis: Using Procedure A Chromatographic System, separately inject (using one of the headspace operating parameter sets described in Table 9) equal volumes of headspace of the Class 1 standard solution, Class 1 system suitability solution, Class 2 mixture A standard solution, Class 2 mixture B standard solution, and Sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements (see Figure 4): The signal-to-noise (S/N) ratio for 1,1,1-trichloroethane in the Class 1 standard solution is NLT 5. The S/N ratio of each peak in the Class 1 system suitability solution is NLT 3, and the resolution between acetonitrile and methylene chloride in the Class 2 mixture A standard solution is NLT 1.0.
Acceptance criteria: If a response of any peak, other than the peak for 1,1,1-TCE, in the Sample solution is greater than or equal to a corresponding peak in either the Class 1 standard solution, or either Class 2 mixture A standard solution or Class 2 mixture B standard solution, or a peak response of 1,1,1-trichloroethane is greater than or equal to 150 times the peak response corresponding to 1,1,1-trichloroethane in the Class 1 standard solution, proceed to Procedure B to verify the identity of the peak, otherwise the article meets the requirements of this test.
PROCEDURE B
Analysis: Using Procedure B Chromatographic System, separately inject (using one of the headspace operating parameter sets described in Table 9) equal volumes of headspace of the Class 1 standard solution, Class 1 system suitability solution, Class 2 mixture A standard solution, Class 2 mixture B standard solution, and Sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements (see Figure 4): The S/N ratio for benzene in the Class 1 standard solution is NLT 5. The S/N ratio of each peak in the Class 1 system suitability solution is NLT 3, and the resolution between methylisobutylketone and cis-dichloroethene in the Class 2 mixture A standard solution is NLT 1.0.
Acceptance criteria: If the response(s) in the Sample solution of the peak(s) identified in Procedure A is/are greater than or equal to corresponding peak(s) in either the Class 1 standard solution, or either Class 2 mixture A standard solution or Class 2 mixture B standard solution, proceed to Procedure C to quantify the peak(s); otherwise the article meets the requirements of this test.
8.3 Screening of Water-Insoluble Articles
STOCK SOLUTIONS
[NOTE-Dimethyl sulfoxide may be substituted as an alternative solvent to dimethylformamide. When transferring solutions, place the tip of the pipet just below the surface of the liquid and mix.]
Class 1 standard stock solution: Transfer 1.0 mL of USP Residual Solvents Mixture-Class 1 RS to a 100-ml volumetric flask previously filled with about 80 mL of dimethylformamide, dilute with dimethylformamide to volume, and mix. Transfer 1.0 mL of this solution to a 100-mL volumetric flask, previously filled with about 80 mL of dimethylformamide, dilute with dimethylformamide to volume, and mix (reserve a portion of this solution for the Class 1 system suitability solution). Transfer 1.0 mL of this solution to a 10-ml volumetric flask, dilute with dimethylformamide to volume, and mix.
Class 2 mixture A standard stock solution: Transfer 1.0 mL of USP Residual Solvents Class 2-Mixture A RS to a 100-ml, volumetric flask containing about 80 mL of dimethylformamide, dilute with dimethylformamide to volume, and mix.
Class 2 mixture B standard stock solution: Transfer 0.5 mL of USP Residual Solvents Class 2-Mixture B RS to a 10-mL volumetric flask, dilute with dimethylformamide to volume, and mix.
Sample stock solution: Transfer about 500 mg of the article under test, accurately weighed, to a 10-mL volumetric flask, dissolve in and dilute with dimethylformamide to volume, and mix.
SYSTEM SUITABILITY SOLUTION
Class 1 system suitability solution: Mix 5 mL of Sample stock solution with 0.5 mL of the intermediate dilution reserved from Class 1 standard stock solution. Transfer 1 mL of this solution to an appropriate headspace vial containing 5 mL of water. Apply the stopper, cap, and mix.
STANDARD SOLUTIONS
Class 1 standard solution: Transfer 1.0 mL of Class 1 standard stock solution to an appropriate headspace vial containing 5.0 mL of water, apply the stopper, cap, and mix.
Class 2 mixture A standard solution: Transfer 1.0 mL of Class 2 mixture A standard stock solution to an appropriate headspace vial containing 5.0 ml of water, apply the stopper, cap, and mix.
Class 2 mixture B standard solution: Transfer 1.0 mL of Class 2 mixture B standard stock solution to an appropriate headspace vial containing 5.0 mL of water, apply the stopper, cap, and mix.
SAMPLE SOLUTION
Sample solution: Transfer 1.0 mL of Sample stock solution to an appropriate headspace vial containing 5.0 mL of water, apply the stopper, cap, and mix.
PROCEDURE A
Analysis: Proceed as directed for Procedure A in 8.2 Screening of Water-Soluble Articles with a split ratio of 1:3. [NOTE-The split ratio can be modified in order to optimize sensitivity.] Separately inject (using one of the headspace operating parameter sets described in Table 9 with a recommended vial pressure of 10 psi) equal volumes of headspace of the Class 1 standard solution, Class 2 mixture A standard solution, Class 2 mixture B standard solution, and Sample solution into the chromatograph, record the chromatograms, and measure the responses for the major peaks.
System suitability requirements (see Figure 4): The S/N ratio for 1,1,1-trichloroethane in the Class 1 standard solution is NLT 5. The S/N
ratio of each peak in the Class 1 system suitability solution is NLT 3; and the resolution between acetonitrile and methylene chloride in the Class 2 mixture A standard solution is NLT 1.0.
Acceptance criteria: If a response of any peak, other than the peak for 1,1,1-TCE in the Sample solution is greater than or equal to a corresponding peak in either the Class 1 standard solution, or either Class 2 mixture A standard solution or Class 2 mixture B standard solution, or a peak response of 1,1,1-trichloroethane is greater than or equal to 150 times the peak response corresponding to 1,1,1-trichloroethane in the Class 1 standard solution, proceed to Procedure B to verify the identity of the peak; otherwise the article meets the requirements of this test.
PROCEDURE B
Analysis: Proceed as directed for Procedure B in 8.2 Screening of Water-Soluble Articles with a split ratio of 1:3. Separately inject (using one of the headspace operating parameter sets described in Table 9 with a recommended vial pressure of 10 psi) equal volumes of headspace of the Class 1 standard solution, Class 2 mixture A standard solution, Class 2 mixture B standard solution, Sample solution, and Class 1 system suitability solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements (see Figure 4): The S/N ratio for benzene in the Class 1 standard solution is NLT 5. The S/N ratio of each peak in the Class 1 system suitability solution is NLT 3; and the resolution between methylisobutylketone and cis-dichloroethene in the Class 2 mixture A standard solution is NLT 1.0.
Acceptance criteria: If the peak response(s) in the Sample solution of the peak(s) identified in Procedure A is/are greater than or equal to corresponding peak(s) in either the Class 1 standard solution, or either Class 2 mixture A standard solution or Class 2 mixture B standard solution, proceed to Procedure C to quantify the peak(s); otherwise the article meets the requirements of this test.
8.4 Limit Tests When Solvents LTBP Are Known
SOLUTIONS
Prepare the standard solution and the Sample solution as directed in 8.2 Screening of Water-Soluble Articles or 8.3 Screening of Water-Insoluble Articles according to the procedure chosen based on the sample solubility. Use USP Reference Standards for those solvents LTBP to prepare the standard solution.
When solvents LTBP are known, only the standards of those solvents LTBP are needed, and determination may be done using a limit test or a quantitative test. Only quantitative tests can be used for Option 2.
System suitability solution: Prepare the System suitability solution according to the solubility of the sample for screening for water-soluble or water-insoluble articles at the concentration of the standard solutions there. Use only those solvents LTBP.
PROCEDURE A
Analysis (limit test): Separately inject (using one of the headspace operating parameter sets described in Table 9) equal volumes of headspace of the System suitability solution, Standard solution, and Sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements (see Figure 4): The S/N ratio for 1,1,1-trichloroethane, if present, is NLT 5. The S/N ratio is NLT 3 for any other solvent. If there is more than one solvent LTBP, the resolution between a peak of interest and any adjacent peak is NLT 1.0.
Acceptance criteria: If the peak response of every peak of interest in the Sample solution is smaller than the corresponding peak in the Standard solution, the article meets the requirements of this test.
PROCEDURE B
Analysis (limit test): Separately inject (following one of the headspace operating parameter sets described in Table 9) equal volumes of headspace of the Standard solution and Sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements (see Figure 4): The S/N ratio for benzene, if present, is NLT 5. The S/N ratio is NLT 3 for any other solvent. If there is more than one solvent LTBP, the resolution between a peak of interest and any other peak is NLT 1.0.
Acceptance criteria: If the peak response of every peak of interest in the Sample solution is smaller than the corresponding peak in the Standard solution, the article meets the requirements of this test.
8.5 Quantitative Tests-Procedure C
QUANTIFICATION FOR WATER-SOLUBLE ARTICLES
(Use the requirement for General or Targeted test, as applicable.)
Sample stock solution: Prepare a Sample stock solution as directed in 8.2 Screening of Water-Soluble Articles, Stock Solutions.
Standard stock solution: (NOTE-Prepare a separate Standard stock solution for each solvent LTBP or potentially LTBP, or identified and verified by Procedure A and Procedure B. For the Class 1 solvents other than 1,1,1-trichloroethane, prepare the first dilution as directed for the first dilution under Class 1 standard stock solution.] Transfer an accurately measured volume of each individual USP Reference Standard corresponding to each residual solvent peak LTBP or identified and verified by Procedure A and Procedure B to a suitable container, and dilute quantitatively and stepwise, if necessary, with water to obtain a solution with a final concentration of 1/20 of the value stated in Table 2 or Table 3 under Concentration Limit.
Standard solution: Transfer 1.0 mL of Standard stock solution to an appropriate headspace vial, add 5.0 mL of water, apply the stopper, cap, and mix.
Sample solution: Prepare as directed in 8.2 Screening of Water-Soluble Articles, Sample Solution.
Spiked sample solution: [NOTE-Prepare a separate Spiked sample solution for each peak LTBP or potentially LTBP or identified and verified by Procedure A and Procedure B.] Transfer 5.0 mL of each Sample stock solution to an appropriate headspace vial, add 1.0 mL of the Standard stock solution, apply the stopper, cap, and mix. The use of a vial containing the sample spiked with multiple standards is permitted, provided that the procedure is validated accordingly.
Analysis: Proceed as directed for Procedure A. [NOTE-If the performance of Procedure B is better than that shown by Procedure A, then Procedure B may be used for quantification.] Separately inject (using one of the headspace operating parameter sets described in Table 9 with a recommended vial pressure of 10 psi) equal volumes of headspace of the Standard solution, Sample solution, and Spiked sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest.
System suitability requirements for Procedure A (see Figure 4):
General: Same as in 8.2 Screening of Water Soluble Articles, Procedure A.
Targeted test: The S/N ratio is NLT 10 for any solvent except NLT 5 for 1,1,1-trichloroethane and NLT 3 for any other Class 1 solvent, if present. If there is more than one solvent LTBP, the resolution between a peak of interest and any adjacent peak is NLT 1.0.
System suitability requirements for Procedure B (see Figure 4):
General: Same as in 8.2 Screening of Water-Soluble Articles, Procedure B.
Targeted test: The S/N ratio is NLT 10 for any solvent except NLT 5 for benzene and NLT 3 for any other Class 1 solvent, if present. If there is more than one solvent LTBP, the resolution between a peak of interest and any adjacent peak is NLT 1.0.
Calculate the amount, in ppm, of each residual solvent found in the article under test:
Result = 5(C/W)[rU /(rST − rU )]
C = concentration of the appropriate USP Reference Standard in the Standard stock solution (µg/mL)
W = weight of the article under test taken to prepare the Sample stock solution (g)
rU = peak responses of each residual solvent obtained from the Sample solution
rST = peak responses of each residual solvent obtained from the Spiked sample solution
QUANTIFICATION FOR WATER-INSOLUBLE ARTICLES
(Use the requirement for General or Targeted test as applicable.)
Sample stock solution: Prepare a Sample stock solution as directed in 8.3 Screening of Water-Insoluble Articles, Stock Solutions. Only the Reference Standard for those solvents identified by Procedure A or Procedure B or LTBP need to be used.
Standard stock solution: [NOTE-Prepare a separate Standard stock solution for each solvent LTBP or potentially LTBP, or identified and verified by Procedure A and Procedure B. For the Class 1 solvents other than 1,1,1-trichloroethane, prepare the first dilution as directed for the first dilution under Class 1 standard stock solution.] Transfer an accurately measured volume of each individual USP Reference Standard corresponding to each residual solvent peak LTBP or identified and verified by Procedure A and Procedure B to a suitable container, and dilute quantitatively and stepwise, if necessary, with water to obtain a solution with a nal concentration of 1/20 of the value stated in Table 2 or Table 3 under Concentration Limit.
Standard solution: Transfer 1.0 mL of Standard stock solution to an appropriate headspace vial, containing 5.0 mL of water, apply the stopper, cap, and mix.
Sample solution: Transfer 1.0 mL of Sample stock solution to an appropriate headspace vial, containing 5.0 mL of water, apply the stopper, cap, and mix.
Spiked sample solution: [NOTE-Prepare a separate Spiked sample solution for each peak LTBP or potentially LTBP, or identified and verified by Procedure A and Procedure B.] Transfer 1.0 mL of each Sample stock solution to an appropriate headspace vial, add 1.0 mL of Standard stock solution and 4.0 mL of water, and apply the stopper, cap, and mix. The use of a vial containing the sample spiked with multiple standards is permitted, provided that the procedure is validated accordingly.
Analysis: Proceed as directed for Procedure A. [NOTE-If the performance of Procedure B is better than that shown by Procedure A, then Procedure B may be used for quantification.] Separately inject (using one of the headspace operating parameter sets described in Table 9 with a recommended vial pressure of 10 psi) equal volumes of headspace of the Standard solution, Sample solution, and Spiked sample solution into the chromatograph, record the chromatograms, and measure the responses for the peaks of interest. If Class 1 solvents are LTBP, also inject the Class 1 system suitability solution.
System suitability requirements for Procedure A (see Figure 4):
General: Same as in 8.3 Screening of Water-Insoluble Articles, Procedure A.
Targeted test: The S/N ratio is NLT 10 for any solvent except NLT 5 for 1,1,1-trichloroethane and NLT 3 for any other Class 1 solvent, if present. If there is more than one solvent LTBP, the resolution between a peak of interest and any adjacent peak is NLT 1.0.
System suitability requirements for Procedure B (see Figure 4):
General: Same as in 8.3 Screening of Water-Insoluble Articles, Procedure B.
Targeted test: The S/N ratio is NLT 10 for any solvent except NLT 5 for benzene and NLT 3 for any other Class 1 solvent, if present. If there is more than one solvent LTBP, the resolution between a peak of interest and any adjacent peak is NLT 1.0.
Calculate the amount, in ppm, of each residual solvent found in the article under test:
Result = 10(C/W)[rU /(rST − rU )]
C = concentration of the appropriate USP Reference Standard in the Standard stock solution (µg/mL)
W = weight of the article under test taken to prepare the Sample stock solution (g)
rU = peak responses of each residual solvent obtained from the Sample solution
rST = peak responses of each residual solvent obtained from the Spiked sample solution
9 ANALYTICAL PROCEDURES FOR CLASS 3 RESIDUAL SOLVENTS
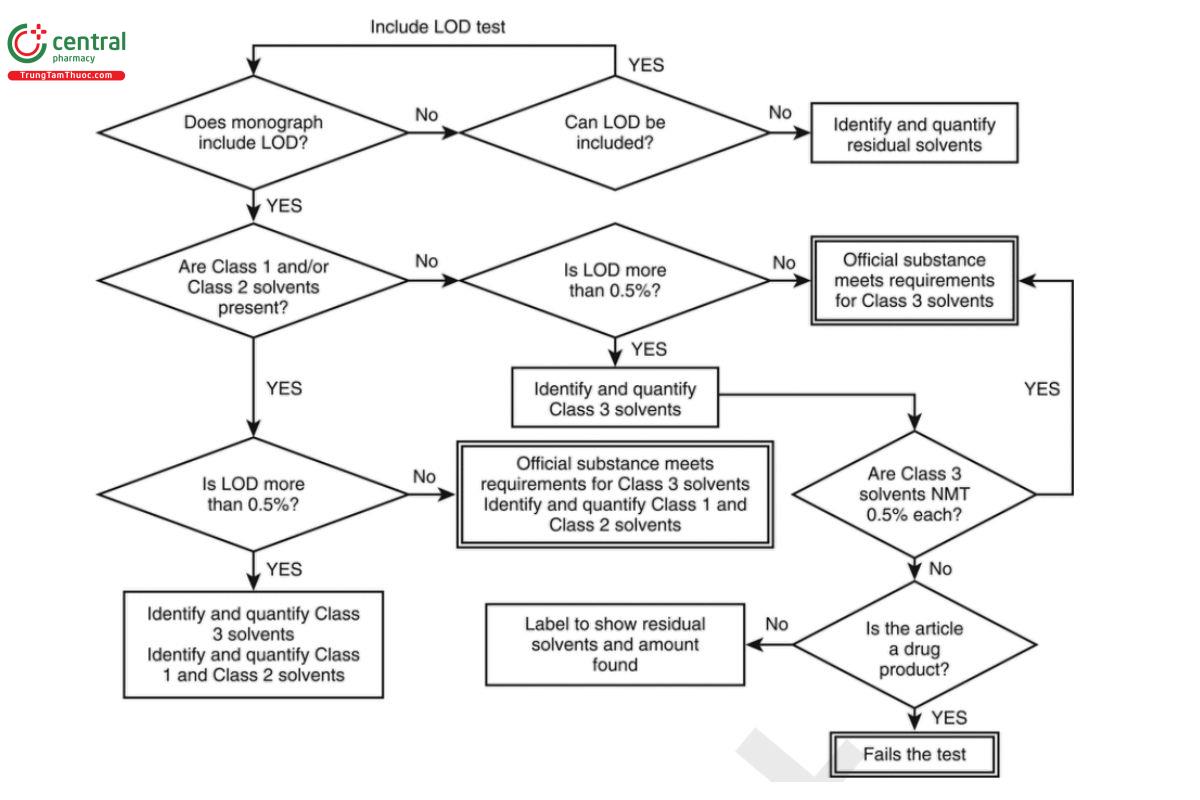
If only Class 3 solvents are present, and the monograph for the article under test contains an LOD procedure specifying an upper limit of NMT 0.5% (per Option 1 in this chapter), then the level of residual solvent can be determined as directed in Loss on Drying (731). If Class 3 solvents are present with other solvents, e.g., Class 1 or Class 2 solvents, or if there is no LOD procedure in the monograph for the article under test, or if the limit for Class 3 solvent(s) in an individual monograph is greater than 50 mg/day (corresponding to 5000 ppm or 0.5% under Option 1), then the Class 3 residual solvent or solvents present in the article under test should be identified and quantified. In that case, the procedures described in this chapter, with appropriate adjustments, are to be applied wherever possible and USP Reference Standards, where available, should be used in these procedures. Such procedures should be properly validated as described in (1467), If Class 1 or Class 2 solvents are present and LOD is less than 0.5%, then identification of Class 3 solvents is not required and only Class 1 and Class 2 solvents should be identified and quantified. See Figure 5.
GLOSSARY
Genotoxic carcinogens: Carcinogens that produce cancer by affecting genes or chromosomes.
Likely to be present (LTBP): Those solvents that are 1) used or produced in the final manufacturing step; 2) used or produced in earlier manufacturing steps but they are not consistently removed by a validated process; 3) the solvents properly declared by a validated supplier of a drug substance, excipient, or dietary ingredient.
Lowest-observed-effect level (LOEL): The lowest dose of a substance in a study or group of studies that produces biologically significant increases in frequency or severity of any effects in exposed humans or animals.
Modifying factor: A factor determined by professional judgment of a toxicologist and applied to bioassay data so that the data can be safely related to humans.
Neurotoxicity: The ability of a substance to cause adverse effects on the nervous system.
No-observed-effect level (NOEL): The highest dose of a substance at which there are no biologically significant increases in frequency or severity of any effects in exposed humans or animals.
Permitted daily exposure (PDE): The maximum acceptable intake per day of a residual solvent in pharmaceutical products or dietary supplements
Reversible toxicity: The occurrence of harmful effects that are caused by a substance and then disappear after the exposure to the substance ends.
Strongly suspected human carcinogen: A substance for which there is no epidemiological evidence of carcinogenesis but for which there are positive genotoxicity data and clear evidence of carcinogenesis in rodents.
Teratogenicity: The occurrence of structural malformations in a developing fetus when a substance is administered during pregnancy.
APPENDICES
Appendix 1: Residual Solvents Included in this General Chapter
See Table A-1 for a list of residual solvents included in this chapter.
Table A-1. List of Residual Solvents Included in this General Chapter
| Solvent | Other Names | Structure | Class |
| Acetic acid | Ethanoic acid | CH3COOH | Class 3 |
| Acetone | 2-Propanone; Propan-2-one | CH3COOH3 | Class 3 |
| Acetonitrile | AcN | CH3CN | Class 2 |
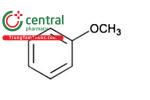
| Anisole | Methoxybenzene |
| Class 3 |
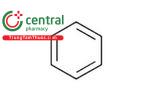
| Benzene | Benzal |
| Class 1 |
| 1-Butanol | n-Butyl alcohol; Butan-1-ol | CH3(CH2)3OH | Class 3 |
| 2-Butanol | sec-Butyl alcohol; Butan-2-ol | CH3CH2CH(OH)CH3 | Class 3 |
| Butyl acetate | Acetic acid butyl ester | CH3COO(CH2)3CH3 | Class 3 |
| tert-Butylmethyl ether | 2-Methoxy-2-methylpropane | (CH3)3COCH3 | Class 3 |
| Carbon tetrachloride | Tetrachloromethane | CCl4 | Class 1 |
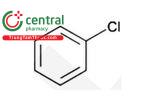
| Chlorobenzene |
| Class 2 | |
| Chloroform | Trichloromethane | CHCl3 | Class 2 |
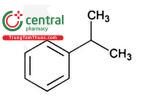
| Cumene | Isopropylbenzene, (1-Methylethyl)benzene |
| Class 2 |
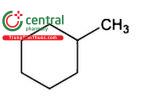
| Cyclohexane | Hexamethylene |
| Class 2 |
| 1,2-Dichloroethane | sym-Dichloroethane, Ethylene dichloride; Ethylene chloride | CH2ClCH2Cl | Class 1 |
| 1,1-Dichloroethene | 1,1-Dichloroethylene, Vinylidene chloride. | H2C=CCl2 | Class 1 |
| 1,2-Dichloroethene | 1,2-Dichloroethylene, Acetylene dichloride, DCE | ClHC=CHCl | Class 2 |
| 1,2-Dimethoxyethane | Ethyleneglycol dimethyl ether, Monoglyme: Dimethyl cellosolve | H3COCH2CH2OCH3 | Class 2 |
| N,N-Dimethylacetamide | DMA | CH3CON(CH3)2 | Class 2 |
| N,N-Dimethylformamide | Dimethylformamide | HCON(CH3)2 | Class 2 |
| Dimethyl sulfoxide | Methylsulfinylmethane, Methyl sulfoxide; Dimethyl sulfoxide | (CH3)2SO | Class 3 |
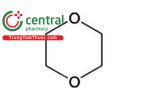
| 1,4-Dioxane | p-Dioxane, [1,4] Dioxane |
| Class 2 |
| Ethanol | Ethyl alcohol | CH3CH2OH | Class 3 |
| 2-Ethoxyethanol | Cellosolve | CH3CH2OCH2CH2OH | Class 2 |
| Ethyl acetate | Acetic acid ethyl ester | CH3COOCH2CH3 | Class 3 |
| Ethylene glycol | 1,2-Dihydroxyethane 1,2-Ethanediol | HOCH2CH2OH | Class 2 |
| Ethyl ether | Diethyl ether, Ethoxyethane: 1,1-Oxybisethane | CH3CH2OCH2CH3 | Class 3 |
| Ethyl formate | Formic acid ethyl ester | HCOOCH2CH3 | Class 3 |
| Formamide | Methanamide | HCONH2 | Class 2 |
| Formic acid | HCOOH | Class 3 | |
| Heptane | n-Heptane | CH3(CH2)5CH3 | Class 3 |
| Hexane | n-Hexane | CH3(CH2)4CH3 | Class 2 |
| Isobutyl acetate | Acetic acid isobutyl ester | CH3COOCH2CH(CH3)2 | Class 3 |
| Isopropyl acetate | Acetic acid isopropyl ester | CH3COOCH(CH3)2 | Class 3 |
| Methanol | Methyl alcohol | CH3OH | Class 2 |
| 2-Methoxyethanol | Methyl cellosolve | CH3OCH2CH2OH | Class 2 |
| Methyl acetate | Acetic acid methyl ester | CH3COOCH3 | Class 3 |
| 3-Methyl-1-butanol | Isoamyl alcohol: Isopentyl alcohol, 3-Methylbutan-1-ol | (CH3)2CHCH2CH2OH | Class 3 |
| Methylbutylketone | 2-Hexanone, Hexan-2-one | CH3(CH2)3COCH3 | Class 2 |
| Methylcyclohexane | Cyclohexylmethane |
| Class 2 |
| MethyMethylene chloridelcyclohexane | Dichloromethane, MC | CH2Cl2 | Class 2 |
| Methylethylketone | 2-Butanone MEK Butan-2-one | CH3CH2COCH3 | Class 3 |
| Methylisobutylketone | 4-Methylpentan-2-one, 4-Methyl-2-pentanone MIBK | CH3COCH2CH(CH3)2 | Class 2 |
| 2-Methyl-1-propanol | Isobutyl alcohol; 2-Methylpropan-1-ol | (CH3)2CHCH2OH | Class 3 |
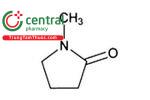
| N-Methylpyrrolidone | 1-Methylpyrrolidin-2-one, 1-Methyl-2-pyrrolidinone |
| Class 2 |
| Nitromethane | CH3NO2 | Class 2 | |
| Pentane | n-Pentane | CH3(CH2)3CH3 | Class 3 |
| 1-Pentanol | Amyl alcohol: Pentan-1-ol; Pentyl alcohol | CH3(CH2)3CH2OH | Class 3 |
| 1-Propanol | Propan-1-ol Propyl alcohol | CH3CH2CH2OH | Class 3 |
| 2-Propanol | Propan-2-ol Isopropyl alcohol | (CH3)2CHOH | Class 3 |
| Propyl acetate | Acetic acid propyl ester | CH3COOCH2CH2CH3 | Class 3 |
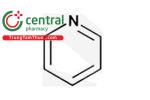
| Pyridine |
| Class 2 | |
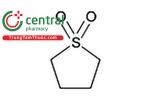
| Sulfolane | Tetrahydrothiophene 1,1-dioxide |
| Class 2 |
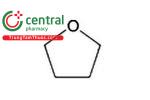
| Tetrahydrofuran | Tetramethylene oxide, Oxacyclopentane |
| Class 2 |
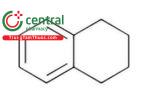
| Tetralin | 1,2,3,4-Tetrahydronaphthalene |
| Class 2 |
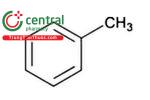
| Toluene | Methylbenzene |
| Class 2 |
| 1,1,1-Trichloroethan | Methylchloroform; 1,1,1-TCE | CH3CCl3 | Class 1 |
| Trichloroethylene | 1,1,2-Trichloroethene | HClC=CCl2 | Class 2 |
| Triethylamine | N,N-Diethylethanamine | (CH3CH2)3N | Class 3 |
| Xylenea | Dimethylbenzene, Xylol |
| Class 2 |
a Usually 60% m-xylene, 14% p-xylene, and 9% o-xylene with 17% ethyl benzene
Appendix 2: Additional Background
environmental regulation of organic volatile solvents
Several of the residual solvents frequently used in the production of pharmaceuticals are listed as toxic chemicals in Environmental Health Criteria (EHC) monographs and in the Integrated Risk Information System (IRIS) and other similar reference material. The approach to risk assessment described therein relates primarily to long-term or lifetime exposure of the general population via the ambient environment (je inhaled air, food, drinking water, and other sources). The objectives of such groups as the International Programme on Chemical Safety (IPCS), the U.S. Environmental Protection Agency (EPA), and the U.S. Food and Drug Administration (FDA) include the determination of acceptable exposure levels. The goal is maintenance of environmental integrity and protection of human health against the possible deleterious effects of chemicals resulting from long-term environmental exposure. The procedures involved in the estimation of maximum safe exposure limits are usually based on long-term studies. When long-term study data are unavailable, shorter-term study data can be used with modification of the approach, such as use of larger safety factors.
RESIDUAL SOLVENTS IN PHARMACEUTICALS
Exposure limits in this chapter were established by referring to methodologies and toxicity data described in EHC and IRIS monographs. However, the following specific assumptions about residual solvents to be used in the synthesis and formulation of pharmaceutical products should be taken into account in establishing exposure limits.
1. Patients (not the general population) use pharmaceuticals to treat their diseases or for prophylaxis to prevent infection or disease.
2. The assumption of lifetime patient exposure is not necessary for most pharmaceutical products but may be appropriate as a working hypothesis to reduce risk to human health.
3. Residual solvents are unavoidable components in pharmaceutical production and will often be a part of pharmaceutical drug products.
4. Residual solvents should not exceed recommended levels except in exceptional circumstances
5. Data from toxicological studies that are used to determine acceptable levels for residual solvents should have been generated using appropriate protocols such as those described by the Organization for Economic Cooperation and Development (OECD), the EPA, and the FDA Redbook or derived from alternative sources also considered to be adequate to support PDE limit setting.
Appendix 3: Procedures for Establishing Exposure Limits
The Gaylor-Kodell method of risk assessment (Gaylor, DW, Kodell, RL. Linear interpolation algorithm for low dose risk assessment of toxic substances. J Environ Pathol, Toxicol, 1980;4:305-312) is appropriate for Class 1 carcinogenic solvents. Only in cases where reliable carcinogenicity data are available should extrapolation by the use of mathematical models be applied to setting exposure limits. Exposure limits for Class 1 residual solvents could be determined with the use of a large safety factor (ie 10,000-100,000) with respect to the no observed-effect level (NOEL). Detection and quantification of these residual solvents should be performed using state-of-the-art analytical techniques.
Acceptable exposure levels in this chapter for Class 2 residual solvents were established by calculation of PDE values according to the procedures for setting exposure limits in pharmaceuticals (page 5748 of Pharmacopeial Forum (Nov.-Dec. 1989]), and the method adopted by IPCS for Assessing Human Health Risks of Chemicals (Environmental Health Criteria 170, WHO, 1994), detailed calculations are published in Pharmeuropa, Vol. 9, No. 1, Supplement, April 1997. These procedures are similar to those used by the U.S. EPA (IRIS) and the U.S. FDA (Redbook) and others. The method is outlined here to give a better understanding of the origin of the PDE values. It is not necessary to perform these calculations in order to use the PDE values presented in Table 3 of this document.
PDE is derived from the NOEL, or the lowest-observed-effect level (LOEL), in the most relevant animal study as follows:
PDE = (NOEL × Weight Adjustment)/(F1 × F2 × F3 × F4 × F5)
The PDE is derived preferably from a NOEL. If no NOEL is obtained, the LOEL may be used. Modifying factors proposed here, for relating the data to humans, are the same kind of “uncertainty factors” used in Environmental Health Criteria (Environmental Health Criteria 170, WHO, 1994) and “modifying factors” or “safety factors” used in Pharmacopeial Forum. The assumption of 100% systemic exposure is used in all calculations regardless of the route of administration.
The modifying factors are as follows:
F1 = A factor to account for extrapolation between species.
F1 = 5 for extrapolation from rats to humans
F1 = 12 for extrapolation from mice to humans
F1 = 2 for extrapolation from dogs to humans
F1 = 2.5 for extrapolation from rabbits to humans
F1 = 3 for extrapolation from monkeys to humans
F1 = 10 for extrapolation from other animals to humans
F1 takes into account the comparative surface area to body weight ratios for the species concerned and for humans. Surface area (S) is
calculated as:
S = kM0.67
k = a constant that has been taken to be 10
M = body weight
The body weights used in the equation are those shown below in Table A-2:
F2 = A factor of 10 to account for variability between individuals.
A factor of 10 is generally given for all organic solvents, and 10 is used consistently in this chapter.
F3 = A variable factor to account for toxicity studies of short-term exposure
F3 = 1 for studies that last at least one half lifetime (1 year for rodents or rabbits; 7 years for cats, dogs, and monkeys)
F3 = 1 for reproductive studies in which the whole period of organogenesis is covered
F3 = 2 for a 6-month study in rodents, or a 3.5-year study in non-rodents
F3 = 5 for a 3-month study in rodents, or a 2-year study in non-rodents
F3 = 10 for studies of a shorter duration
In all cases, the higher factor has been used for study durations between the time points (e.g., a factor of 2 for a 9-month rodent study).
F4 = A factor that may be applied in cases of severe toxicity, e.g., nongenotoxic carcinogenicity, neurotoxicity, or teratogenicity. In studies of reproductive toxicity, the following factors are used:
F4 = 1 for fetal toxicity associated with maternal toxicity
F4 = 5 for fetal toxicity without maternal toxicity
F4 = 5 for a teratogenic effect with maternal toxicity
F4 = 10 for a teratogenic effect without maternal toxicity
F5 = A variable factor that may be applied if the no-effect level was not established.
When only a LOEL is available, a factor of up to 10 can be used, depending on the severity of the toxicity. The weight adjustment assumes an arbitrary adult human body weight for either sex of 50 kg. This relatively low weight provides an additional safety factor against the standard weights of 60 or 70 kg that are often used in this type of calculation. It is recognized that some adult patients weigh less than 50 kg; these patients are considered to be accommodated by the built-in safety factors used to determine a PDE. If the solvent was present in a
formulation specically intended for pediatric use, an adjustment for a lower body weight would be appropriate.
As an example of the application of this equation, consider a toxicity study of acetonitrile in mice that is summarized in Pharmeuropa, 1997;9(1): S24. The NOEL is calculated to be 50.7 mg kg−1 day−1. The PDE for acetonitrile in this study is calculated as follows:
PDE = (50.7 mg kg−1 day−1 × 50 kg)/(12 × 10 × 5 × 1 × 1) = 4.22 mg day−1
In this example,
F1 = 12 to account for the extrapolation from mice to humans
F2 = 10 to account for differences between individual humans
F3 = 5 because the duration of the study was only 13 weeks
F4 = 1 because no severe toxicity was encountered
F5 = 1 because the no-effect level was determined
Table A-2. Values Used in the Calculations in This Chapter
| Rat body weight | 425 g |
| Pregnant rat body weight | 330 g |
| Mouse body weight | 28 g |
| Pregnant mouse body weight | 30 g |
| Guinea-pig body weight | 500 g |
| Rhesus monkey body weight | 2.5 kg |
| Rabbit body weight (pregnant or not) | 4 kg |
| Beagle dog body weight | 11.5 kg |
| Rat respiratory volume | 290 L/day |
| Mouse respiratory volume | 43 L/day |
| Rabbit respiratory volume | 1440 L/day |
| Guinea-pig respiratory volume | 430 L/day |
| Human respiratory volume | 28,800 L/day |
| Dog respiratory volume | 9000 L/day |
| Monkey respiratory volume | 1150 L/day |
| Mouse water consumption | 5mL/day |
| Rat water consumption | 30 mL/day |
| Rat food consumption | 30 g/day |
The equation for an ideal gas, PV = nRT, is used to convert concentrations of gases used in inhalation studies from units of ppm to units of mg/L or mg/m3. Consider as an example the rat reproductive toxicity study by inhalation of carbon tetrachloride (molecular weight, 153.84) summarized in Pharmeuropa, 1997:9(1):S9.
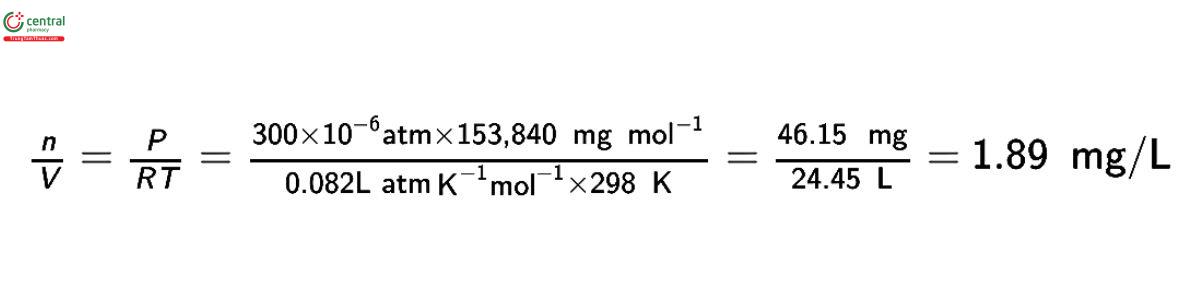
The relationship 1000 L = 1 m3 is used to convert to mg/m3.
Change to read:
USP Reference Standards 〈11〉
USP 1-Butanol RS
USP 2-Butanol RS
USP 2-Methyl-1-Propanol RS
USP 3-Methyl-1-butanol RS
USP 1-Pentanol RS
USP 1-Propanol RS
USP 2-Propanol RS
USP Acetone RS
USP Alcohol Determination±Alcohol RS
USP Anisole RS
USP Butyl Acetate RS
USP tert-Butylmethyl Ether RS
USP Dimethyl Sulfoxide RS
USP Ethyl Acetate RS
USP Ethyl Formate RS
USP Formic Acid RS
USP Glacial Acetic Acid RS
USP Heptane RS
USP Isobutyl Acetate RS
USP Isopropyl Acetate RS
USP Methyl Acetate RS
USP Methyl Ethyl Ketone RS
USP Pentane RS
USP Propyl Acetate RS
USP Residual Solvents Mixture—Class 1 RS
USP Residual Solvent Class 1—Benzene RS
USP Residual Solvent Class 1—Carbon Tetrachloride RS
USP Residual Solvent Class 1—1,2-Dichloroethane RS
USP Residual Solvent Class 1—1,1-Dichloroethene RS
USP Residual Solvent Class 1—1,1,1-Trichloroethane RS
USP Residual Solvents Class 2—Mixture A RS
USP Residual Solvents Class 2—Mixture B RS
USP Residual Solvents Class 2—Mixture C RS (ERR 1-Sep-2022)
USP Residual Solvent Class 2—Acetonitrile RS
USP Residual Solvent Class 2—Chlorobenzene RS
USP Residual Solvent Class 2—Chloroform RS
USP Residual Solvent Class 2—Cumene RS
USP Residual Solvent Class 2—Cyclohexane RS
USP Residual Solvent Class 2—1,2-Dichloroethene RS
USP Residual Solvent Class 2—1,2-Dimethoxyethane RS
USP Residual Solvent Class 2—N,N-Dimethylacetamide RS
USP Residual Solvent Class 2—N,N-Dimethylformamide RS
USP Residual Solvent Class 2—1,4-Dioxane RS
USP Residual Solvent Class 2—2-Ethoxyethanol RS
USP Residual Solvent Class 2—Ethylene Glycol RS
USP Residual Solvent Class 2—Hexane RS
USP Residual Solvent Class 2—Methanol RS
USP Residual Solvent Class 2—2-Methoxyethanol RS
USP Residual Solvent Class 2—Methylbutylketone RS
USP Residual Solvent Class 2—Methylcyclohexane RS
USP Residual Solvent Class 2—Methylene Chloride RS
USP Residual Solvent Class 2—Methylisobutylketone RS
USP Residual Solvent Class 2—N-Methylpyrrolidone RS
USP Residual Solvent Class 2—Nitromethane RS
USP Residual Solvent Class 2—Pyridine RS
USP Residual Solvent Class 2—Tetrahydrofuran RS
USP Residual Solvent Class 2—Tetralin RS
USP Residual Solvent Class 2—Toluene RS
USP Residual Solvent Class 2—Trichloroethylene RS
USP Residual Solvent Class 2—Xylenes RS
USP Triethylamine RS