Mucosal Drug Products—Performance Tests
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
Mucosal drug products deliver drug substances to the body via the mucosal route. For the purposes of this chapter, the mucosal route of drug administration is divided into seven membrane surfaces characterized as otic, ophthalmic, nasal, oropharyngeal, urethral, vaginal, and rectal. Mucosal drug products include a wide variety of dosage forms such as solutions, suspensions, emulsions, creams, ointments, gels, inserts, strips, aerosols, sprays, lms, medicated chewing gums, lozenges, tablets, and suppositories. Some of these dosage forms are also administered by other routes. For example, creams can be administered by the mucosal route (vaginal) and also by the topical route. Two categories of tests—product quality and product performance—are performed on these products. These tests provide assurances of batch to-batch quality, reproducibility, reliability, and performance of a drug product. Product quality tests are performed to assess attributes such as assay, identification, and content uniformity and are part of the compendial monograph (see Mucosal Drug Products—Product Quality Tests 〈4〉). Product performance tests are conducted to assess the drug release from the dosage form. For certain mucosal drug products, determination of aerodynamic particle size or globule size may serve as a product performance test.
Where a compendial performance test exists for a dosage form administered by a nonmucosal route, such as a dissolution test for an oral tablet, that test may have application for the dosage form administered by a mucosal route (e.g., buccal tablets or sublingual tablets).
2 PERFORMANCE TESTS FOR MUCOSAL DRUG PRODUCTS
The performance tests for the various mucosal drug products can be broadly divided into two categories: 1) test procedures that use or can adopt methodology in existing general chapters, and 2) tests that need additional developmental work before they can be recommended. The Dissolution Procedure: Development and Validation 〈1092〉 should be a reference when developing a drug release test (e.g., selecting the drug release medium, apparatus/procedure, or analytical method). For several mucosal drug products, drug release procedures described in Dissolution 〈711〉, Drug Release 〈724〉, and Semisolid Drug Products—Performance Tests 〈1724〉 may be applicable. In some instances, mini-basket or mini-paddle apparatuses may be suitable. These apparatuses resemble Dissolution 〈711〉, Apparatus 1 (Basket Apparatus) and Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus), with dimensions scaled down to accommodate medium volumes of <500 mL (1,2). Several designs are commercially available. However, as of now, these apparatuses are not standardized. Because of the varied and specific environments characterizing the mucosal route of administration, researchers may be inclined to use “physiological medium” for the drug release of the specific dosage form. It may not be essential to use such a medium for the performance test of the product, and in many instances a simple buffer may suffice. A reference (3) for the composition of such media is provided for additional information.
2.1 Aerosols and Nasal Sprays
Performance tests for nasal and lingual aerosols and nasal sprays are largely concerned with droplet or particle size distribution and aerodynamic size distribution. The procedures in Inhalation and Nasal Drug Products: Aerosols, Sprays, and Powders—Performance Quality Tests 〈601〉 can be applied to products administered by mucosal routes. When the drug substance is present as a solid with a modified release mechanism in the administered dose, attempts should be made to determine the dissolution of the particles (4).
2.2 Creams, Gels, and Ointments
Drug release tests for creams, gels, and ointments can be performed using a procedure described in 〈1724〉.
Change to read:
2.3 Emulsions
Performance tests for emulsions include globule size determination and dissolution/drug release testing. Globule size can be determined using a procedure described in Globule Size Distribution in Lipid Injectable Emulsions 〈729〉. The drug release test can be performed using Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus) or a vertical diffusion cell as described in Semisolid Drug Products—Performance Tests 〈1724〉, In Vitro Release Test (IVRT) (CN 1-Dec-2023) .
2.4 Films
Drug Release 〈724〉, Apparatus 5 (Paddle over Disk) can be used to determine the drug release from lm dosage forms. A mini-basket can be used for drug release testing of lms.
For gum products, the performance test includes drug release from the formulation. A device is described in the European Pharmacopoeia (5). The release of drug from the formulation requires masticatory activity that renews the surface exposed to the medium. The rate of release will be a function in part of the frequency of chewing that is simulated by the test apparatus. Gums can require conditioning at the temperature of the mouth to deform plastically under the action of the oscillating platens of the test apparatus.
Important parameters for the apparatus include: dissolution medium volume, distance between upper and lower chewing surfaces, recommended rotation angle, temperature, and chewing frequency. The dissolution medium chosen, the test time(s), and the volume sampled are also important considerations.
2.5 Inserts
Drug release testing for inserts can be performed using Dissolution 〈711〉, Apparatus 1 (Basket Apparatus) or Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus).
2.6 Lozenges
Drug release testing of lozenges can be performed using either Dissolution 〈711〉, Apparatus 1 (Basket Apparatus); Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus) at high agitation (175 rpm); or Dissolution 〈711〉, Apparatus 3 (Reciprocating Cylinder).
2.7 Suppositories
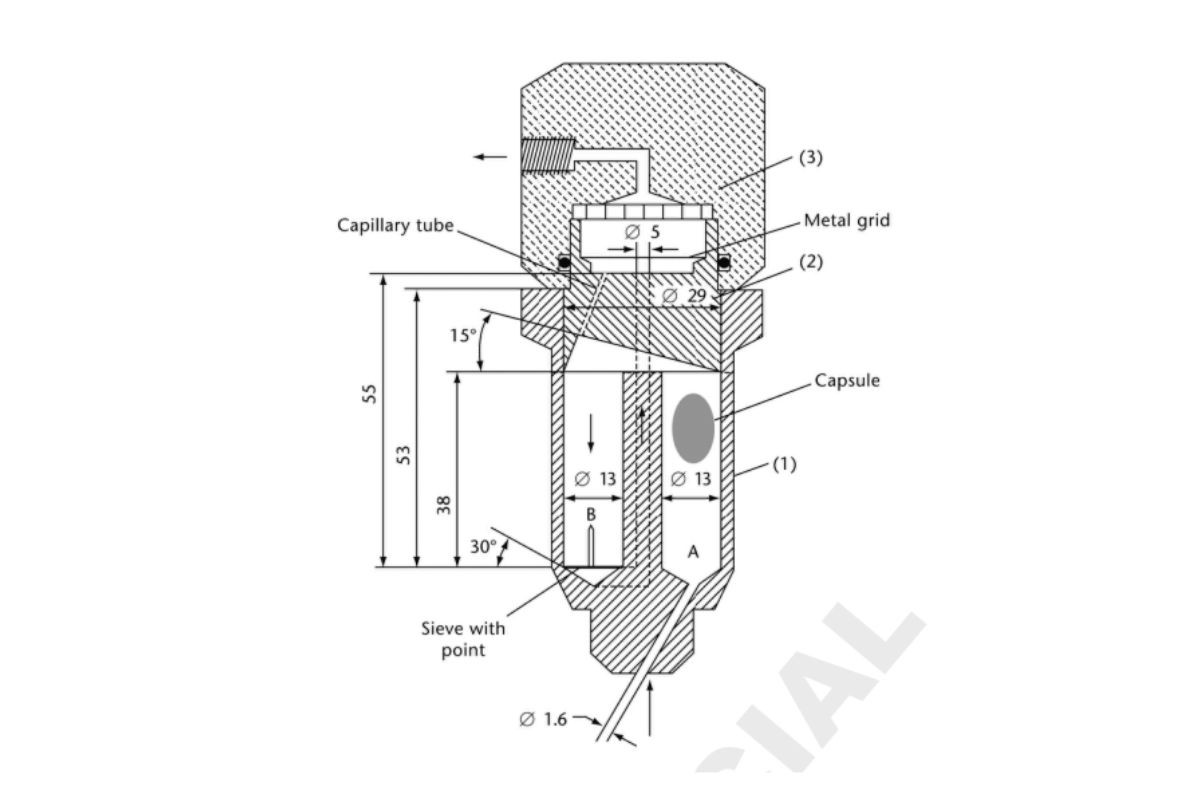
There are two types of suppositories: 1) hydrophilic (water soluble), and 2) lipophilic (oil soluble or melting). Drug release (dissolution) for water-soluble suppositories can be performed using Dissolution 〈711〉, Apparatus 1 (Basket Apparatus); Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus); or Dissolution 〈711〉, Apparatus 4 (Flow-Through Cell). Drug release testing for lipophilic suppositories may need modification of the dissolution procedure to avoid analytical interference from the oil globules. Several alternative methods have been proposed (6–8). The flow-through cell apparatus using the cell for suppositories may be useful. The selection of the method will be dependent on the nature of the formulation. Figure 1 shows the schematic view of a ow-through cell [Dissolution 〈711〉, Apparatus 4 (Flow-Through Cell)] specifically intended for dissolution of suppositories. The lower part (1) is made up of two adjacent chambers connected to an overflow device. The dissolution medium passes through chamber A and is subjected to an upward flow. The flow in chamber B is directed downward to a small size bore exit that leads upward to a filter assembly. The middle part (2) of the cell has a cavity designed to collect lipophilic excipients that float on the dissolution medium. A metal grid serves as a rough filter. The upper part (3) holds a filter unit for paper, glass ber, or cellulose filters.
2.8 Suspensions
The dissolution test for suspensions can be performed using Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus). A small-volume, mini paddle apparatus may be used.
2.9 Sublingual Tablets and Buccal Tablets
Drug release for these dosage forms can be performed using Dissolution 〈711〉, Apparatus 1 (Basket Apparatus) or Dissolution 〈711〉, Apparatus 2 (Paddle Apparatus). Mini-baskets or the mini-paddles can also be used for drug release testing of buccal and sublingual tablets.
3 REFERENCES
1. Klein S, Shah VP. A standardized mini paddle apparatus as an alternative to the standard paddle. AAPS PharmSciTech. 2008;9(4):1179–1184.
2. Crist GB. Trends in small-volume dissolution apparatus for low-dose compounds. Dissolut Technol. 2009;16(1):19–22. 3. Marques MRC, Loebenberg R, Almukainzi M. Simulated biological uids with possible application in dissolution testing. Dissolut Technol. 2011;18(3):15–28.
4. Son Y-J, Horng M, Copley M, McConville JT. Optimization of an in vitro dissolution test method for inhalation formulations. Dissolut Technol. 2010;17(2):6–13.
5. Dissolution test for medicated chewing gums. European Pharmacopoeia 8.5, 2.9.25. Strasbourg, France: Directorate for the Quality of Medicines of the Council of Europe (EDQM); 2015: 325–328.
https://www.edqm.eu/en
6. Yamazaki M, Itoh S, Sasaki N, Tanabe K, Uchiyama M. Comparison of three test methods for suppositories. Pharmacopeial Forum. 1991;17(5):2427–2437.
7. Gjellan K, Graffner C. Comparative dissolution studies of rectal formulations using the basket, the paddle, and the flow-through methods. I. Paracetamol in suppositories and soft gelatin capsules of both hydrophilic and lipophilic types. Acta Pharm Nord. 1989;1(6):343–354.
8. Palmieri A. Suppository dissolution testing: apparatus design and release of Aspirin. Drug Dev Ind Pharm.


