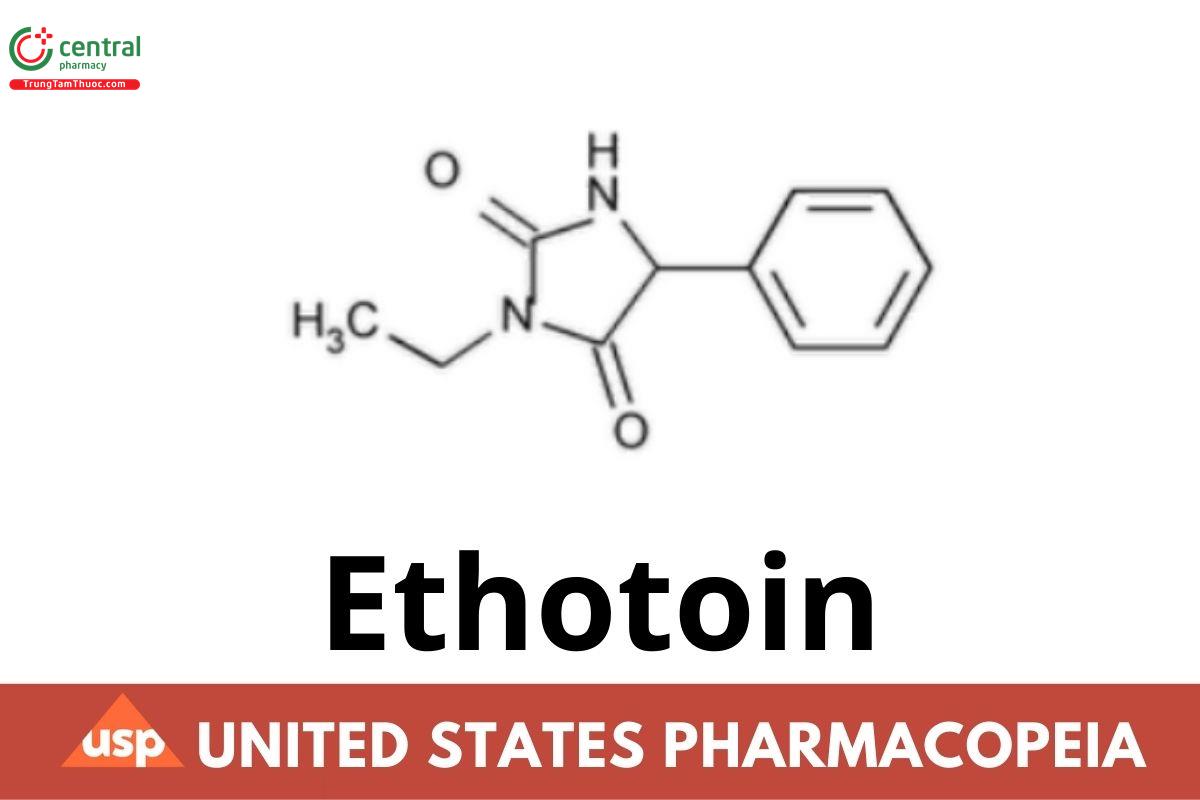
Ethotoin
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
DOWNLOAD PDF HERE
Ethotoin contains not less than 97.5 percent and not more than 102.0 percent of C11H12N2O2, calculated on the dried basis.
1 Packaging and storage
—Preserve in tight containers.
USP Reference standards 〈11〉—
USP Ethotoin RS
USP 5-Phenylhydantoin RS
2 Identification—
Change to read:
A: Spectroscopic Identification Tests 〈197〉, Ultraviolet-Visible Spectroscopy: 197U (CN 1-May-2020) —
Solution: 1 mg per mL.
Medium: alcohol.
B: The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Loss on drying 〈731〉—Dry it in vacuum at 60° for 4 hours: it loses not more than 1.0% of its weight.
Residue on ignition 〈281〉: not more than 0.1%.
3 Chloride
—Transfer 1.0 g of Ethotoin to a suitable separator, and dissolve in 50 mL of ether. Extract with three 15-mL portions of water, collect the combined extracts in a beaker, heat on a steam bath to expel any traces of ether, and allow to cool to room temperature. Transfer the solution to a 50-mL color-comparison tube, add 2 N nitric acid until the solution is acidic, add 1 mL of 2 N nitric acid in excess, mix, add 1 mL of silver nitrate TS, dilute with water to 50 mL, and allow to stand for 5 minutes, protected from direct sunlight. The turbidity produced does not exceed that of a solution prepared by mixing 2 mL of freshly prepared 0.002 N hydrochloric acid, 1 mL of 2 N nitric acid, 1 mL of silver nitrate TS, and 46 mL of water (0.014%).
4 Related compounds—
Buffer solution—Dissolve about 1 g of monobasic sodium phosphate in 1 L of water. Adjust the solution with 1.5 M phosphoric acid to a pH of 3.5 ± 0.1.
Diluent—Prepare a mixture of Buffer solution and methanol (65:35).
Solution A—Prepare a filtered and degassed mixture of Buffer solution and acetonitrile (80:20).
Solution B—Prepare a filtered and degassed mixture of Buffer solution and acetonitrile (60:40).
Mobile phase—Use variable mixtures of Solution A and Solution B as directed for Chromatographic system. Make adjustments if necessary (see System Suitability under Chromatography 〈621〉).
Standard solution—Dissolve an accurately weighed quantity of USP Ethotoin RS, and dilute quantitatively, and stepwise if necessary, with Diluent to obtain a solution having a known concentration of about 2.5 μg of ethotoin per mL.
Test solution—Transfer about 50 mg of Ethotoin, accurately weighed, to a 200-mL volumetric flask, dissolve in about 100 mL of Diluent with sonication, dilute with Diluent to volume, and mix.
Chromatographic system (see Chromatography 〈621〉)—The liquid chromatograph is equipped with a 210-nm detector and a 4.6-mm × 15-cm column that contains 5-μm packing L1. The column temperature is maintained at 40°. The flow rate is about 0.8 mL per minute. The chromatograph is programmed as follows.
Time (minutes) | Solution A (%) | Solution B (%) | Elution |
| 0 | 100 | 0 | equilibration |
| 0–10 | 100 | 0 | isocratic |
| 10–30 | 100→0 | 0→100 | linear gradient |
| 30–40 | 0 | 100 | isocratic |
| 40–42 | 0→100 | 100→0 | linear gradient |
| 42–55 | 100 | 0 | re-equilibration |
Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the tailing factor is not more than 2.0 for ethotoin; and the relative standard deviation for replicate injections is not more than 1.0%.
Procedure—Separately inject equal volumes (about 100 μL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the responses for the major peaks. [Note—Discard any peak due to the Diluent.] Calculate the percentage of any impurity in the portion of Ethotoin taken by the formula:
(20,000/F)(C/W)(ri/rs)
in which C is the concentration, in mg per mL, of USP Ethotoin RS in the Standard solution; F is the response factor for the impurity as shown in the table below; W is the weight, in mg, of the portion of Ethotoin taken; ri is the peak area for any impurity in the Test solution; and rs is the peak area for ethotoin in the Standard solution. The impurities meet the requirements given in the table below.
| Compound name | RRT1 | RRF2 | Limit (%) |
| 5-Phenylhydantoin | about 0.4 | 1.0 | 1.5 |
| 3-Methyl-5-phenylhydantoin | about 0.6 | 1.0 | 0.9 |
| Ethotoin | 1.0 | - | - |
Ethotoin/5-Phenylhydantoin dimer | about 1.9 | 1.0 | 0.3 |
| Ethotoin dimer | about 2.5 | 0.58 | 0.4 |
| Unknown impurities | - | 1.0 | 0.1 Individual |
| 1.0 Total Unknown | |||
| Total | - | - | 2.0 |
1 RRT—Relative retention time.
2 RRF—Relative response factor.
5 Assay—
Mobile phase—Dissolve 0.65 g of monobasic potassium phosphate in 600 mL of water, adjust with phosphoric acid solution (1 in 10) to a pH of 3.5 ± 0.1, and dilute with water to 650 mL. Add 350 mL of methanol, mix, filter through a membrane filter of 0.5 μm or finer porosity, and degas. Make adjustments if necessary (see System Suitability under Chromatography 〈621〉).
5-Phenylhydantoin stock solution—Dissolve, with the aid of sonication if necessary, an accurately weighed quantity of USP 5-Phenylhydantoin RS in Mobile phase to obtain a solution having a known concentration of about 0.37 mg per mL.
Standard preparation—Dissolve, with the aid of sonication if necessary, an accurately weighed quantity of USP Ethotoin in Mobile phase to obtain a solution having a known concentration of about 0.25 mg of ethotoin per mL.
System suitability solution—Transfer 25 mg of USP Ethotoin RS, accurately weighed, to a 100-mL volumetric flask, add about 1.0 mL of 5-Phenylhydantoin stock solution, add Mobile phase to volume, and sonicate to dissolve.
Assay preparation—Transfer about 50 mg of Ethotoin, accurately weighed, to a 200-mL volumetric flask, dissolve in Mobile phase, dilute with Mobile phase to volume, and sonicate to dissolve.
Chromatographic system (see Chromatography 〈621〉)—The liquid chromatograph is equipped with a 210-nm detector and a 4.6-mm × 30-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the resolution, R, between the 5-phenylhydantoin and ethotoin peaks is not less than 6.0. The relative retention times are about 0.4 for 5-phenylhydantoin and 1.0 for ethotoin. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections is not more than 3.0%.
Procedure—Separately inject equal volumes (about 20 μL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C11H12N2O2 in the portion of
Ethotoin taken by the formula:
200C(rU/rS)
in which C is the concentration, in mg per mL, of USP Ethotoin RS in the Standard preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.


