Pyrrolizidine Alkaloids as Contaminants
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
This informational general chapter is intended to provide an overview of pyrrolizidine alkaloids (PAs), heterocyclic organic compounds that are synthesized by many plant species (1–2) and can contaminate or be present in many plant-related products. The most potent PAs exhibit hepatotoxic, genotoxic, carcinogenic, or mutagenic activities. PAs can be found as contaminants in botanical drugs, herbal medicines, botanical dietary ingredients, and botanical dietary supplements, thereby posing a potentially serious threat to human health. PAs are derived from ornithine and are characterized by bicyclic pyrrolizidine moieties that are referred to as “necines” (3–4). PAs occur in two forms: pyrrolizidine alkaloids N-oxide (PANO) and tertiary bases (4–5). More than 660 PAs have been identified in approximately 6000 plant species (2) or in 3% of the world’s flowering plants (6–7). PAs are reported to be present mainly in the Boraginaceae (all genera), Asteraceae or Compositae (specifically in the tribes Senecioneae and Eupatorieae), and Leguminosae or Fabaceae (genus Crotalaria) families (5,8–9). PAs are mainly concentrated in the seeds and flowering parts of plants and, in lesser amounts, the leaves, stems, and roots (5). PAs of concern have been detected in dietary supplements (6); teas and herbal infusions (2); honey and pollen (4,10), predominantly due to the presence of PA-containing plants growing near plants of interest and being accidentally coharvested with plants used in these products. Because of the wide distribution of PAs in many plant species, dietary exposures to potent PAs are a safety concern (6). Published literature reports suggest that tea and herbal infusions were the main contributors to total unsaturated PA exposure. Therefore, efforts should be made to obtain information specifically on the sources of PA contamination in tea, such as the adventitious PA-containing plants responsible for the contamination, and to use measures to control infestation and coharvest (11).
2 PA CHEMISTRY
[Hình ảnh cấu trúc hóa học – Figure 1 tại đây]
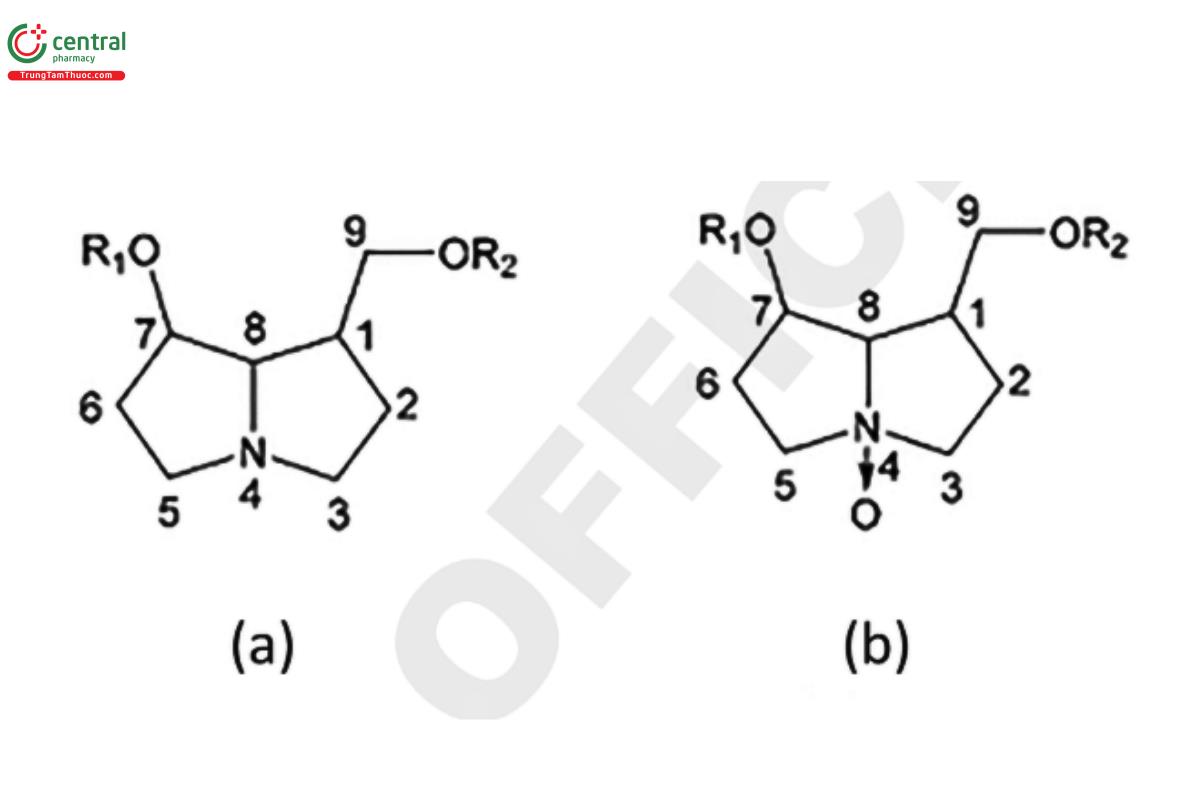
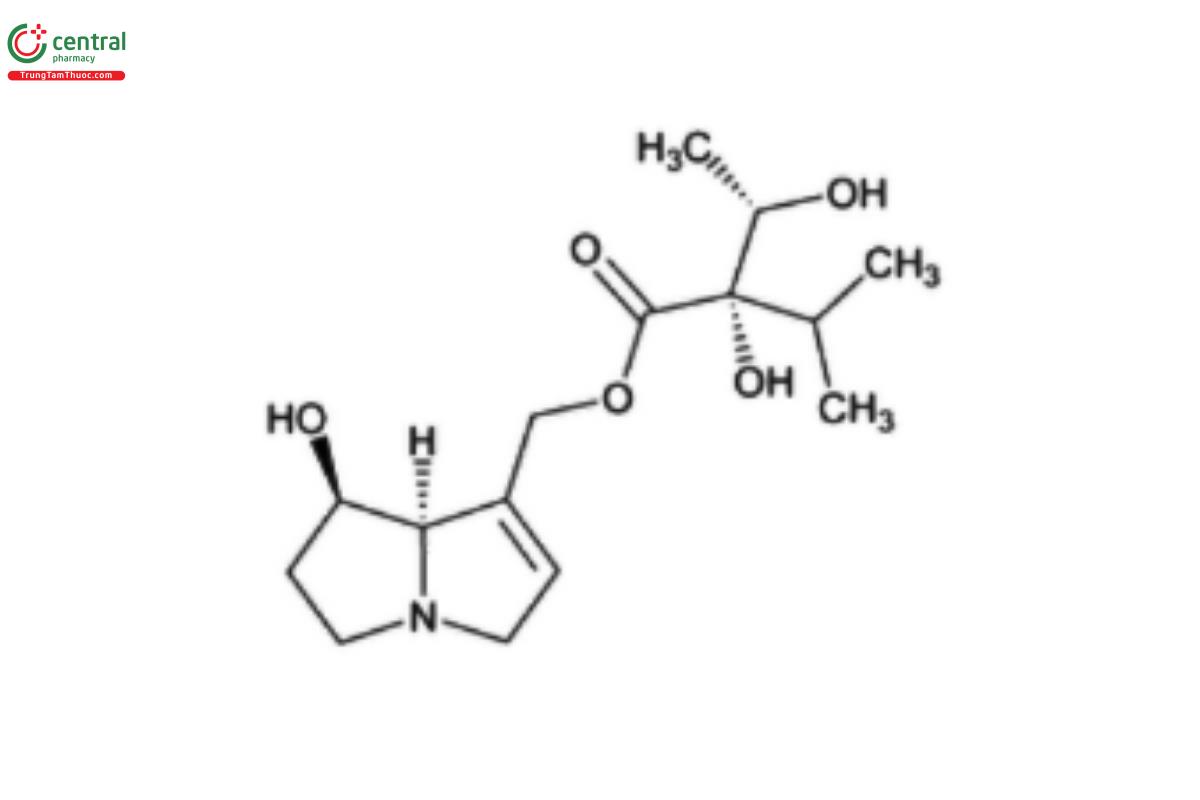
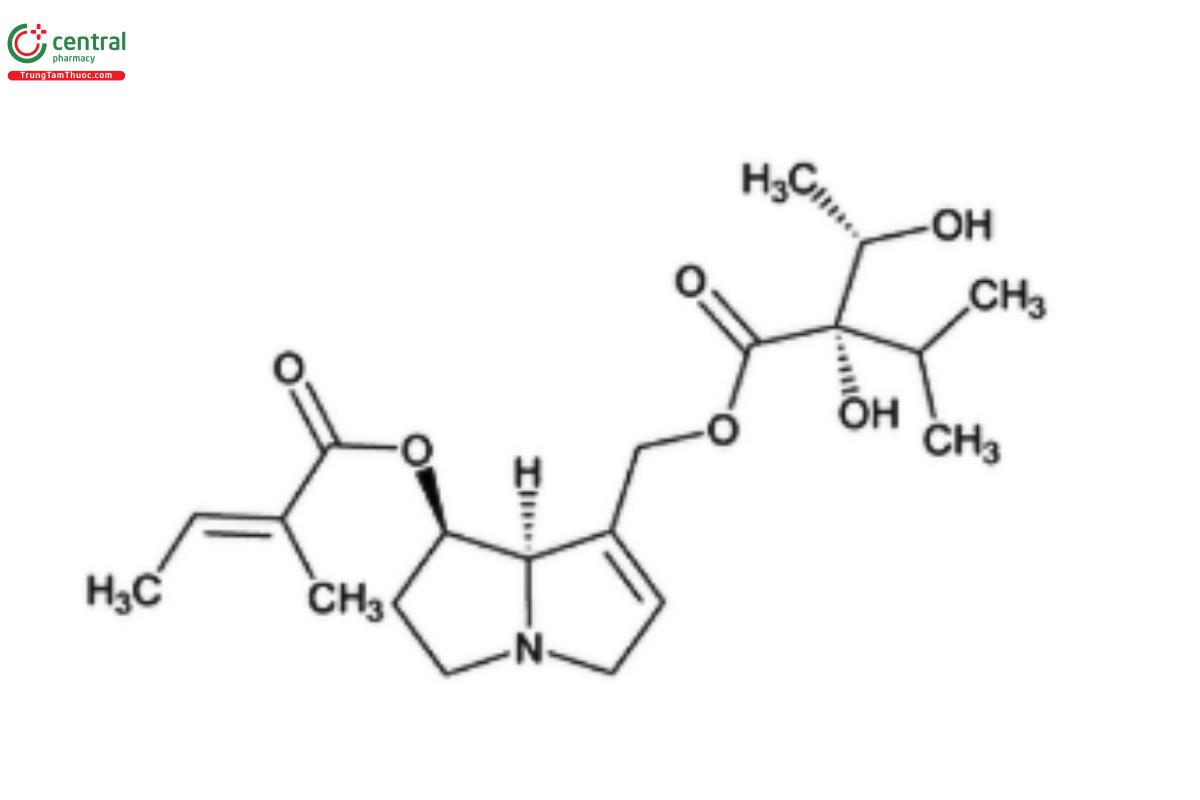
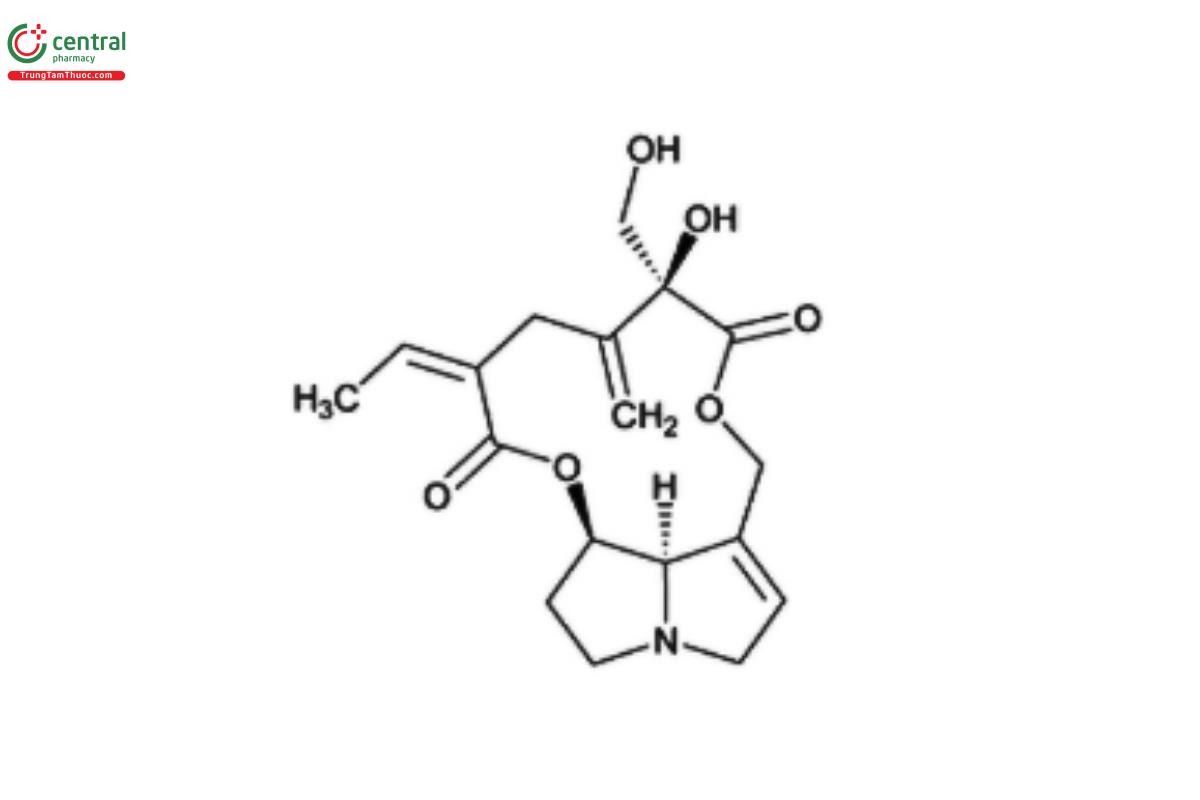
PAs are typically found as esters composed of a necine base and one or more necic acids (Figure 1). The necine base typically includes pyrrolizidine, a bicyclic aliphatic hydrocarbon comprised of two fused 5-membered rings with a nitrogen atom at position 4. The necine base usually has a hydroxymethyl group at C-1 and a hydroxyl group at C-7 (12) (when R₁ and R₂ are hydrogens in Figure 1[a]). Esterification with necic acids occurs at these hydroxyl groups forming monoester (Figure 2), open-chain diester (Figure 3), or macrocyclic diester alkaloids (Figure 4) (13). Necic acids are either mono- or dicarboxylic acids with branched carbon chains bearing hydroxy, epoxy, carboxy, acetoxy, methoxy, or other alkoxy groups as substituents (14). The high diversity in the structures of PAs is due in part to combinations of various necine bases and necic acids and the formation of monoesters at different positions (5). The diversity is further enhanced with their presence in plants as both tertiary bases and as N-oxides (PANO) Figure 1(b).
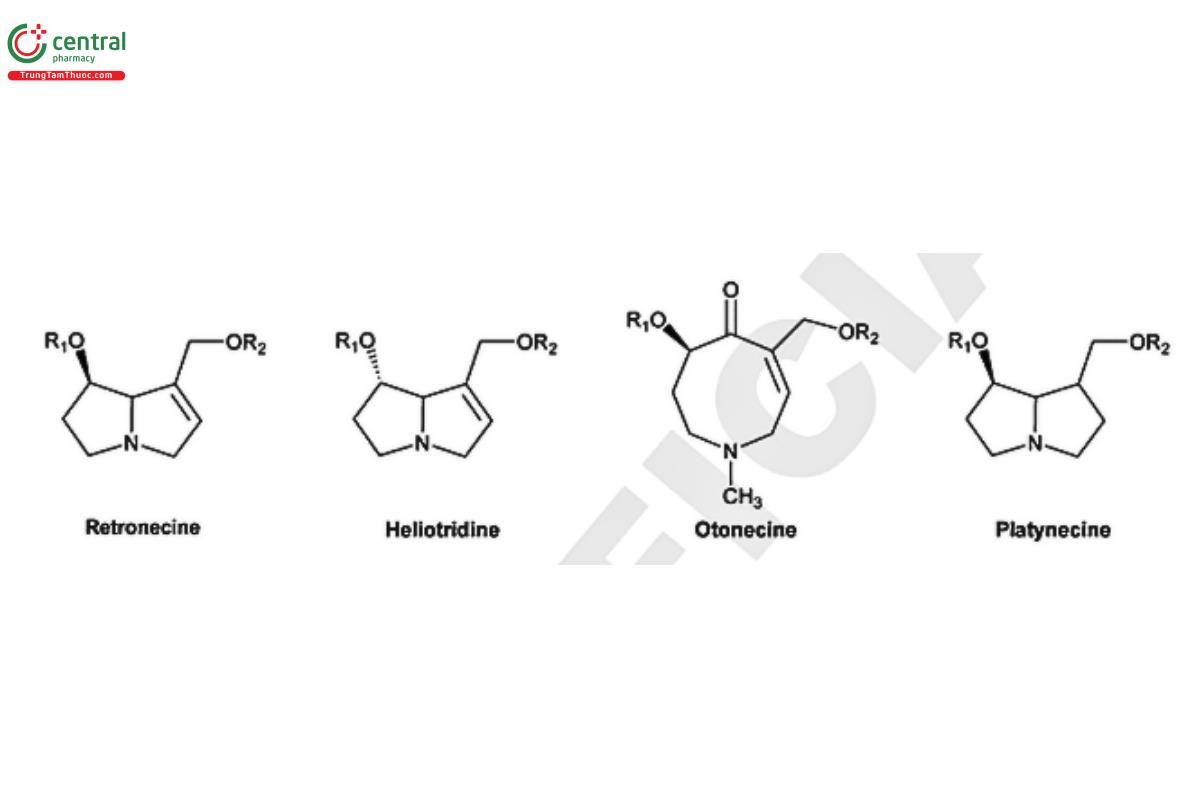
Based on the structure of necine base, PAs are further categorized into four groups: retronecine, heliotridine, otonecine, and platynecine types (Figure 5) (15). Retronecine, heliotridine and otonecine are unsaturated bases, i.e., they contain a double bond in the 1,2-position, whereas platynecine is a saturated base (16–17). Structurally, otonecine stands as the most distinct among all types because it is oxidized at C-8 and contains a monocyclic ring, thus deviating from other types that contain a bicyclic ring (18–19). Some esters of 1,2-unsaturated necine bases are toxic while esters with platynecine type are considered nontoxic PAs (14,20).
2.1 Structural Requirements for Toxicity
Not all PAs are toxic to humans at the levels found in herbal products. The toxic potential of a PA varies considerably with chemical structure (21). The structural requirements for PAs to exhibit toxicity are outlined below:
- The presence of a 1,2-double bond in the necine base (22). Cytochrome P-450 mediated metabolism of 1,2-unsaturated PAs forms reactive pyrroles, which can then form protein and DNA adducts (17,23). In contrast, the saturated structure of the platynecine-type PAs forms nonreactive polar compounds that are water soluble and readily excreted.
- The presence of one or two hydroxyl groups in the necine base.
- The esterification of one or two hydroxyl groups in the necine base and branching in at least one of the carboxylic acids (24). Due to this, cyclic diesters exert the highest toxicity, followed by open-chain diesters (intermediary), with monoesters having the lowest toxicity (25).
For the remainder of this paper, the term "PAs" will refer to more-potent and most-potent pyrrolizidine alkaloid molecules with the chemical characteristics described above, not all PAs.
2.2 Stability of PAs
It has been suggested that PAs, particularly their N-oxides, are heat sensitive, and thus, heat treatment could be utilized to control PA levels (26). Although high concentrations of some 1,2-unsaturated PAs and their N-oxides may occur in certain plants, much lower and variable concentrations of these substances are present in finished products due to processing (26). Drying plant materials is reported to cause significant decomposition of 1,2-unsaturated PAs, especially their N-oxides (27).
3 DIETARY EXPOSURE TO PAS
Seeds of PA-containing plants can contaminate grains, and prolonged consumption of such grains or grain products have resulted in rare but serious large-scale outbreaks of PA poisoning (pyrrolizidine alkaloidosis). One such example is a serious outbreak in 1974 that occurred in Afghanistan due to the consumption of bread made from wheat contaminated with seeds of Heliotropium sp. plants (28).
Lately there has been a growing interest in the use of botanical ingredients, leading to concern about the potential for PA poisoning primarily due to contamination from PA-containing plants in widely distributed botanical and herbal products. Recent literature reports have revealed the presence of PAs in herbal teas, predominantly senecionine-type, followed by lycopsamine- and heliotrine-types (29). Cross-contamination with plants containing PAs during the harvesting of herbal ingredients is responsible for the occurrence of PAs in ingredients of botanical drugs and botanical dietary supplements such as Matricaria chamomilla and Mentha × piperita L, which are not known to contain PAs (30–31).
Apart from exposure to PAs through herbal products, secondary contamination in foods of animal origin such as milk, eggs, honey, and pollen products was also observed.
Honey and supplement products containing bee pollen frequently contain PAs (5,11) such as senecionine, echimidine, lycopsamine and their N-oxides (32–33).
3.1 Measures to Avoid PA Contamination
Total eradication of PA-containing plants is not possible. It is necessary to identify potential risks associated with every step involved in the process chain, such as cultivation, harvesting, inspection of incoming goods, and drug processing, until the release of the botanical drug, botanical dietary ingredient, or botanical dietary supplement. For example, the risk of coharvesting PA-containing weeds during the harvest of the cultivated plant material can be controlled by optimization of timing, technology, cutting height, and other such factors associated with harvesting technology. Apart from coharvesting of PA-containing weeds, an alternate path of contamination that involves horizontal transfer and exchange of PA and/or PANO between living plants via the soil was recently discovered (34). To avoid PA contamination through this process, alkaloidal weeds have to be extracted completely from the soil and entirely removed from the fields rather than just leaving them to rot (34).
4 TOXICOLOGY
PAs are absorbed in the gastrointestinal tract and approximately 80% of PAs are excreted in the urine, feces, and breast milk (4). Because PAs are highly lipophilic, some are able to cross the placenta (4). The toxicity of potent PAs is mainly associated with their metabolic byproducts (2,4). PAs require metabolic activation to form highly reactive electrophiles (4,35). The liver is the main target organ of toxicity because the metabolic activation of PAs primarily occurs in the liver (4). Metabolic activation occurs through the oxidation of PAs, mostly catalyzed by the cytochrome P450 enzymes CYP3A and CYP2B, to form reactive pyrrolic esters. The hydrolysis of pyrrolic esters produces dihydropyrrolizines (DHP) (4,5). Liver toxicity results from the electrophilic pyrrolic esters and DHP, which further binds or forms adducts with vital cellular macromolecules such as proteins and DNA (5,36). The lungs are also a target organ of PA exposure as a result of reactive metabolites reaching the lungs through the systemic circulation (4–5,37). Experimental animal studies on potent PAs have reported hepatotoxicity, developmental toxicity, genotoxicity, and carcinogenicity (37). Both acute and chronic toxicity of PAs have been described and are characterized by different symptoms (2,4) as outlined below.
4.1 Acute and Subchronic Toxicity
Acute liver toxicity in humans and animals due to PA exposure is characterized by hepatic veno-occlusive disease (HVOD), which is considered a marker of PA toxicity (2,5,37–38). It is estimated that in 20% of acute PA poisoning cases humans die, 50% recover clinically, and 20% may develop chronic HVOD and cirrhosis years later (39). Acute PA poisoning symptoms include hemorrhagic necrosis, hepatomegaly, and ascites, while subacute liver effects present as hepatic sinusoidal obstruction syndrome (HSOS) due to blockage of the hepatic veins (4). HSOS is characterized by portal hypertension and liver dysfunction (40). Acute PA toxicity may also target the lungs (5,37,39) causing damage similar to HVOD, resulting in pulmonary hypertension and congestive heart failure (4,38–39). The lowest exposures of PAs associated with acute or short-term toxicity range from 0.8–3 mg/kg per day (5). However, based on available human and experimental data, even one-time exposure to unsaturated PAs may lead to liver cirrhosis; the level of exposure was not provided (41). Importantly, long-term exposure to much lower levels of PAs (15 µg/kg per day for 6 months) may cause chronic liver diseases including HVOD (5,35).
4.2 Chronic Toxicity
Animal studies have shown that long-term exposure to some PAs is associated with carcinogenicity, primarily in the liver (5), although other organs have also been rarely implicated. The occurrence of PA-associated tumors has also been reported in the lungs, bladder, skin, brain, spinal cord, pancreas, and gastrointestinal tract (4). Tumorigenic PAs belong to heliotridine-, retronecine- and otonecine-type structural classes. The DHPs formed by metabolism of these PAs form DNA adducts, which are thought to be the main mechanism responsible for tumorigenesis (4–5). Lasiocarpine, monocrotaline, and riddelliine are classified as possibly carcinogenic to humans (Category 2B) based on carcinogenic effects observed in rodent studies. Riddelliine has been shown to form DNA adducts in human liver microsomes similar to the DNA adducts observed in rodents, suggesting that PAs could be genotoxic and carcinogenic in humans (4).
5 PAS RECOMMENDED FOR MONITORING
Twenty-eight PAs, shown in Table 1, are selected for monitoring in botanical drugs, botanical dietary ingredients, and botanical supplements.
Table 1. List of PAs Selected for Monitoring
| PA | CAS number | PA | CAS number |
| Echimidine | 520-68-3 | Lycopsamine | 10285-07-1 |
| Echimidine-N-oxide | 41093-89-4 | Lycopsamine-N-oxide | 95462-15-0 |
| Erucifoline | 40158-95-0 | Monocrotaline | 315-22-0 |
| Erucifoline-N-oxide | 123864-94-8 | Monocrotaline-N-oxide | 35337-98-5 |
| Europine | 570-19-4 | Retrorsine | 480-54-6 |
| Europine-N-oxide | 65582-53-8 | Retrorsine-N-oxide | 15503-86-3 |
| Heliotrine | 303-33-3 | Seneciphylline | 480-81-9 |
| Heliotrine-N-oxide | 6209-65-0 | Seneciphylline-N-oxide | 38710-26-8 |
| Intermedine | 10285-06-0 | Senecionine | 130-01-8 |
| Intermedine-N-oxide | 95462-14-9 | Senecionine-N-oxide | 13268-67-2 |
| Jacobine | 6870-67-3 | Senecivernine | 72755-25-0 |
| Jacobine-N-oxide | 38710-25-7 | Senecivernine-N-oxide | 101687-28-9 |
| Lasiocarpine | 303-34-3 | Senkirkine | 2318-18-5 |
| Lasiocarpine-N-oxide | 127-30-0 | Trichodesmine | 548-90-3 |
6 REGULATORY AND PROPOSED TOLERABLE DAILY INTAKE LEVELS
The US current good manufacturing practice (cGMP) regulations for dietary supplements (21 CFR §111) require manufacturers to take certain measures to prevent contamination in the manufacturing process. The cGMP regulations do not specifically require testing for PAs, but manufacturers are required to set limits on the types of contamination that may adulterate or may lead to adulteration of the product within the meaning of the Federal Food, Drug and Cosmetic Act (FFDCA). Manufacturers also generally have a responsibility to ensure that their dietary supplement products do not present a significant or unreasonable risk of illness or injury that would cause the FDA to deem the product adulterated under §402(f)(1)(A) of the FFDCA. Additionally, under §402(a)(1) of the FFDCA, certain foods, including dietary supplements, are deemed adulterated if they contain poisonous or deleterious substances. Currently, there are no required FDA limits for PAs. In the absence of required FDA limits, the Division of Risk and Decision Analysis evaluates the safety of contaminants in food on a case-by-case basis using a safety assessment (SA) approach. The SA considers exposure and toxicity of the contaminant in the food product. Exposure is estimated using the contaminant level reported in the food and consumption or recommended intake amounts for specific subpopulations.
Regulations governing levels of PAs in food and food supplements have been established in several countries based on the potential exposure to contaminants from the increased use of herbal medicines leading to PA intoxication as a public health concern (4,42). The range of tolerable daily intake levels for PAs provided in Table 2 may provide some guidance to manufacturers on the highly potent PAs (e.g., cyclic diesters).
Table 2. Proposed Tolerable Daily Intake Levels for PAs
| Regulatory Authority | Tolerable Daily Intake | Reference |
| German Federal Department of Health Bundesanzeiger (1992) | ≤1 µg/day (maximum 6 weeks use per year) ≤0.1 µg/day (for prolonged use) | (4,42,43) |
| US FDA (2001) | Ban on PA-containing comfrey supplements; no tolerable intake level established | (42,45) |
| Netherlands (2001) | ≤1 µg/kg or ≤1 µg/L PA concentration in final product (herbal preparation or plant extract) | (4,42,46) |
| Food Standards Australia New Zealand (FSANZ) (2001) | ≤1 µg/kg/day (provisional) based on veno-occlusive disease (cancer risk considered not proven in humans) | (41,47) |
| EFSA (2011) | MOE of 10,000 or higher than BMDL10 of 70 µg/kg/day considered a “low concern from a public health point of view” (based on NTP carcinogenicity data) | (5) |
| BfR (2011) | 0.007 µg/kg/day | (48) |
| JECFA (2015) | MOE of 10,000 or higher than BMDL10 of 182 µg/kg/day (based on NTP carcinogenicity data) | (49) |
| EFSA (2017) | MOE of 10,000 or higher than BMDL10 of 237 µg/kg/day considered a “low concern from a public health point of view” (based on updated BMD modeling approach and NTP carcinogenicity data) | (37) |
| European Commission | 400 µg/kg in food supplements and 500 µg/kg in pollen-based food supplements | (50) |
| EMA (2020) | Accepted limit of 1.0 µg/day with no temporary restrictions | (44) |
7 RELATIVE POTENCIES OF PAS
The risk-based decisions for tolerable daily intake levels presented above are based on approaches that assume all PAs are equally potent, i.e., as potent as the highly toxic PAs such as lasiocarpine (5) or riddelliine (49). There is another risk-based approach based on relative risk factors (RRF) that may be appropriate. The RRF approach may be applicable because large structure-dependent differences have been documented between subgroups of unsaturated PA congeners with respect to their genotoxicity and cytotoxicity (51). The concept of relative potency (REP) was developed to address cases where a group of compounds share the same major mode of action, but individual members exhibit extraordinarily different potencies sometimes spanning several orders of magnitude. REP factors represent the potency of the congener relative to the most-toxic congener(s). The latter is assigned an REP factor of 1.0, and then all other congeners have REP factors of ≤1.0. For a given sample (e.g., a mixture of PAs) the sum potency can be derived by multiplying the amount of the specific congeners and their respective REP factors, which are then summed up for all congeners present in the sample. This REP concept has been successfully applied in evaluating the toxicology of polychlorinated dioxins, furans, and PCBs (52), and has also been suggested for phototoxic furanocoumarins (53). In an attempt to factor in the varied PA potencies in risk assessment, efforts have been made to derive PA potency factors relative to riddelliine. REP factors for PAs can be derived based on an approach that correlated fundamental structural differences between PAs and acute toxicity data from rodents, cytotoxicity data in mammalian cell culture, and genotoxicity data in Drosophila. Cyclic PA diesters are markedly more toxic than open-chain diesters followed by monoesters, consistent with experimental data. Generally, PA monoesters and open diesters showed lower toxicity than cyclic diesters. However, some PAs such as lasiocarpine and lasiocarpine N-oxide (PA monoesters) had the same REP factors as monocrotaline, riddelliine, and retrorsine (cyclic diesters, most potent) (54).
Manufacturers of dietary supplements containing PAs should aim at minimizing the presence of PAs in their products.
8 ANALYTICAL METHODS AND CHALLENGES
A wide range of analytical techniques to quantitate PAs and PANOs—such as chromatography, nuclear magnetic resonance (NMR), enzyme-linked immunosorbent assay (ELISA), capillary electrophoresis (CE), droplet countercurrent chromatography (DCCC), and miscellaneous electrokinetic chromatography (MEKC) methods—have been reported in the literature (55–57). However, selective and sensitive analytical methods are needed to meet the limits set by different regulatory bodies as the methods cited above should be validated for the different matrices at the analytical range required to establish the level of PA contaminants. Chromatographic separation methods, particularly high- or ultra-high performance liquid chromatography (HPLC, UHPLC) and gas chromatography (GC), are preferred to quantitate PAs. Detection methods including ultraviolet (UV), flame ionization (FI), evaporative light scattering (ELS), and refractive index (RI) could not achieve the required limit of detection (LoD) and limit of quantitation (LoQ). Mass spectrometric techniques offer improved detection limits and provide the advantage of mass selectivity via detectors such as quadrupole, triple quadrupole, time of flight (ToF), and orbital ion-trap along with various ionization techniques. It is not feasible to quantitate PANOs using GC without prior derivatization or reduction due to their instability at high temperatures. Further, the extraction and clean-up procedures associated with GC methods are time consuming and require very large volumes of solvent. As a result, high-performance liquid chromatography–tandem mass spectrometry (HPLC–MS/MS) has become the method of choice because of the extraction methods, which are more appropriate for the matrix, and the capability to simultaneously analyze PANOs along with PAs.
The most preferred analytical technique is LC–MS/MS, but this also has limitations. A baseline chromatographic separation is not always achieved for a few PA isomers such as intermedine/lycopsamine or senecionine/senecivernine, as well as their respective N-oxides (37). Also, because these isomers have the same molecular weight, it is not possible to distinguish them by mass spectrometry. An accurate quantification of individual PAs is not always possible for the above reasons. This necessitates the use of multiple methods for the accurate quantification of isomeric PAs. Furthermore, coelution of indicine could happen with the intermedine/lycopsamine pair (58). Similarly, coelution of indicine N-oxide has been observed with N-oxides of the intermedine/lycopsamine pair.
There is a need for highly sensitive and selective analytical methods to provide the level of quantification required to control contaminant PAs in botanical drugs, botanical dietary ingredients, and botanical dietary supplements. Apart from the above-mentioned challenges associated with analytical methods for the quantitation of PAs, there are challenges related to the reference standards: most of them are not commercially available, are very expensive to procure, are difficult to synthesize or isolate, and are not stable.
9 CONCLUSIONS
PAs are naturally occurring heterocyclic phytotoxins that have been reported to be hepatotoxic, mutagenic, and carcinogenic. PAs have been detected in various dietary sources such as grains, herbal teas, honey, milk, and eggs. We included a list of PAs to be monitored in Table 1. Manufacturers should evaluate the levels of PAs in their product considering their potential presence arising from the plant itself, from contamination with external PAs (e.g., due to coharvesting, absorbed from soil), and/or from supply chain vulnerabilities that may increase the risk of contamination of ingredients. Manufacturers should use a risk-based approach for determining appropriate limits and testing requirements, which should take into consideration that select PAs are more toxic and their levels should be minimized. The range of tolerable daily intake levels for PAs provided in Table 2 may provide some guidance to manufacturers for the highly potent PAs (e.g., cyclic diesters). Extraction and analytical quantitation of PAs is associated with several challenges and would benefit from evolving strategies for both efficient extraction and advanced selective analytical quantification techniques. Future developments will hopefully lead to a greater availability of reference standards and method validation as regulatory bodies create requirements for accurate quantitation of PAs.
10 REFERENCES
- Chen L, et al. Risk assessment for pyrrolizidine alkaloids detected in (herbal) teas and plant food supplements. Regulatory Toxicology and Pharmacology, 2017. 86:292-302.
- Habs M, et al. A Balanced Risk-Benefit Analysis to Determine Human Risks Associated with Pyrrolizidine Alkaloids (PA)-The Case of Tea and Herbal Infusions. Nutrients, 2017. 9(7).
- Roeder E, Wiedenfeld H, and Edgar JA. Pyrrolizidine alkaloids in medicinal plants from North America. Pharmazie, 2015. 70(6):357-67
- Moreira R, et al. Pyrrolizidine Alkaloids: Chemistry, Pharmacology, Toxicology and Food Safety. International Journal of Molecular Sciences, 2018. 19(6):1668.
- EFSA. Scientific Opinion on Pyrrolizidine alkaloids in food and feed. EFSA Panel on Contaminants in the Food Chain (CONTAM). EFSA Journal, 2011. 9(11):2406.
- Fu PP. Pyrrolizidine alkaloids-Tumorigenic components in Chinese herbal medicines and dietary supplements. Journal of Food and Drug Analysis, 2002. 10(4):198-211.
- Smith LW and Culvenor CCJ. Plant Sources of Hepatotoxic Pyrrolizidine Alkaloids. Journal of Natural Products, 1981. 44(2):129-152.
- Chen T, Mei N, and Fu PP. Genotoxicity of pyrrolizidine alkaloids. Journal of Applied Toxicology, 2010. 30(3):183-196.
- WHO. Pyrrolizidine Alkaloids. Environmental Health Criteria 80. International Programme on Chemical Safety. World Health Organization. 1998; Available from: http://www.inchem.org/documents/ehc/ehc/ehc080.htm.
- Kempf, M, et al. Pyrrolizidine alkaloids in pollen and pollen products. Molecular Nutrition & Food Research, 2010. 54(2):292-300.
- Dietary exposure assessment to pyrrolizidine alkaloids in the European population. EFSA Journal, 2016. 14(8):4572.
- Hartmann T and Witte L. Chemistry, biology and chemoecology of the pyrrolizidine alkaloids. Alkaloids Chem Biol Perspect, 1995. 9:155-233.
- Xia Q, et al. Formation of DHP-derived DNA adducts from metabolic activation of the prototype heliotridine-type pyrrolizidine alkaloid, heliotrine. Toxicol Lett, 2008. 178(2):77-82.
- Committee on Herbal Medicinal Products, European Medicines Agency. Public statement on the use of herbal medicinal products containing toxic, unsaturated pyrrolizidine alkaloids (PAs). EMA/HMPC, Editor. 2011.
- Gottschalk C, Ronczka S, Preiss-Weigert A, Ostertag J, Klaffke H, Schafft H, Lahrssen-Wiederholt M. Pyrrolizidine alkaloids in natural and experimental grass silages and implications for feed safety. Anim Feed Sci Technol, 2015. 207:253-261.
- Zhou Y, et al. A new approach for simultaneous screening and quantification of toxic pyrrolizidine alkaloids in some potential pyrrolizidine alkaloid-containing plants by using ultra performance liquid chromatography-tandem quadrupole mass spectrometry. Anal Chim Acta, 2010. 681(1-2):33-40.
- Wang YP, Fu PP, and Chou MW. Metabolic activation of the tumorigenic pyrrolizidine alkaloid, retrorsine, leading to DNA adduct formation in vivo. Int J Environ Res Public Health, 2005. 2(1):74-9.
- Chen T, Mei N, and Fu PP. Genotoxicity of pyrrolizidine alkaloids. J Appl Toxicol, 2010. 30(3):183-96.
- Rowell-Rahier MWL, Ehmke A, Hartmann T, Pasteels JM. Sequestration of plant pyrrolizidine alkaloids by chrysomelid beetles and selective transfer into the defensive secretions. Chemoecology, 1991. 2:41-48.
- Dreger MSM, Krajewska-Patan A, Mielcarek S, Mikołajczak PŁ, Buchwald W. Pyrrolizidine alkaloids-chemistry, biosynthesis, pathway, toxicity, safety and perspectives of medicinal usage. Journal Herba Polonica, 2009. 55(4):127-147.
- Lester C, et al. Intrinsic relative potency of a series of pyrrolizidine alkaloids characterized by rate and extent of metabolism. Food Chem Toxicol. 2019. 131:110523.
- Cheeke PR. Toxicity and metabolism of pyrrolizidine alkaloids. J Anim Sci, 1988. 66(9):2343-50.
- Fu PP, et al. Pyrrolizidine alkaloids-genotoxicity, metabolism enzymes, metabolic activation, and mechanisms. Drug Metab Rev, 2004. 36(1):1-55.
- Wiedenfeld H. Toxicity of pyrrolizidine alkaloids-a serious health problem. Journal of Marmara University Institute of Health Sciences, 2011. 1(2):79-87.
- Luckert C, et al. Disturbance of gene expression in primary human hepatocytes by hepatotoxic pyrrolizidine alkaloids: A whole genome transcriptome analysis. Toxicol In Vitro, 2015. 29(7):1669-82.
- World Health Organization. Safety evaluation of certain food additives and contaminants: supplement 2: pyrrolizidine alkaloids, prepared by the 18th meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 2020.
- Bull LB, Culvenor CCJ, and Dick AT. The pyrrolizidine alkaloids: their chemistry, pathogenicity and other biological properties. 1968(9).
- Mohabbat O, et al. An outbreak of hepatic veno-occlusive disease in north-western Afghanistan. Lancet, 1976. 2(7980):269-71.
- Mulder PPJ, López Sánchez P, These A, Preiss-Weigert A, and Castellari M. Occurrence of Pyrrolizidine Alkaloids in food. EFSA Supporting Publication 2015:EN-859, 2015. 12(8):114.
- Schulz M, et al. Detection of pyrrolizidine alkaloids in German licensed herbal medicinal teas. Phytomedicine, 2015. 22(6):648-56.
- Bodi D, et al. Determination of pyrrolizidine alkaloids in tea, herbal drugs and honey. Food Addit Contam Part A Chem Anal Control Expo Risk Assess, 2014. 31(11):1886-95.
- Boppre M, Colegate SM, and Edgar JA. Pyrrolizidine alkaloids of Echium vulgare honey found in pure pollen. J Agric Food Chem, 2005. 53(3):594-600.
- Orantes-Bermejo FJ, et al. Pyrrolizidine alkaloids: their occurrence in Spanish honey collected from purple viper's bugloss (Echium spp.). Food Addit Contam Part A Chem Anal Control Expo Risk Assess, 2013. 30(10):1799-806.
- Selmar D, et al. Transfer of pyrrolizidine alkaloids between living plants: A disregarded source of contaminations. Environ Pollut, 2019. 248:456-461.
- Schrenk D, et al. Pyrrolizidine alkaloids in food and phytomedicine: Occurrence, exposure, toxicity, mechanisms, and risk assessment-A review. Food and Chemical Toxicology, 2020. 136:111107.
- Lin G, et al. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. Journal of Hepatology, 2011. 54(4):666-673.
- EFSA. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements. EFSA Journal, European Food Safety Authority Panel on Contaminants in the Food Chain (CONTAM). 2017. 15(7):4908.
- Barnes JM, Magee PN, and Schoental R. Lesions in the lungs and livers of rats poisoned with the pyrrolizidine alkaloid fulvine and its N-oxide. Journal of Pathology and Bacteriology, 1964. 88:521-31.
- Prakash AS, et al. Pyrrolizidine alkaloids in human diet. Mutation Research-Fundamental and Molecular Mechanisms of Mutagenesis, 1999. 443(1-2):53-67.
- Barcelos STA, et al. Sinusoidal obstruction syndrome secondary the intake of Senecio brasiliensis: A case report. Ann Hepatol, 2019.
- EMA. Public statement on the use of herbal medicinal products containing toxic, unsaturated pyrrolizidine alkaloids (PAs). European Medicines Agency (EMA). Committee on Herbal Medicinal Products (HMPC). EMA/HMPC/893108/2011. Adopted on November 24, 2014 [cited July 24, 2020]; Available from: https://www.ema.europa.eu/en/use-herbal-medicinal-products-containing-toxic-unsaturated-pyrrolizidine-alkaloids-pas.
- Wiedenfeld H. Toxicity of pyrrolizidine alkaloids-a serious health problem. Clinical and Experimental Health Sciences, 2011. 1(2):79.
- Dusemund, et al. Risk assessment of pyrrolizidine alkaloids in food of plant and animal origin. Food and Chemical Toxicology, 2018. 115:63-72.
- EMA. Public statement on the use of herbal medicinal products containing toxic, unsaturated pyrrolizidine alkaloids (PAs) including recommendations regarding contamination of herbal medicinal products with pyrrolizidine alkaloids. European Medicines Agency (EMA). Committee on Herbal Medicinal Products (HMPC). EMA/HMPC/893108/2011 Rev. 1. July 8, 2020. (Draft document open for consultation) [cited November 11, 2020]; Available from: https://www.ema.europa.eu/en/documents/public-statement/draft-public-statement-use-herbal-medicinal-products-containing-toxic-unsaturated-pyrrolizidine en-0.pdf.
- FDA. FDA Advises Dietary Supplement Manufacturers to Remove Comfrey Products From the Market. Safety Alerts & Advisories. Issued July 6, 2001 [cited July 23, 2020]; US Food and Drug Administration. Available from: http://wayback.archive-it.org/7993/20171114115012/https://www.fda.gov/Food/Recalls Outbreaks Emergencies/SafetyAlerts Advisories/ucm111219.htm.
- Netherlands, O.G.o.t.K.o.t. Decree of 19 January 2001, establishing the Commodities Act Decree on Herbal Preparations (Translated). Ministry of Health, Wellbeing and Sports, 2001 [cited July 23, 2020 ]; Available from: https://zoek.officielebekendmakingen.nl/stb-2001-56.
- Australia New Zealand Food Authority (ANZF) Pyrrolizidine Alkaloids In Food. A Toxicological Review and Risk Assessment. Technical Report Series No. 2. 2001 [cited July 23, 2020]; Available from: https://www.foodstandards.gov.au/publications/documents/TR2.pdf.
- German Federal Institute for Risk Assessment (BfR). Chemical analysis and toxicity of pyrrolizidine alkaloids and assessment of the health risks posed by their occurrence in honey. Germany, 2011; Available from: https://www.bfr.bund.de/cm/349/chemical-analysis-and-toxicity-of-pyrrolizidine-alkaloids-and-assessment-of-the-health-risks-posed-by-their-occurence-in-honey.pdf.
- JECFA. World Health Organization: Joint FAO/WHO Expert Committee on Food Additives (JECFA). Eightieth meeting Rome, June 16-25, 2015. Summary and Conclusions. Issued 6 July 2015. JECFA/80/SC. 2015 [cited July 24, 2020]; Available from: https://www.fao.org/3/ax942e/ax942e.pdf.
- IASDA. IADSA Newsflash. February 2020 [cited July 24, 2020]; International Alliance of Dietary/Food Supplement Associations. Available from: https://ahpa.gomembers.com/Portals/0/PDFs/IADSA%20Newsflash%2011%202020-FF.pdf.
- Merz KH and Schrenk D. Interim relative potency factors for the toxicological risk assessment of pyrrolizidine alkaloids in food and herbal medicines. Toxicol Lett, 2016. 263:44-57.
- Van den Berg M, et al. The 2005 World Health Organization reevaluation of human and mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicological sciences, 2006. 93(2):223-241.
- Raquet N and Schrenk D. Application of the equivalency factor concept to the phototoxicity and genotoxicity of furocoumarin mixtures. Food and chemical toxicology, 2014. 68:257-266.
- Allemang A, et al. Relative potency of fifteen pyrrolizidine alkaloids to induce DNA damage as measured by micronucleus induction in HepaRG human liver cells. Food and chemical toxicology, 2018. 121:72-81.
- Crews C, Berthiller F, and Krska R. Update on analytical methods for toxic pyrrolizidine alkaloids. Anal Bioanal Chem, 2010. 396(1):327-38.
- Monteiro-Silva F, Gonzalez-Aguilar G. Evolution through time of Pyrrolizidine Alkaloids detection and quantification. Biomolecules, Switzerland. 2013.
- Kopp T, Abdel-Tawab M, and Mizaikoff B. Extracting and Analyzing Pyrrolizidine Alkaloids in Medicinal Plants: A Review. Toxins (Basel), 2020. 12(5).
- Mulder PPJ, López Sánchez P, These A, Preiss-Weigert A, and Castellari M. Occurrence of Pyrrolizidine Alkaloids in food. EFSA Supporting Publication 2015:EN-859, 2015. 12(8):114.
(USP 1-Dec-2023)


