OPHTHALMIC PRODUCTS-QUALITY TESTS
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
Change to read:
1 INTRODUCTION
Ophthalmic products are sterile products that are intended for application to any ocular structure, including any space adjacent to an ocular structure and its immediate surrounding spaces.
The routes of administration of ophthalmic products fall into three general categories: topical, intraocular injections, and extraocular injections. Topical drug products are intended to be administered to an ocular surface component, such as the eyelid, conjunctiva, or cornea, and can produce local or systemic effects. Intraocular and extraocular injections are administered through external boundary tissue. Due to the potential sensitivity of some ocular tissues, injections have additional requirements for subvisible particulate matter content (see Particulate and Foreign Matter, Subvisible Particulate Matter in Intraocular Solutions (789) (CN 1-May-2024), and Visible Particulates in Injections (790)). The ophthalmic routes of administration include, but are not limited to: topical, subconjunctival, sub-Tenons, subretinal, subchoroidal, intracorneal, intrascleral, suprachoroidal, intravitreal, intracameral, juxtascleral, and retrobulbar routes (see Figure 1). Ophthalmic products are administered to the eye in a wide variety of dosage forms, including but not restricted to: solutions, suspensions, ointments, gels, emulsions, strips, injections, inserts, and implants.
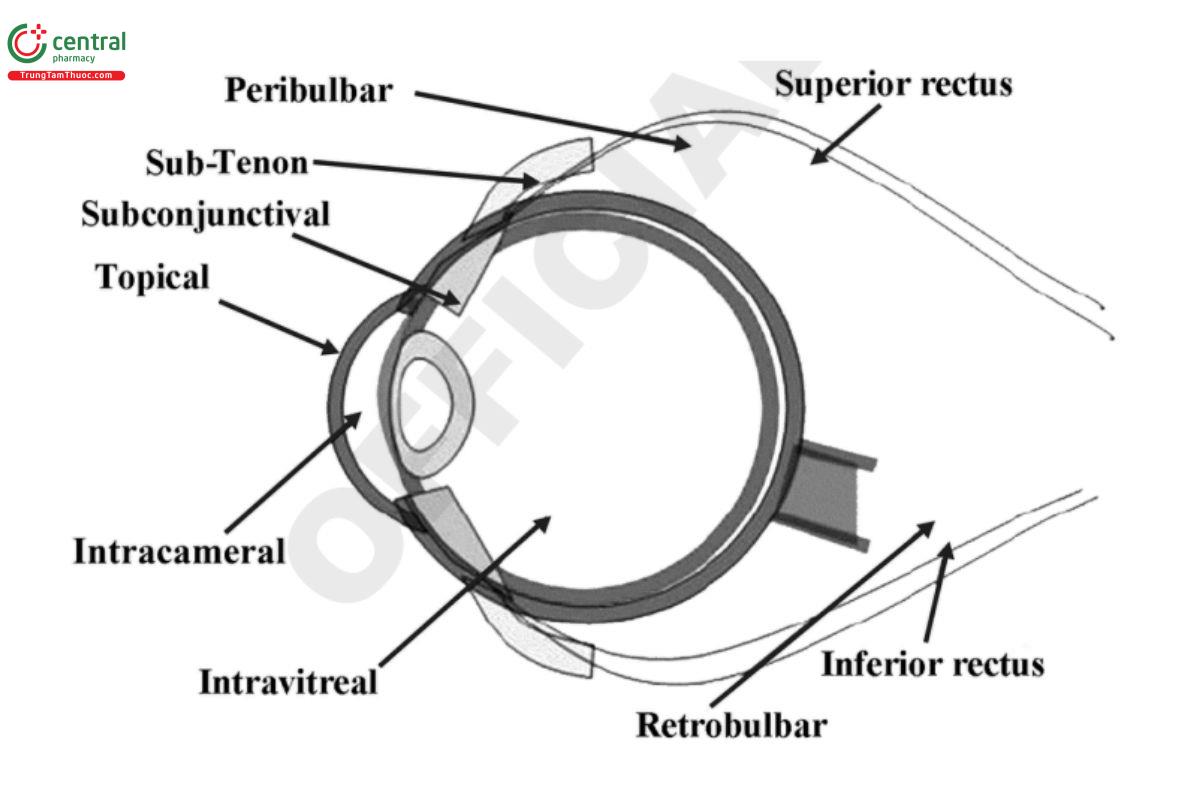
This chapter provides lists of consolidated common product quality test requirements in a concise and coherent fashion. This chapter applies, in part or in its entirety, when referenced in a drug product monograph (see General Notices, 3.10 Applicability of Standards). This chapter includes the quality tests for the specific route of administration. The quality tests listed can be used, as appropriate, by manufacturers toward the development of new drug product monographs for submission to the USP-NF.
2 OPHTHALMIC DOSAGE FORMS
This chapter discusses the specific characteristics of dosage forms that are administered to any structure in the eye. See Pharmaceutical Dosage Forms (1151) for additional information pertaining to the description and manufacturing of pharmaceutical dosage forms.
Ophthalmic products have the same or similar requirements as injectables and implants. For more information on the requirements for these dosage forms, see Injections and Implanted Drug Products (1).
Degradation and/or morphological changes of the drug product can occur during the sterilization step of the manufacturing process depending on the formulation, container-closure system, and sterilization process. These changes should be minimized. Temperatures required for autoclaving can cause irreversible damage to certain dosage forms such as suspensions, semisolids, emulsions, and others, whereas filtration is applicable only to formulations containing particulates with particle sizes <0.2 µm. An alternative approach is to manufacture the drug product from sterile ingredients maintained within an aseptic environment.
Multi-dose products shall contain a suitable antimicrobial preservative, and the effectiveness of the preservative over the shelf life of the product must be verified. Alternatively, preservative-free multi-dose products require a packaging system that has been demonstrated to maintain sterility over the shelf-life of the product.
2.1 Solutions
[NOTE-Included in this section are solutions and those solid products that, when reconstituted according to the instructions in the labeling, result in a solution.]
The corneal contact time of topical ophthalmic solutions increases with the viscosity of the formulations. Several synthetic polymers, including but not limited to polyvinyl alcohol, polyvinylpyrrolidone, polyethylene glycol, poly(Acrylic acid), and cellulose derivatives such as hypromellose and hydroxyethyl cellulose, are commonly used as viscosity enhancers because of their physiologic compatibility and desirable physicochemical properties.
Another approach to increase the corneal contact time involves the use of polymers that provide the liquid formulation with semisolid consistency only when the liquid formulation is placed in the conjunctival or corneal area. In this way, instillation of the solution is followed by prolonged permanence as a result of the viscoelastic properties of the formed hydrogel. "Hydrogel", or "water-based gels", are three-dimensional networks of polymer chains containing water within the network. The structure of the polymer network can be physically crosslinked (e.g., via ionic or hydrogen bonds) or chemically crosslinked (via a covalent bond). The water content of hydrogels can be adjusted by modulating the composition and conformation of polymers, such as the hydrophilic/hydrophobic balance of polymer chains and pendant groups, and the degree of cross-linking. The in-situ gelling phenomenon is caused upon delivery to the eye by a change in the conformation of the polymer(s) that can be triggered by external stimuli such as temperature, pH, ionic content, and lacrimal fluid.
Additionally, some polymers can interact, via noncovalent bonds, with conjunctival mucin and maintain the formulation in contact with corneal tissues.
Solutions can be injected not only by the intravitreal route but also by other routes, such as subconjunctival, sub-Tenons, retrobulbar, suprachoroidal, and subretinal routes.
2.2 Suspensions
[NOTE-Included in this section are suspensions and those solid products that, when reconstituted according to the instructions in the labeling, result in a suspension.]
Use of aqueous or oily suspensions may be considered for a number of reasons such as the following: 1) a drug is poorly soluble in water,
2) a drug has poor stability in water, and 3) in cases where there is a need to increase contact time with the eye and to increase wetting of the eye. A drug particle size of <10 µm is desirable to reduce irritation.
After topical instillation, particles are expected to be retained in the cul-de-sac, and the drug slowly dissolves or is released from the polymeric structures by diffusion, dissolution, polymer degradation, or ion-exchange.
Suspensions can be injected not only by the intravitreal route but also through other routes, such as subconjunctival, sub-Tenons, retrobulbar, suprachoroidal, and subretinal.
2.3 Ointments
Eye ointments are semisolid products usually intended for application to the conjunctiva, cornea, or eyelid. They have longer contact time when compared to many solutions. A suitable ophthalmic ointment base is nonirritating to the eye and permits diffusion of the drug substance throughout the secretions bathing the eye.
The majority of water-free, oleaginous eye ointment bases are composed of white petrolatum and liquid petrolatum (Mineral oil), or the eye ointment base is a modification of the petrolatum-based formula. They are designed to melt at body temperature. Other types of lipophilic ointment bases can also be used. An anhydrous vehicle can be advantageous for moisture-sensitive drugs.
2.4 Gels
Semisolid gel products are an alternative to traditional ointments and are based on the effect of increasing the viscosity to prolong the retention of the drug in the eye. Several types of gelling agents can be used, such as poly(acrylic acid) derivatives (e.g., Carbomer) or hypromellose.
2.5 Emulsions
Topical ophthalmic emulsions are generally prepared by dissolving or dispersing the drug substance into an oil phase, adding suitable emulsifying and suspending agents, and vigorously mixing these components with water to form a uniform oil-in-water emulsion. Each phase is typically sterilized before or during charging into the mixing vessel. High-shear homogenization may be used to reduce oil droplet size to submicron size, which may improve the physical stability of the oil micelles so that they do not coalesce. The resulting dosage form should contain small oil droplets, uniformly suspended.
Limited aqueous solubility of the drug substance(s) is the most common rationale for developing an ophthalmic emulsion. The drug substance(s) can be added to the phase in which it is soluble at the beginning of the manufacturing process, or the drug substance can be added after the emulsion is prepared by a suitable dispersion process.
The physical stability of the emulsion can be measured by light-scattering techniques that characterize oil-phase, globule-size distribution. Suitable surfactants may be added to improve emulsion stability.
2.6 Strips
Ophthalmic strips can have different ophthalmic uses including, but not limited to, use as a diagnostic tool to visualize defects or aberrations in the corneal epithelium or to measure the amount of tear production. They are made of filter paper, and they can contain compounds such as Fluorescein Sodium. They are individually packed to preserve sterility until the moment of use.
2.7 Injections
Although injections are considered a dosage form for nomenclature purposes, they are referred to in this chapter by other dosage forms such as solution, suspension, emulsion, and other appropriate terminology based on physical form.
2.8 Inserts
Ophthalmic inserts and ocular systems are solid dosage forms of appropriate size and shape that are placed in the conjunctival fornix, in the lacrimal punctum (Figure 2), or on the cornea. Inserts can usually be removed, if adverse effects develop, and can provide extended release of the drug over a certain period of time. Inserts can be classified as bioabsorbable (soluble) or non-bioabsorbable (insoluble). Drug release from soluble inserts involves two steps: 1) fast release of a portion of the drug as the tear fluid penetrates into the system and 2) slow release as a gel layer is formed on the surface of the insert. Collagen shields made from porcine sclera collagen or bovine corium tissue-and devices obtained by molding, extrusion, or compression (minitablets) of gelling polymers-belong to this category of soluble inserts. Bioabsorbable polymers (e.g., cross-linked gelatin derivatives and polyesters) can be used to prepare bioabsorbable inserts; these matrices act as reservoirs or interact with the drug molecules through labile bonds.
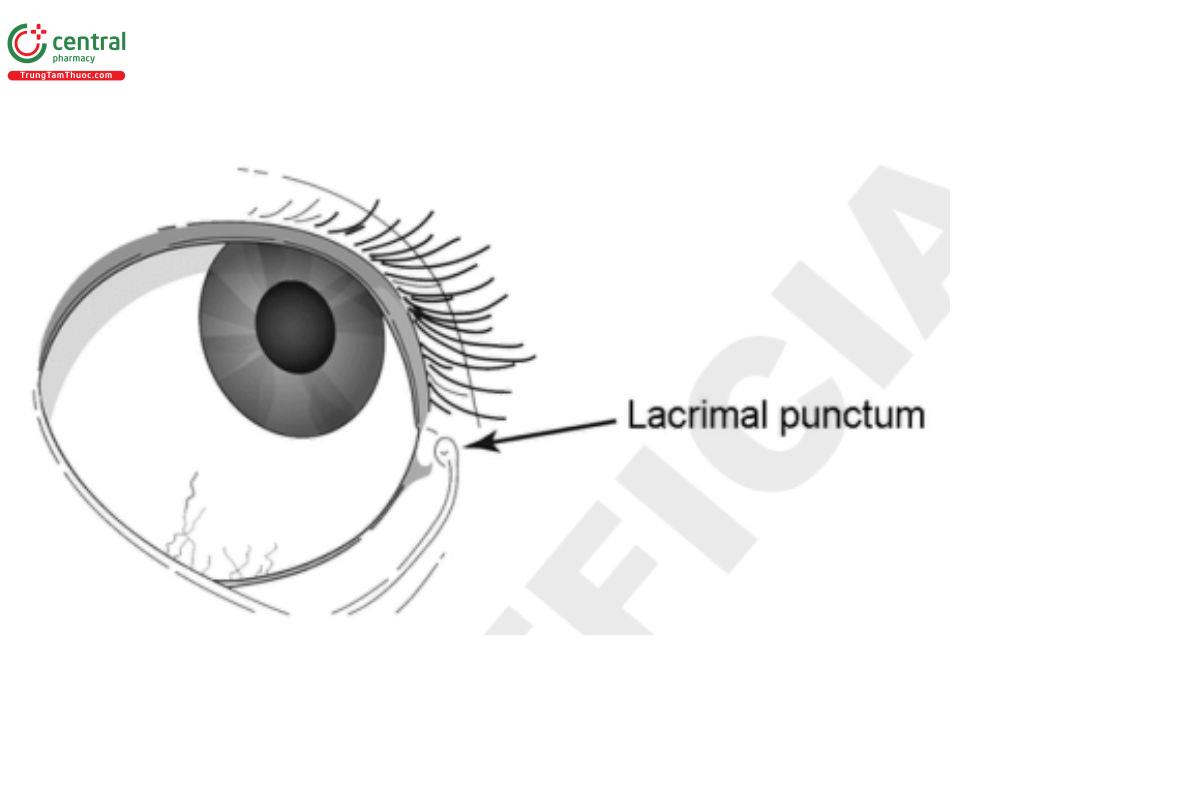
Soluble inserts can have the drug incorporated into a bioabsorbable matrix, such as hydroxypropyl cellulose, Hyaluronic Acid, or poly(acrylic acid) matrices. Soluble inserts may be placed in the lower cul-de-sac and generally dissolve within 12-24 h. Bioabsorbable polymeric products undergo gradual dissolution while releasing the drug, and the patient does not have to remove the bioabsorbable polymeric insert after use.
Insoluble inserts can have a reservoir or matrix structure. Their mechanism of action is based on diffusion of a fluid from the eye into the device, dissolution of the drug, and the creation of a saturated solution released to the medium by diffusion out of the insert. Insoluble inserts need to be removed after a certain period of time.
2.9 Implants
Implants are injected or implanted into the anterior segment (intracameral, subconjunctival, sub-Tenon, or other parts of the anterior segment) or intravitreal cavities, and the implants can be in different shapes, such as a disk or a thin rod. Implants can be made of non-bioabsorbable or bioabsorbable polymers.
Among the types of non-bioabsorbable solid (polymer) implants are reservoir-type implants anchored to the sclera, in which the drug is released across a non-bioabsorbable, semipermeable polymer. The reservoir-type implants can be made of a variety of materials, including poly(vinyl alcohol-ethylene vinyl acetate). Bioabsorbable polymers can be used to form injectable solid implants.
2.10 Drug-Device Combination Products
An ophthalmic drug-device combination product is constituted, in most cases, of two components. One is a pharmaceutical dosage form containing the drug substance(s), and the other is a device that will activate or facilitate the penetration of the drug substance(s) from the dosage form into a particular region of the eye. Some examples of the devices are those that generate waveforms (heat or light). Light sources can activate photoresponsive drugs.
This chapter is applicable only to the pharmaceutical dosage form component of the ophthalmic drug-device combination product. The appropriate FDA regulations regarding medical devices should be used for the device component.
Change to read:
3 DRUG PRODUCT QUALITY
Procedures and acceptance criteria for testing ophthalmic products are divided into two categories: 1) those that assess general quality attributes, e.g., identification, potency, purity (and impurities), sterility, and particulate matter; and 2) those that assess in vitro product performance, i.e., dissolution or drug release of the drug substance from the drug product. Quality tests assess the integrity of the dosage form, whereas the performance tests assess drug release and other attributes that relate to in vivo drug performance. Taken together, quality and performance tests ensure the identity, strength, quality, purity, and efficacy of the drug product. This chapter addresses the quality tests for ophthalmic products. The performance tests (dissolution/drug release) are addressed by Ophthalmic Products-Performance Tests (1771).
Universal Tests
In this chapter, the division of the product quality tests into universal and specific tests does not strictly follow the ICH guidance Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances (available at www.ich.org). Universal tests in this chapter mean the tests that are applicable to all ophthalmic products, regardless of the dosage form type.
DESCRIPTION
A qualitative description of the drug product should be provided. The acceptance criteria should contain the final acceptable appearance, including clarity and color, of the dosage form and packaging. If the color changes during storage, a quantitative procedure may be appropriate. This is not a compendial test but is part of the manufacturer's specification of the drug product.
IDENTIFICATION
Identification tests are discussed in General Notices, 5.40 Identification. Identification tests should establish the identity of the drug or drugs present in the article and should discriminate between compounds of closely related structures that are likely to be present. Identity tests should be specific for the drug substance(s). If the test is nonspecific, at least two orthogonal nonspecific tests should be used. Identification by use of a single chromatographic retention time does not constitute a specific test.
ASSAY
A specific and stability-indicating test should be used to determine the strength (content) of the drug product. In cases where the use of a nonspecific assay test is justified, other supporting analytical procedures should be used to achieve overall specificity. A specific procedure should be used when there is evidence of excipient interference with the nonspecific assay test. Additional information on specific assays may be found in Antibiotics-Microbial Assays (81), Chromatography (621), and lon Chromatography (1065).
IMPURITIES
Process impurities, synthetic byproducts, and other inorganic and organic impurities may be present in the drug substance and excipients used in the manufacture of the drug product. These impurities are controlled by the drug substance and excipient monographs. Organic impurities arising from the degradation of the drug substance in the drug product and those impurities arising during the manufacturing process of the drug product should be monitored. All articles meet the requirements in Elemental Impurities-Limits (232) and Residual Solvents (467).
PH
See pH (791). Normal tears have a pH of about 7.4. The eye can tolerate products over a range of pH values from about 3.0 to about 8.6, depending on the buffering capacity of the formulation. The pH value of the formulation should be the one where the drug product is the most stable. Formulations that target the extremes of the acceptable pH range will have better patient acceptability if the formulations have a low buffering capacity.
OSMOLARITY
See Osmolality and Osmolarity (785). Ophthalmic products may be tolerated over a wide range of tonicity (0.5%-5% sodium chloride, equivalent to about 171-1711 mOsm/kg). Hypotonic solutions are better tolerated than hypertonic solutions. Precautions should be taken to ensure that the product maintains its osmolarity during shelf life. Any possible contributions or interferences from the packaging system should be considered.
PARTICULATE AND FOREIGN MATTER
As described in Visual Inspection of Injections (1790), 4.1 Extrinsic, Intrinsic, or Inherent Particles, unwanted particulate matter is categorized as extrinsic (foreign matter) and intrinsic (formulation-, process-, or container-related matter or particulate matter due to product instability or product package interactions). A third category, inherent, describes a physical state that is an expected attribute of the product, such as a suspension or emulsion. It is expected that the manufacturer has established an acceptable control level for any inherent attribute, such as solution color, haze, or drug/excipient-related particle content.
The term 100% unit inspection is defined in (1790), 3.1 100% Inspection as the complete nondestructive inspection of the container-closure system and its contents. Therefore, evaluation of package and fill acceptability is shown by using the methodology in Visible Particulates in Injections (790), and this methodology is applied to all ophthalmic products: topical, intraocular injections or ophthalmic products used in association with a surgical procedure, and extraocular injections.
Intraocular administration includes all ophthalmic products that cross (penetrate) boundary tissue such as the cornea and sclera. Extraocular administration of ophthalmic products includes all the other ocular components and spaces.
For all intraocular solutions, conformance to (790), and Subvisible Particulate Matter in Intraocular Solutions (789) (CN 1-May-2024) is required. Additionally, for extraocular (nontopical) solution products, conformance to (790), and evaluation by Particulate Matter in Injections (788) is required. For topical products, conformance to (790) is required. See the summary of this information in Table 1.
Table 1. Particulate and Foreign Matter Requirements for Ophthalmic Solutions
| Route of Administration (See Figure 1) | Must comply witha |
| Topical | (790) |
| Peribulbar | (790), (788) |
| Superior rectu | (790). (788) |
| Sub-Tenon | (790), (788) |
| Subconjunctiv | (790), (788) |
| Inferior rectus | (790), (788) |
| Retrobulbar | (790), (788) |
| Suprachoroida | (790), (789) |
| Juxtascleral | (790), (788) |
| Intrascleral | (790), (789) |
| Intracorneal | (790), (789) |
| Subchoroidal | (790), (789) |
| Subretinal | (790), (789) |
| Intracameral | (790), (789) |
| Intravitreal | (790), (789) |
a Chapter (787) may be used instead of (788) for protein biotherapeutic formulas. For further information on difficult-to-inspect products, see (1790).
For nonsolution dosage forms (e.g., ointments, gels, suspensions, and emulsions) and dosage forms packaged in opaque containers (e.g., opaque bottles, metal tubes), the approach must be modified because these products are not directly amenable to the tests listed in Table 1. It is recommended to use 100% unit inspection per (790), for package acceptability (including but not limited to integrity, form, dimensions, cleanliness, label); whereas, an alternate testing strategy may be used for foreign particles where the dosage form hinders or precludes the use of these tests. For these products, a statistically relevant sample, examined by a destructive or other alternative method, will determine batch acceptability for visible particles as required by (790) and further described in (1790).
All ophthalmic products will benefit from evaluation of unwanted particulate matter content through the control strategy discussed in (1790), and in the FDA's current good manufacturing practices guidance. The final product, therefore, benefits from a control strategy including comprehensive evaluation of excipient and active ingredient quality, processing effects, package quality, and fill control. These concepts are discussed in (1790).
STERILITY
Ophthalmic dosage forms must meet the requirements of Sterility Tests (71). If the specific ingredients used in the formulation do not lend themselves to routine sterilization techniques, ingredients that meet the sterility requirements described in (71), along with aseptic manufacture, may be used. The immediate container for ophthalmic products shall be sterile at the time of filling and closing. See also Universal Tests, Container-Closure Integrity.
ANTIMICROBIAL PRESERVATIVES
Antimicrobial agents must be added to products that are packaged in containers that allow for the withdrawal or administration of multiple doses, unless one of the following conditions prevails: 1) there are different directions in the individual monograph; 2) the radiopharmaceutical drug product contains a radionuclide with a physical half-life of <24 h; 3) the drug product, without additional agents, is sufficiently microbicidal to meet the requirements of Antimicrobial Effectiveness Testing (51), or 4) the container-closure system is capable of maintaining sterility of the product throughout its shelf life until opened or accessed. Once opened or accessed, it must maintain antimicrobial effectiveness during the intended use period. Antimicrobial agents must meet the requirements of (51) and Antimicrobial Agents-Content (341). Acceptance criteria for antimicrobial preservative content in multi-dose products should be established.
BACTERIAL ENDOTOXINS
All injected ophthalmic drug products shall be prepared in a manner designed to minimize bacterial endotoxins as defined in Bacterial Endotoxins Test (85). The limits are not more than 0.5 EU/mL for ophthalmic irrigation products and not more than 2.0 EU/dose/eye for injected or implanted drug products. Typically, this test is not required for topically applied ophthalmic products. This chapter does not address the endotoxin limits for devices that are injected or implanted.
UNIFORMITY OF DOSAGE UNITS
This test is applicable to dosage forms packaged in single-unit containers. It includes both the mass of the dosage form and the content of the drug substance(s) in the dosage form. The test can be performed by either content uniformity or weight variation (see Uniformity of Dosage Units (905)).
CONTAINER CONTENTS
Container contents of ophthalmic products should be determined (see Minimum Fill (755)).
LEACHABLES AND EXTRACTABLES
The packaging system should not interact physically or chemically with the product in any manner to alter the strength, quality, or purity of the drug product. The packaging system should meet the requirements in Elastomeric Components in Injectable Pharmaceutical Product Packaging/Delivery Systems (381), Containers-Glass (660), and, depending on which standards the manufacturer has implemented, Plastic Packaging Systems and Their Materials of Construction (661) or Plastic Materials of Construction (661.1) and Plastic Packaging Systems for Pharmaceutical Use (661.2). Further information regarding packaging systems testing can be found in Assessment of Extractables Associated with Pharmaceutical Packaging/Delivery Systems (1663) and Assessment of Drug Product Leachables Associated with Pharmaceutical Packaging/Delivery Systems (1664). The assessment of possible leachables and/or extractables and the establishment of any acceptance criteria for these compounds should consider the risk assessment of the product, its indication, and its packaging system.
This assessment is done in a case-by-case approach during product development and re-evaluated when any changes are made to the product including, but not restricted to, changes in manufacturing process, formulation, and packaging material.
Furthermore, it should be noted that a risk assessment of extractable/leachable impact on the topical or intraocular route of administration is a challenging undertaking. Toxicological or safety assessments of primary or secondary packaging component extractables and leachables are not typically available for ophthalmic routes of administration. A risk assessment may include evaluation of toxicology and safety from other routes of administration and an assessment of the total daily intake of the extractable/leachable being evaluated. The preponderance of such assessments leads to an estimate of extractable/leachable risk via ocular delivery to the patient.
CONTAINER-CLOSURE INTEGRITY
The packaging system should be closed or sealed in such a manner as to prevent contamination or loss of contents and should incorporate a tamper-evident design. Validation of container integrity must demonstrate no penetration of microbial contamination or of chemical or physical impurities (see Package Integrity Evaluation-Sterile Products (1207)).
3.1 Specific Tests
VISCOSITY
An increase in viscosity increases the residence time of ocular formulations. However, drug diffusion out of these formulations into the ocular tissue may be modified due to high product viscosity. Ophthalmic ointments are designed to be of very high viscosity to prolong the residence time of the formulation in the eye. The inclusion of viscosity evaluation in the specification of the product should be based on the type of dosage form and whether changes in product viscosity will affect its performance. This is not a compendial test but is part of the manufacturer's specification of the drug product. See Viscosity-Capillary Methods (911), Viscosity-Rotational Methods (912), and Viscosity, -Rolling Ball Method (913).
ANTIOXIDANT CONTENT
If antioxidants are present in the drug product, tests of their content should be established, unless oxidative degradation can be detected by another test method such as impurity testing. Acceptance criteria for antioxidant content should be established. They should be based on the levels of antioxidant necessary to maintain the product's stability at all stages throughout its proposed usage and shelf life.
RESUSPENDABILITY/REDISPERSIBILITY
Consideration must be given to establishing good physical stability of a suspension. If the particles settle and eventually produce a cake at the bottom of the container, they must redisperse readily at the time of use to achieve dosage uniformity. Quantitative demonstration of the active ingredient's resuspendability/redispersibility during product development is also recommended.
PARTICLE SIZE AND PARTICLE SIZE DISTRIBUTION
The potential for any changes in the particle size of ophthalmic suspensions and emulsions needs to be evaluated through stability testing (see Light Diffraction Measurement of Particle Size (429)).
DROP SIZE
For ophthalmic drug products dispensed as drops, drop sizes may typically range from 20-70 µL. Drop size can be controlled by weight or by volume, and it is typically evaluated during product development. Drop size can be determined by any appropriate validated procedure.
ADDED SUBSTANCES
Suitable substances may be added to ophthalmic products to increase stability, unless proscribed in the individual monograph, provided they are harmless in the amounts administered and do not interfere with therapeutic efficacy or with responses to the specified assays and tests. Typically, these substances should be evaluated during product development. The use of ingredients solely to impart an odor or flavor is prohibited. The use of ingredients solely to impart a color is prohibited unless coloring is needed to visualize the administration location of the product.


