Quinidine Sulfate
If you find any inaccurate information, please let us know by providing your feedback here
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
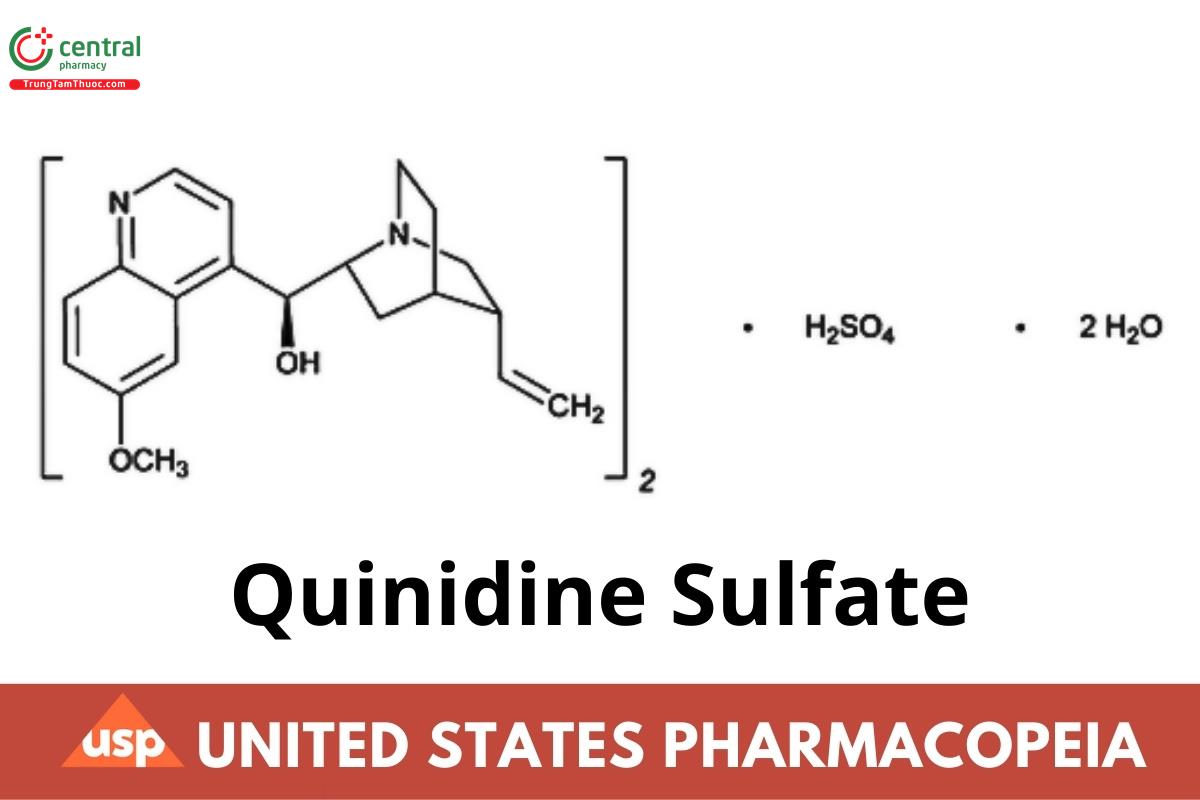
(C20H24N2O2)2 · H2SO4 · 2H2O 782.94
(C20H24N2O2)2 · H2SO4 746.93
Cinchonan-9-ol, 6′-methoxy-, (9S)-, sulfate (2:1) (salt), dihydrate;
Quinidine sulfate (2:1) (salt) dihydrate
CAS RN®: 6591-63-5; UNII: J13S2394HE.
Anhydrous CAS RN®: 50-54-4; UNII: 140CU2322K.
1 DEFINITION
Quinidine Sulfate is the sulfate of an alkaloid obtained from various species of Cinchona and their hybrids and from Remijia pedunculata Flückiger (Fam. Rubiaceae), or prepared from Quinine. It contains NLT 99.0% and NMT 101.0% of total alkaloid salt, calculated as Quinidine Sulfate [(C20H24N2O2)2 · H2SO4 · 2H2O], on the anhydrous basis.
2 IDENTIFICATION
2.1 A.
Sample solution: 1-in-2000 solution of Quinidine Sulfate in dilute sulfuric acid (1 in 350)
Acceptance criteria: The Sample solution exhibits a vivid blue fluorescence. On the addition of a few drops of hydrochloric acid, the fluorescence disappears.
2.2 B.
The R value of the principal spot from the Sample solution corresponds to that from the Standard solution, as obtained in Organic Impurities.
2.3 C. Identification Tests—General, Sulfate 〈191〉
Sample solution: 20-mg/mL solution made with the aid of a few drops of hydrochloric acid
Acceptance criteria: Meets the requirements
3 ASSAY
3.1 Procedure
Sample solution: Dissolve 200 mg of Quinidine Sulfate in 20 mL of glacial acetic acid, heating if necessary, and cool the solution.
Titrimetric system
Mode: Titrimetry
Titrant: 0.1 N perchloric acid VS
Endpoint detection: Visual
Analysis: To the Sample solution add 20 mL of acetic anhydride and 4 drops of p-naphtholbenzein TS, and titrate with Titrant from a 10-mL microburet to a green endpoint. Perform a blank determination, and make any necessary corrections. Each mL of 0.1 N perchloric acid is equivalent to 24.90 mg of total alkaloid salt, calculated as quinidine sulfate [(C20H24N2O2)2 · H2SO4 · 2H2O].
Acceptance criteria: 99.0%–101.0% on the anhydrous basis
4 IMPURITIES
4.1 Residue on Ignition 〈281〉: NMT 0.1%
4.2 Organic Impurities
Standard solution A: 6 mg/mL of USP Quinidine Sulfate RS in diluted alcohol
Standard solution B: 0.06 mg/mL of USP Quinidine Sulfate RS in diluted alcohol from Standard solution A
Standard solution C: 0.05 mg/mL of USP Quininone RS (corresponding to 0.06 mg of the sulfate) and 0.10 mg/mL of cinchonine (corresponding to 0.12 mg of the sulfate) in diluted alcohol
Sample solution: 6 mg/mL of Quinidine Sulfate in diluted alcohol
Chromatographic system
(See Chromatography 〈621〉, Thin-Layer Chromatography.)
Mode: TLC
Adsorbent: 0.25-mm layer of chromatographic silica gel mixture
Application volume: 10 µL
Developing solvent system: Chloroform, acetone, and diethylamine (50:40:10)
Analysis
Samples: Standard solution A, Standard solution B, Standard solution C, and Sample solution
Proceed as directed in the chapter. The solvent chamber is used without previous equilibration. When the solvent front has moved 15 cm, remove the plate from the chamber, mark the solvent front, and allow the solvent to evaporate. Spray with glacial acetic acid. Locate the spots on the plate by examination under long-wavelength UV light. Then spray the plate with potassium iodoplatinate TS.
Acceptance criteria: Any spot produced by the Sample solution at the R value of a spot produced by Standard solution C is not greater in size or intensity than that corresponding spot. Apart from these spots and from the spots appearing at the R value of quinidine sulfate and dihydroquinidine sulfate (the two spots most evident from Standard solution A), any additional fluorescent spot is not greater in size or intensity than the principal spot of Standard solution B. After the treatment with potassium iodoplatinate TS, any spot produced by the Sample solution is not greater in size or intensity than a corresponding spot from Standard solution C.
4.3 Limit of Dihydroquinidine Sulfate
Solution A: Add 35.0 mL of methanesulfonic acid to 20.0 mL of glacial acetic acid, and dilute with water to 500 mL.
Solution B: Dilute 10.0 mL of diethylamine with water to 100 mL.
Mobile phase: Acetonitrile, Solution B, Solution A, and water (100:20:20:860). Adjust with diethylamine to a pH of 2.6.
System suitability solution: 0.2 mg/mL each of USP Quinidine Sulfate RS and dihydroquinidine hydrochloride prepared as follows. Dissolve a suitable quantity of each USP Quinidine Sulfate RS and dihydroquinidine hydrochloride in about 10% of total volume of methanol, and dilute with Mobile phase to volume.
Sample solution: 0.2 mg/mL of Quinidine Sulfate in Mobile phase
Chromatographic system
(See Chromatography 〈621〉, System Suitability.)
Mode: LC
Detector: UV 235 nm
Column: 3.9-mm × 30-cm; packing L1
Injection volume: 50 µL
System suitability
Sample: System suitability solution
[Note—The relative retention times for quinidine and dihydroquinidine are 1 and 1.5, respectively.]
Suitability requirements
Resolution: NLT 2.5 between quinidine and dihydroquinidine
Relative standard deviation: NMT 2.0%
Analysis
Sample: Sample solution
Acceptance criteria: 20.0%; the response of the dihydroquinidine peak is NMT 0.25 that of the quinidine peak.
5 SPECIFIC TESTS
5.1 Chloroform–Alcohol-Insoluble Substances
Sample solution: Warm 2 g of Quinidine Sulfate with 15 mL of chloroform and dehydrated alcohol mixture (2:1) at 50° for 10 min.
Analysis: Pass the Sample solution through a tared, sintered-glass filter, using gentle suction. Wash the filter with five 10-mL portions of the chloroform–alcohol mixture (2:1), dry at 105° for 1 h, and weigh.
Acceptance criteria: NMT 0.1%; the weight of the residue does not exceed 2 mg.
5.2 Optical Rotation, Specific Rotation 〈781S〉
Sample solution: 20 mg/mL of Quinidine Sulfate in 0.1 N hydrochloric acid
Acceptance criteria: +275° to +288° on the anhydrous basis
5.3 Water Determination, Method I 〈921〉: 4.0%–5.5%
6 ADDITIONAL REQUIREMENTS
Packaging and Storage: Preserve in well-closed, light-resistant containers.
USP Reference Standards 〈11〉
USP Quinidine Sulfate RS
USP Quininone RS
Cinchonan-9-one, 6′-methoxy-, (8-alpha)-.
C20H22N2O2 322.40


