Oxygen Flask Combustion
If you find any inaccurate information, please let us know by providing your feedback here

This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
The oxygen ask combustion procedure is provided as the preparatory step in the determination of bromine, chlorine, iodine, Selenium, and sulfur in some Pharmacopeial articles. Combustion of the material under test (usually organic) yields water-soluble inorganic products, which are analyzed for specific elements as directed in the individual monograph or general chapter.
The caution statement given for Procedure covers minimum safety precautions only, and serves to emphasize the need for exceptional care throughout.
Change to read:
1 APPARATUS
The apparatus (USP 1-Aug-2024) consists of a heavy-walled conical, deeply lipped or cupped 500-mL flask (unless a larger flask is specified), fitted with a ground-glass stopper to which is fused a test specimen carrier consisting of heavy-gauge platinum wire and a piece of welded platinum gauze measuring about 1.5 cm × 2 cm.
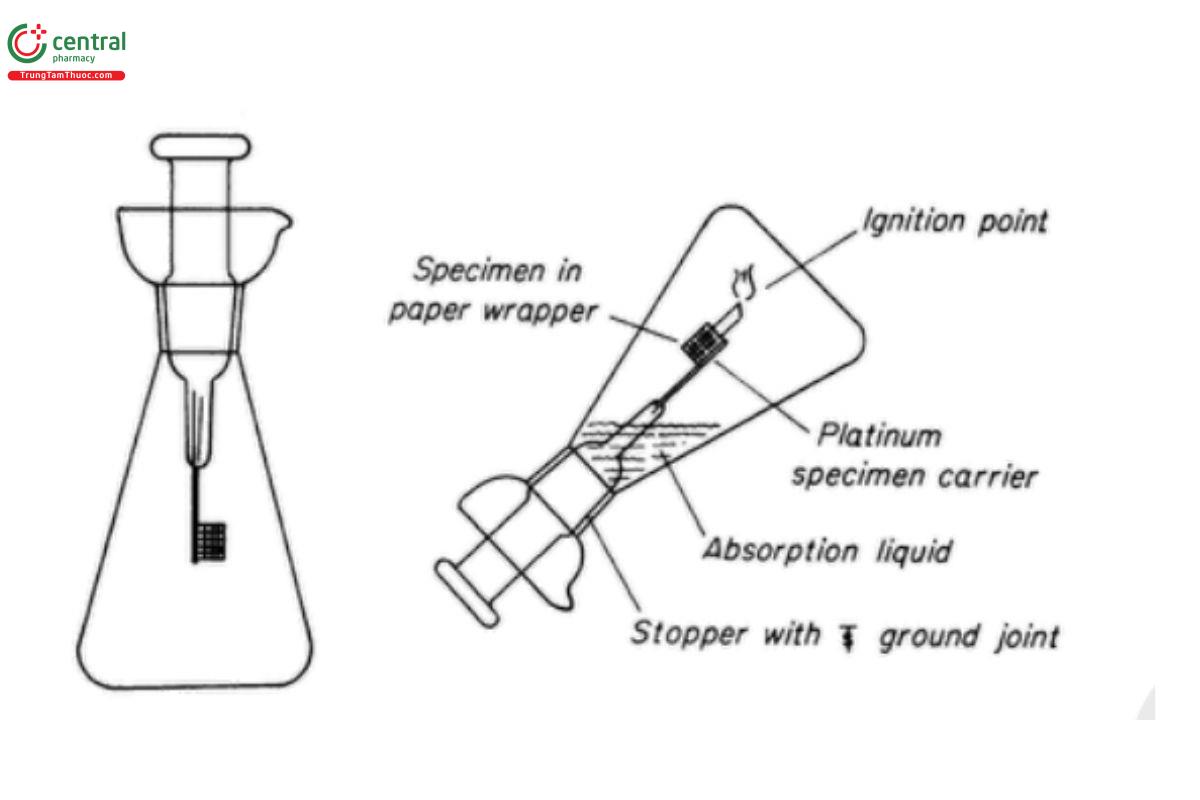
Change to read:
2 PROCEDURE
[Caution—Wear safety glasses and use a suitable safety shield between yourself and the apparatus and/or perform this test in a well- ventilated hood. (USP 1-Aug-2024) Exercise care to ensure that the flask is scrupulously clean and free from even traces of organic solvents.]
Weigh the substance, if a solid, on a piece of halide-free filter paper measuring about 4 cm square, and fold the paper to enclose it. Liquid substances are weighed in tared (USP 1-Aug-2024) gelatin capsules. (USP 1-Aug-2024) [Note—Gelatin capsules may contain signi cant amounts of combined halide or sulfur; therefore, (USP 1-Aug-2024) perform a blank determination, and make any necessary correction.]
Place the specimen, together with a filter paper fuse-strip, in the platinum gauze specimen holder. Place the absorbing liquid specified in the individual monograph or general chapter in the flask, moisten the joint of the stopper with water, and flush the air from the flask with a stream of rapidly flowing oxygen, swirling the liquid to favor its taking up oxygen. [Note—Saturation of the liquid with oxygen is essential for the successful performance of the combustion procedure.] Ignite the fuse-strip by suitable means. If the strip is ignited outside the flask, immediately plunge the specimen holder into the flask, invert the flask so that the absorption solution makes a seal around the stopper, and hold the stopper firmly in place. If the ignition is carried out in a closed system, the inversion of the flask may be omitted. After combustion is complete, shake the flask vigorously, and allow to stand for not less than 10 min with intermittent shaking. Then proceed as directed in the individual monograph or general chapter.


