QUALITY ATTRIBUTES OF TABLETS LABELED AS HAVING A FUNCTIONAL SCORE
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 PURPOSE
This chapter provides guidance for the development, scale up, and some post approval changes of tablets labeled as having a functional score. Specifically, this chapter provides quality attributes for tablet products labeled as functionally scored that can be split into multiple split portions to have an accurate fractional dose. The split portions should be a fractional part of the dose of the intact tablet based on the number of scores and the number of the split portion (e.g., one-half, one-quarter). At the time of splitting, the intact tablets should conform to the product specification. After splitting, with the exception of dose, each split portion from tablets with functional scoring is expected to conform to the quality attributes of the whole tablets. The split portions from tablets having a functional score are to conform to the product specification and the quality attributes of the whole tablets with equivalent strength. The split portions from a functionally scored tablet should conform to the tests described in this chapter.
2 SCOPE
This chapter applies to tablets with a functional score and to their split portions. Functionally scored tablets should be split as described in the procedure for splitting tablets. The storage conditions for the split portions should be defined in the test procedure. Analysts should use only split portions from tablets prepared following the procedure for Splitting Tablets with Functional Scoring for further analyses. These analyses include Loss of Weight, Uniformity of Dosage Units, Tablet Friability (if applicable), and Dissolution (or Disintegration, if applicable).
For further information, see FDA Guidance for Industry Tablet Scoring: Nomenclature, Labeling, and Data for Evaluation.
[NOTE-This chapter is not applicable for modified-release tablets, unless the tablet splitting has been demonstrated to not compromise the intended drug release profile.]
3 SPLITTING TABLETS WITH FUNCTIONAL SCORING
Both Sample Preparation 1 and Sample Preparation 2 should be performed. The resulting split portions should be used for the tests described in this chapter.
3.1 Sample Preparation 1 (splitting by hand)
1. Take a random sample of intact functionally scored tablets, per the number of dosage units as designated in Uniformity of Dosage Units (905), and proceed as follows.
2. Accurately weigh each tablet, and record its weight.
3. Split each tablet by hand into the designed number of split portions, and weigh each split portion.
4. Proceed to Loss of Weight and Uniformity of Dosage Units.
5. If the criteria in step 4 have been met, then proceed to Tablet Friability and Dissolution.
3.2 Sample Preparation 2 (splitting by tablet splitter)
1. Take a random sample of intact functionally scored tablets, per the number of dosage units as designated in (905), and proceed as follows.
2. Accurately weigh each tablet, and record its weight.
3. Split each tablet using a tablet splitter into the designed number of split portions, and weigh each split portion.
4. Proceed to Loss of Weight and Uniformity of Dosage Units.
5. If the criteria in step 4 have been met, then proceed to Tablet Friability and Dissolution.
[NOTE-Each split portion of an intact tablet is considered a unit dose.]
4 LOSS OF WEIGHT
The analyses should be performed on 15 tablets that are split by hand, and 15 tablets that are split with a tablet splitter, for tablets compressed at both the high and low ends of the proposed tablet hardness range.
For each individual whole tablet, weigh that tablet, split that tablet, physically combine all of the split portions from that tablet, and weigh the combined split portions from that individual whole tablet.
Calculate the difference in weight between each whole individual tablet and its combined split portions. The resultant difference in weight must be less than 3.0% of the weight of the whole individual tablet.
Report the difference as the percent of loss in weight for each whole individual tablet. Report each of the 15 tablets.
5 UNIFORMITY OF DOSAGE UNITS
Use split portions from tablets according to Splitting Tablets with Functional Scoring. Uniformity of dosage units must be demonstrated by either content uniformity or weight variation. See (905).
6 TABLET FRIABILITY
Use split portions of tablets that have been compressed both at the high and low ends of the proposed hardness range according to Splitting Tablets with Functional Scoring. See Tablet Friability (1216).
7 DISSOLUTION
Use split portions from tablets according to Splitting Tablets with Functional Scoring.
7.1 Immediate-Release Tablets
Dissolution for immediate-release tablets should start at the S, stage (see Dissolution (711)). Test 12 split tablet portions according to the specified Medium, Apparatus, Times, and Analysis in the test method.
Where appropriate, it may be applicable to apply dissolution profile comparison between the split portions and the intact tablet. (See Assessment of Solid Oral Drug Product Performance and Interchangeability, Bioavailability, Bioequivalence, and Dissolution (1090), and FDA Guidance for Industry Tablet Scoring: Nomenclature, Labeling, and Data for Evaluation).
7.2 Extended-Release Tablets
Perform dissolution testing of the intact tablets and the split tablet portions of extended-release tablets as specified below.
Procedure: See (711), Extended-Release Dosage Forms. Individually test 12 split tablet portions and 12 intact tablets that have been compressed at both the high and low ends of the proposed hardness range.
Medium, Apparatus, and Analysis: As given in the test method, following the appropriate test number found on the labeling. Dissolution profile test time points are determined as follows. From the appropriate dissolution test in the monograph, use the time points given. At a minimum, use 3 time points with no more than 1 time point where the results exceed 85% dissolved.
Calculate the similarity factor (f) for the intact-tablet results (R) and the split-tablet portion results (7), and apply the similarity assessment as described in (1090).
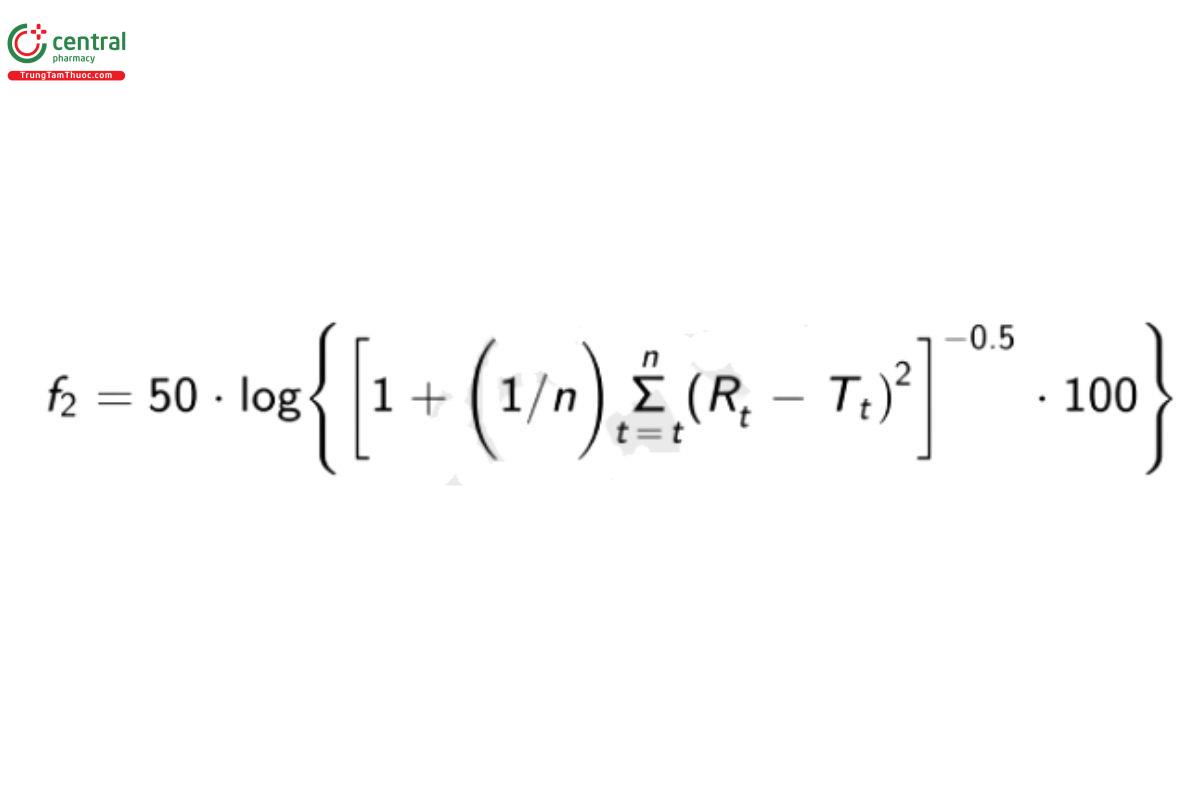
Rt = cumulative percentage of the labeled drug dissolved at each of the selected n time points of the intact tablets
Tt = cumulative percentage of the labeled drug dissolved at each of the selected n time points of the split tablet portions
Acceptance criteria: The calculated f2 is NLT 50 (acceptable range: 50-100) and the percentages of the labeled amount released of split tablet conform to the L2 level criteria of Acceptance Table 2 in (711).
8 DISINTEGRATION
Disintegration testing is necessary only when used as a surrogate for dissolution testing as specified in the monograph. Follow the procedure using split portions from tablets according to Splitting Tablets with Functional Scoring test as the dosage unit (see Disintegration (701))


