Limit on Number of Large Deviations When Assessing Content Uniformity in Large Samples
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
To view the Notice from the Expert Committee that posted in conjunction with this accelerated revision, please click https://www.uspnf.com/rb-gc-1099-20190222
1 INTRODUCTION
The uncertainty around the application of the zero tolerance criterion (ZTC) to sample sizes larger than 30 may inhibit the collection of uniformity data from large samples. This chapter provides a process for limiting the number of observed results that fall outside of the ZTC (c₂), as described in Uniformity of Dosage Units 〈905〉, when samples larger than 30 are collected. It should be noted that the criterion described in this chapter is not intended as a batch release test, nor as a replacement of or alternative to 〈905〉. It also is not intended as an extension to go beyond second tier testing in 〈905〉. The use is solely to help judging if a large data set is consistent with the ZTC element of 〈905〉; whether the large data set complies with the complete set of requirements in 〈905〉 must be decided by other means.
The ZTC in 〈905〉 states that no individual content of any dosage unit can be less than [1 − (0.01)(L₂)]M nor more than [1 + (0.01)(L₂)]M, where L₂ is 25.0% unless otherwise specified in the applicable monograph, and M, the “reference value”, depends on the sample mean, X̄ (expressed as a percentage of the label claim), as follows:
M = 98.5% if X̄ < 98.5, M = 101.5% if X̄ > 101.5, and M = X̄ otherwise, with a sample size (N) of 30
2 PROCEDURE
When a sample that includes the contents of more than 30 units has been collected, the following procedure can be used to confirm that the results in that sample are consistent with the ZTC of 〈905〉. The criterion is applicable both when the content is determined directly by assaying a number of units and when the content is determined indirectly by weighing the units in the situations allowing this as described in 〈905〉. The procedure is as follows:
Express individual results x₁, x₂, ..., xₙ as a percentage of the label claim
Calculate the mean (X̄) of the contents of the N units in the sample
Calculate the reference value M: M = 98.5% if X̄ < 98.5, M = 101.5% if X̄ > 101.5, and M = X̄ otherwise
Determine S₂, the number of sample results less than [1 − (0.01)(L₂)]M or more than [1 + (0.01)(L₂)]M, where L₂ = 25.0% unless otherwise specified in the applicable monograph
The sample is consistent with the ZTC of 〈905〉 if S₂ ≤ c₂, where c₂ depends on the sample size as detailed in Table 1
Table 1. Limit on Number of Observed Results Falling Outside of the ZTC Based on Sample Size
| N | c₂ |
| 31–100 | 0 |
| 101–181 | 1 |
| 182–265 | 2 |
| 266–353 | 3 |
| 354–442 | 4 |
| 443–533 | 5 |
| 534–624 | 6 |
| 625–717 | 7 |
| 718–810 | 8 |
| 811–903 | 9 |
| 904–998 | 10 |
| 999–1092 | 11 |
| 1093–1187 | 12 |
| 1188–1283 | 13 |
| 1284–1379 | 14 |
| 1380–1475 | 15 |
| 1476–1571 | 16 |
| 1572–1667 | 17 |
| 1668–1764 | 18 |
| 1765–1861 | 19 |
Values of c₂ for other sample sizes (N) are determined as
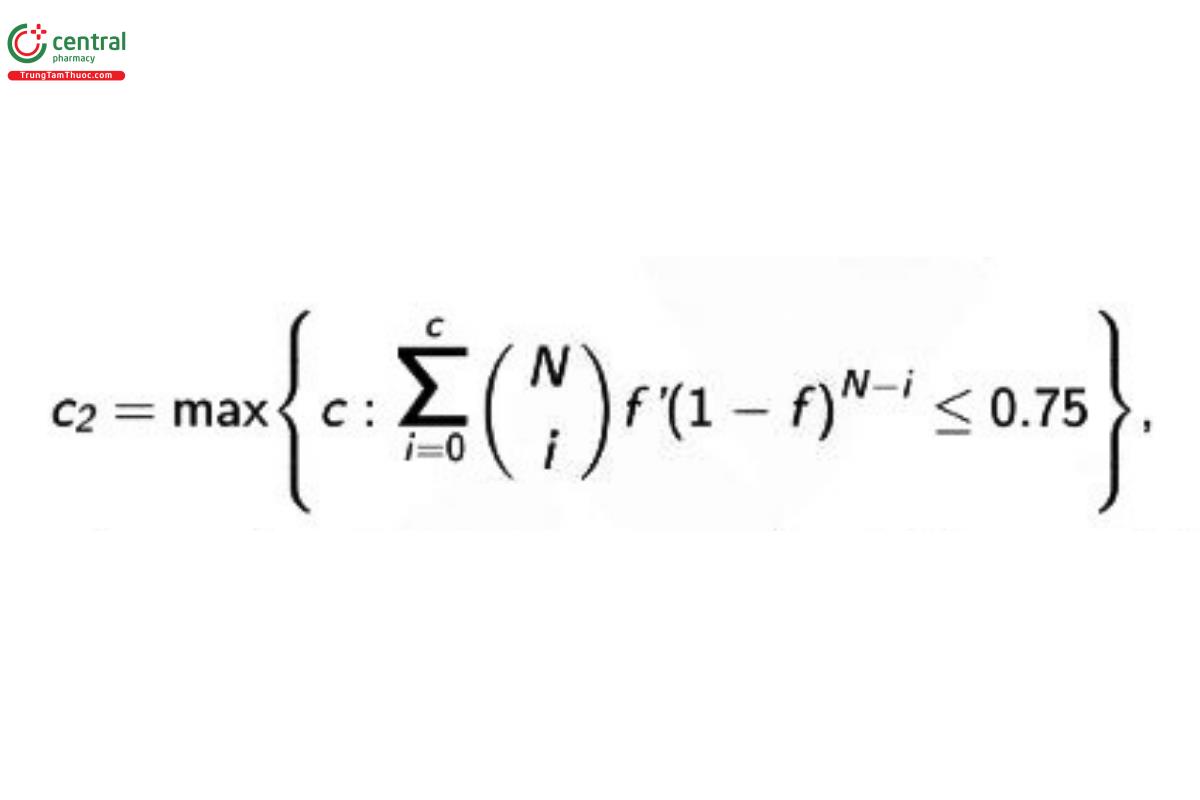
where f = 1 − 0.75¹ᐟ³⁰ = 0.00954357. The c₂ value can be calculated in a spreadsheet with a cumulative binomial function. (Postponed on 1-Mar-2019)


