Mean Kinetic Temperature in the Evaluation of Temperature Excursions During Storage and Transportation of Drug Products
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
The extent of physicochemical degradation of drug products depends on factors such as product stability, how a product is stored and shipped, and how it is packaged. Temperature at which a product is stored or shipped is likely to vary during the life of the product, which can impact its stability and degradation pathways and kinetics, thereby leading to the product failing to maintain its critical quality attributes.
Mean kinetic temperature (MKT) is a way to summarize the time history of a product’s temperature exposure with a single “effective” or “virtual” temperature. It is defined as the single calculated temperature at which the total amount of degradation over a particular period is equal to the sum of the individual degradations that would occur at various temperatures. MKT integrates the time–temperature history by making assumptions about the kinetics of a product’s chemical degradation. Therefore, MKT takes into account the fact that long temperature excursions at slightly elevated temperatures can be just as, or more, impactful than short temperature excursions at elevated temperatures.
2 SCOPE
This chapter applies to every link in the supply chain from the manufacturer through any entity that transports or stores a finished drug product, with the sole exception of the patient. Examples:
- Manufacturers of drug products, radiopharmaceuticals, biological products, and biotechnological products
- Repackaging operations in which the product may be owned by a company other than the primary manufacturer
- Healthcare providers and institutions such as hospitals; outpatient, ambulatory, and urgent care centers; home health providers; vaccine clinics; emergency departments; and medical, dental, and veterinary offices
- Pharmacies including but not limited to retail, infusion/compounding (sterile and nonsterile), specialty, mail order, hospital, nursing home, and hospice
- Importers and exporters
- Wholesale distributors
- Third-party logistics providers, brokers, freight forwarders, consolidators, and other organizations involved in storage; road, rail, sea, and/or air transport services; or mail distributors that offer expedited or controlled-temperature shipping services
Manufacturers of active pharmaceutical ingredients, excipients, medical devices (with the exception of drug combination devices), and dietary supplements are not within the scope of this chapter. However, the principles and mitigation strategies presented in this chapter may be useful for materials other than finished drug products.
3 MEAN KINETIC TEMPERATURE
MKT is the single calculated temperature at which the total amount of degradation over a particular period is equal to the sum of the individual degradations that would occur at various temperatures. MKT may be considered as an isothermal storage temperature that simulates the nonisothermal effects of storage temperature variation.1 Seevers et al. demonstrated that the choice of activation energy doesn’t change the MKT significantly. It is not a simple arithmetic mean. The use of MKT is considered an acceptable practice for storage and can be considered for excursions during transit.
The temperatures used for calculating MKT can be conveniently collected using electronic devices that measure temperatures at frequent intervals (e.g., every 15 min). MKT can be calculated directly or the data can be downloaded to a computer for processing. Software to compute the MKT is available commercially.
The arithmetic mean of the high and low temperatures of the observation period is then used in the calculation of MKT. MKT is calculated by the following equation (derived from the Arrhenius equation):
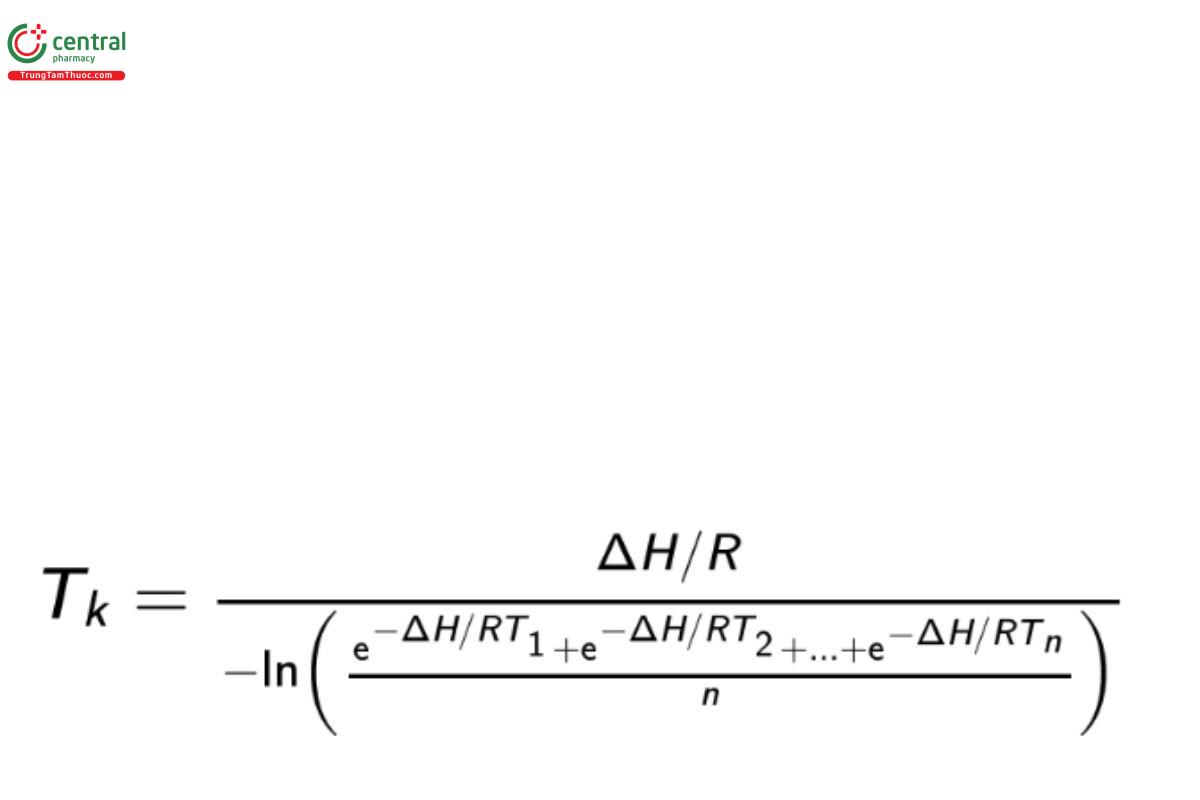
Tk = mean kinetic temperature
ΔH = heat of activation, 83.144 kilojoules · mole−1 (unless more accurate information is available from experimental studies)
o… R = universal gas constant, 8.3144 × 10−3 kj · mole−1 · K−1
n = total number of storage temperatures recorded during the observation period
T1 = value for the temperature recorded during the first time period
T2 = value for the temperature recorded during the second time period
Tn = value for the temperature recorded during the nth time period
[Note—All temperatures, T, are absolute temperatures in degrees Kelvin (K).]
Drug products in the distribution supply chain may be held at temperatures outside of their labeled storage requirements as determined by the appropriate stability data. Drug products stored either in warehouse conditions or during transportation may experience excursions from their acceptable temperature ranges. Each product excursion outside of USP excursion limits must be evaluated and its impact on the nal product determined. The means of evaluation must be scientifically sound with documented justification that the integrity and quality of the drug product have not been affected. One method of analysis for examining temperature excursion is the use of MKT.
Because MKT expresses the cumulative thermal stress that a drug product experiences, it is considered an acceptable practice for storage and can be considered for excursions during transit. The calculation must be justified for use with distribution excursions by confirming that the stability-limiting characteristic of the product follows zero-order or rst-order kinetics over the temperature range encountered.
Change to read:
4 APPLICATION OF MKT
Mean kinetic temperature has been misunderstood and/or misused. The most significant misuse has been utilizing 52 weeks of temperature data to calculate MKT during a temperature excursion. Drug products typically do not spend 52 weeks in a single storage location. Thus, the 52 weeks of data used in the MKT calculation would not be a true reflection of the storage time. This approach skews results and could lead a company to overlook the impact of an excursion on the drug product.1,2 A closely related concern is the idea that a temperature excursion above a product's storage temperature can be "xed" just by lowering the temperature of a warehouse for an appropriate period of time so that the resulting MKT calculation would provide an acceptable value. This ignores the fact that any degradation due to the higher temperature is not reversible.3
Mean kinetic temperature is referenced in the controlled room temperature (CRT) and controlled cold temperature (CCT) definitions in Packaging and Storage Requirements 〈659〉. Within the definitions, the temperature range for an excursion and the maximum excursion time are defined, but the time frame used for calculating MKT is not mentioned. For CRT excursions, USP recommends using 30 days for calculating MKT or the average number of days that a product remains in the holder’s possession. This recommendation is based on temperature data presented by Anderson et al.3 and the fact that products, on average, spend 30 days in warehouse storage in the United States.4
For CCT excursions, USP recommends using 24 h for calculating MKT. While specific products may have stability data to support excursions within wider temperature and time frames, operations involved with storage and distribution (shipping) throughout the supply chain typically do not have access to information beyond the product labeling. This includes dispensing and healthcare providers. Excursions occur and these guidelines are intended to manage these excursions while being conscious of the impact of temperature and time excursions. It is important to note that an excursion is a nonconforming event (except as previously noted when provided by the manufacturer). MKT should not be used to justify a system out of control (e.g., repeated temperature and time excursions).
Table 1. Use of MKT for CRT and CCT Excursionsa
Storage Range | Acceptable Excursion Range | Maximum Temperature | Maximum Excursion Time (NMT) | MKT (NMT) | Time Period of Calculation | |
CRTb | 20°–25° | 15°–30° | 40° | 24 h | 25° | 30 days |
CCTc | 2°–8° | 8°–15° | 15° | 24 h | 8° | 24 h |
a Exposure to higher and/or lower temperatures beyond what is recommended in this chapter should be evaluated using monographs, product labels, or stability data provided by the manufacturer.
b Some CRT- labeled products may have stability issues with the higher and/or lower excursion limits. These products must be evaluated and shipped within the stability limits provided by the manufacturer.
c An article for which storage at CCT is directed may, alternatively, be stored and shipped in a cool place or refrigerated unless otherwise specied in the monograph or on the label (see 〈659〉).
In CRT storage environments, temperature monitoring (see Monitoring Devices—Time, Temperature, and Humidity 〈1079.3〉 (CN 1-Dec-2023) ) captures the temperatures throughout the day (e.g., every 15 min). MKT can be calculated on an ongoing basis or anytime that there has been a temperature excursion using data going back 30 days from (and including) the high excursion temperature. 30 days must be from, and including, the high excursion temperature for the entire period of the excursion.
4.1 Use of MKT for Controlled Cold Temperature Excursions
In CCT storage environments, temperature monitoring (see 〈1079.3〉 (CN 1-Dec-2023) ) captures the temperatures throughout the day (e.g., every 15 min). MKT can be calculated on an ongoing basis or anytime that there has been a temperature excursion using data going back 24 h from the (and including) high excursion temperature. Excursion ranges, time, and MKT within allowances should be documentedand can be reviewed for compliance and trending. The excursion temperature and time allowances for CCT products are much more restrictive than for most CRT products due to the more sensitive nature of these products to temperature excursions. Each excursion should be considered a separate event. A storage or transportation system that has repeated excursions should be considered a system out of control that needs to be corrected.
Excursion ranges, time, and MKT within allowances should be documented and can be reviewed for compliance and trending. Transportation systems that are monitored may also use MKT as outlined in this chapter. For transportation systems that are not monitored (a risk-based decision), lane temperature prole mapping may include the use of MKT when evaluating risks (see Risks and Mitigation Strategies for the Storage and Transportation of Finished Drug Products 〈1079〉).
5 CONCLUSION
While every effort should be made to store and distribute drugs within their labeled temperature range, excursions may occur. The use of MKT in evaluating a short-term excursion (as defined in 〈659〉) and the time period used for calculating MKT as recommended by USP will allow responsible management of excursions. It is important to note that excursions outside of USP allowable limits must be evaluated by the manufacturer, where conveying the excursion temperatures and time is critical to determining product disposition. MKT may not be used to justify a storage or transportation system that has repeated excursions; such a system is not in control and needs to be corrected.
1 Seevers RH, Hofer J, Harber P, Ulrich D, Bishara R. The Use of Mean Kinetic Temperature (MKT) in the Handling, Storage, and Distribution of Temperature Sensitive Pharmaceuticals in Pharmaceutical Outsourcing; May–June 2009, 10(3); 30–35.
2 Anderson C, Seevers R, Hunt D. The use of mean kinetic temperature to aid evaluation of temperature excursions: Proper and improper application. Pharm. Forum 2018;44(4).
3 Anderson C, Seevers R, Hunt D. The use of mean kinetic temperature to aid evaluation of temperature excursions for controlled cold temperature drugs: Proper and improper application. Pharm. Forum 2019;45(5).
4 Center for Healthcare Supply Chain Research. Factbook: The Facts, Figures and Trends in Healthcare (2015–2016). 86th ed. Arlington, VA: Center for Healthcare Supply Chain Research; 2016:15.


