Dipivefrin Hydrochloride
If you find any inaccurate information, please let us know by providing your feedback here
Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
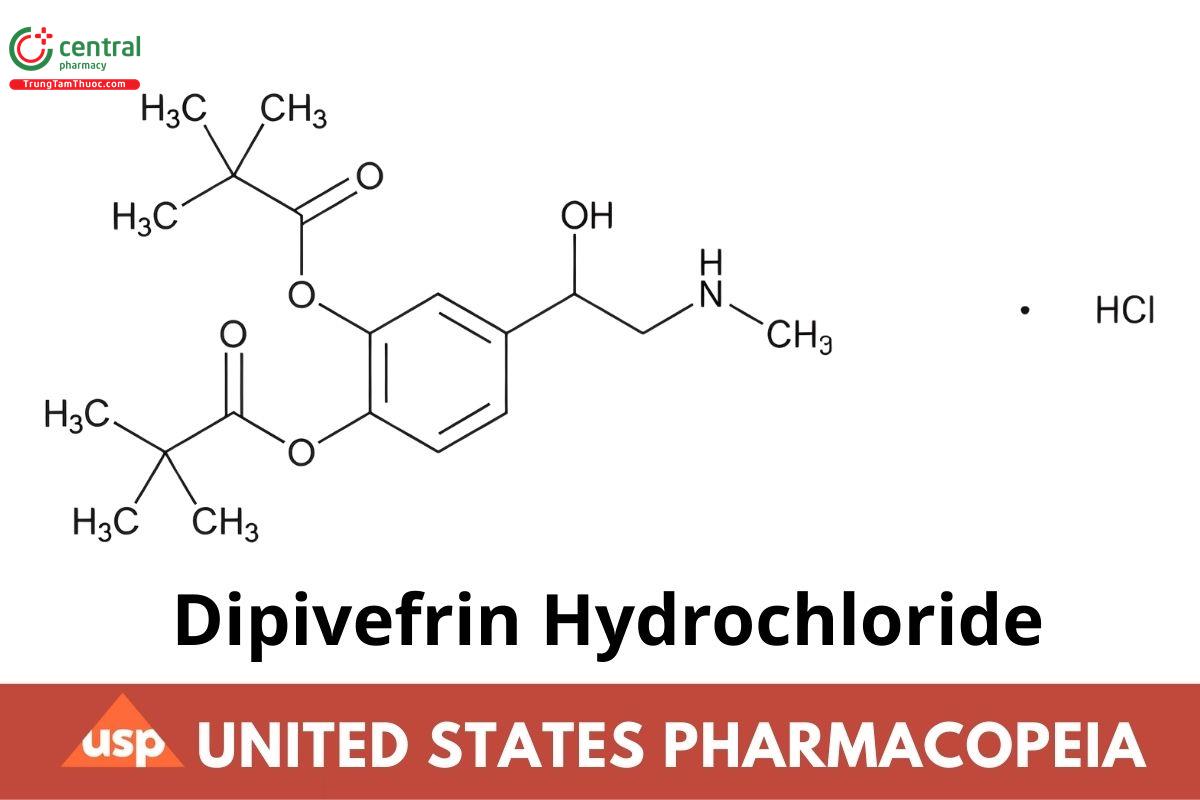
C19H29NO5 · HCI 387.90
Propanoic acid, 2,2-dimethyl-, 4-[1-hydroxy-2-(methylamino) ethyl]-1,2-phenylene ester, hydrochloride, (±);
(±)-3,4-Dihydroxy-a-[(methylamino) methyl benzyl alcohol 3,4-dipivalate hydrochloride CAS RN®: 64019-93-8; UNII: 5QTH9UHV0K.
1 DEFINITION
Dipivefrin Hydrochloride contains NLT 98.5% and NMT 101.5% of dipivefrin hydrochloride (C19H29NO5 · HCI), calculated on the dried basis.
2 IDENTIFICATION
A. SPECTROSCOPIC IDENTIFICATION TESTS (197), Infrared Spectroscopy: 197K
B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
C. IDENTIFICATION TESTS GENERAL, Chloride (191).
Sample solution: 10 mg/mL of Dipivefrin Hydrochloride
Acceptance criteria: Meets the requirements
3 ASSAY
3.1 PROCEDURE
Mobile phase: Acetonitrile, 0.014 M sodium dodecyl sulfate, and glacial acetic acid (24:15:1)
Standard solution: 5 mg/mL of USP Dipivefrin Hydrochloride RS in 0.0015 N hydrochloric acid
Sample solution: 5 mg/mL of Dipivefrin Hydrochloride in 0.0015 N hydrochloric acid
3.1.1 Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 254 nm
Column: 4.6-mm x 25-cm; packing L1
Flow rate: 2 mL/min
Injection volume: 20 µL
3.1.2 System suitability
Sample: Standard solution
3.1.3 Suitability requirements
Tailing factor: NMT 1.5
Relative standard deviation: NMT 0.6%
3.1.4 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of dipivefrin hydrochloride (C19H29NO5 · HCI) in the portion of Dipivefrin Hydrochloride taken:
Result = (rU/rS) x (CS/CU) × 100
rU = peak response from the Sample solution r_{u} = 1
rS = peak response from the Standard solution s
CS = concentration of USP Dipivefrin Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Dipivefrin Hydrochloride in the Sample solution (mg/mL)
Acceptance criteria: 98.5%-101.5% on the dried basis
4 IMPURITIES
4.1 RESIDUE ON IGNITION (281): NMT 0.3%
Change to read:
4.2 IRON (241), Procedures. Procedure 1 ▲(CN 1-JUN-2023)
Standard iron solution: Use the Standard Iron Solution prepared as directed in the chapter.
Hydroxylamine solution: 100 mg/mL of hydroxylamine hydrochloride in water
Triazine solution: 1.25 mg/mL of 2,4,6-tri-(2-pyridyl)-S-triazine in methanol
Standard solution: Into a 50-mL color-comparison tube pipet 1 mL of Standard iron solution, add 42.0 mL of water, and mix.
Sample solution: Into a 50-mL color-comparison tube add 2.0 g of Dipivefrin Hydrochloride, 43.0 mL of water, and mix.
Analysis: To each of the tubes containing the Standard solution and the Sample solution add 5.0 mL of Hydroxylamine solution and 2.0 mL of Triazine solution, and mix.
Acceptance criteria: NMT 5 ppm; the color of the solution from the Sample solution is not darker than that of the solution from the Standard solution.
4.3 LIMITS OF EPINEPHRINE AND ADRENALONE
Protect the Standard solution and the Sample solution from light.
Solution A: 0.1% (v/v) Anhydrous formic acid in water
Solution B: Acetonitrile and methanol (60:40)
Mobile phase: See Table 1. Return to the original conditions and re-equilibrate the system.
Table 1
| Time (min) | Solution A (%) | Solution B (%) |
| 0 | 100 | 0 |
| 3 | 100 | 0 |
| 5 | 40 | 60 |
| 10 | 40 | 60 |
Diluent: 0.01 M hydrochloric acid
Standard solution: 0.02 mg/mL of USP Epinephrine Bitartrate RS and 0.01 mg/mL of USP Adrenalone Hydrochloride RS in Diluent.
Sample solution 10.0 mg/mL of Dipivefrin Hydrochloride in Diluent
4.3.1 Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 260 nm
Column: 4.6-mm x 15-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 10 µL
4.3.2 System suitability
Sample: Standard solution
4.3.3 Suitability requirements
Resolution: NLT 2.0 between epinephrine and adrenalone
Relative standard deviation: NMT 5%
4.3.4 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of epinephrine in the portion of Dipivefrin Hydrochloride taken:
Result = (rU/rS) x (CS/CU) × (Mr1/Mr2) × 100
rU = peak response of epinephrine from the Sample solution
rS = peak response of epinephrine from the Standard solution s
CS = concentration of USP Epinephrine Bitartrate RS in the Standard solution (mg/mL)
CU = concentration of Dipivefrin Hydrochloride in the Sample solution (mg/mL)
Mr1 = molecular weight of epinephrine, 183.20
Mr2 = molecular weight of epinephrine bitartrate, 333.29
Calculate the percentage of adrenalone in the portion of Dipivefrin Hydrochloride taken:
Result = (rU/rS) x (CS/CU) × (Mr1/Mr2) × 100
rU = peak response of adrenalone from the Sample solution
rS = peak response of adrenalone from the Standard solution
CS = concentration of USP Adrenalone Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Dipivefrin Hydrochloride in the Sample solution (mg/mL)
Mr1 = molecular weight of adrenalone, 181.19
Mr2 = molecular weight of adrenalone hydrochloride, 217.65 12
Acceptance criteria: See Table 2.
Table 2
| Name | Relative Retention Time | Relative Retention Time |
| Epinephrinea,b | 1.0 | 0.1 |
| Adrenalonec | 1.4 | 0.1 |
a 4-1[-Hydroxy-2-(methylamino) ethylbenzene-1,2-diol.
b This impurity is a racemate of epinephrine (also known as racepinephrine or [±] adrenaline).
c 3',4'-Dihydroxy-2-(methylamino)acetophenone.
5 ORGANIC IMPURITIES
Solution A: Acetonitrile and methanol (60:40)
Solution B: 2.7 g/L of ammonium hydroxide, adjusted with 2 M acetic acid to a pH of 10.0
Mobile phase: Solution A and Solution B (55:45)
Diluent: Solution A and 0.01 M hydrochloric acid (55:45)
System suitability solution: 10 mg/mL of USP Dipivefrin Hydrochloride RS and 10 µg/mL of USP Dipivefrin Related Compound ERS in Diluent
Standard solution: 10 µg/mL each of USP Dipivefrin Hydrochloride RS and USP Dipivefrin Related Compound E RS in Diluent
Sample solution: 10 mg/mL of Dipivefrin Hydrochloride in Diluent
4.3.5 Chromatographic system
(See Chromatography (621), System Suitability.)
Mode: LC
Detector: UV 260 nm
Column: 4.6-mm x 15-cm; 5-µm packing L1
Flow rate: 1 mL/min
Injection volume: 10 µL
Run time: 2.5 times the retention time of dipivefrin
4.3.6 System suitability
Samples: System suitability solution and Standard solution
4.3.7 Suitability requirements
Resolution: NLT 3.0 between dipivefrin and dipivefrin related compound E, System suitability solution
Relative standard deviation: NMT 5% for the dipivefrin peak, Standard solution
4.3.8 Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of dipivefrin related compound E in the portion of Dipivefrin Hydrochloride taken:
Result = (rU/rS) x (CS/CU) × 100
rU = peak response of dipivefrin related compound E from the Sample solution
rS = peak response of dipivefrin related compound E from the Standard solution
CS = concentration of USP Dipivefrin Related Compound E RS in the Standard solution (mg/mL)
CU = concentration of Dipivefrin Hydrochloride in the Sample solution (mg/mL)
Calculate the percentage of any other individual impurity in the portion of Dipivefrin Hydrochloride taken:
Result = (rU/rS) x (CS/CU) × (1/F) x 100
rU = peak response of each impurity from the Sample solution
rS = peak response of dipivefrin from the Standard solution
CS = concentration of USP Dipivefrin Hydrochloride RS in the Standard solution (mg/mL)
CU = concentration of Dipivefrin Hydrochloride in the Sample solution (mg/mL)
F = relative response factor (see Table 3)
Acceptance criteria: See Table 3. Disregard any impurity peak less than 0.05%.
Table 3
| Name | Relative Retention Time | Relative Response Factor | Acceptance Criteria, NMT (%) |
5-Pivaloyl racepinephrinea and 4-Pivaloyl racepinephrineb | 0.4c | 2.0 | 0.3 |
| Dipivefrin | 1.0 | — | — |
| Dipivefrin related compound Ed | 1.3 | — | 0.1 |
| N-Ethyl dipivefrine | 2.0 | 1.0 | 0.1 |
| Unspecified impurity | — | 1.0 | 0.10 |
| Total impurities | — | — | 0.5 |
a 2-Hydroxy-5-[1-hydroxy-2-(methylamino) ethyl]phenyl pivalate.
b 2-Hydroxy-4-[1-hydroxy-2-(methylamino)ethyl]phenyl pivalate.
c 5-Pivaloyl racepinephrine and 4-Pivaloyl racepinephrine coelutes.
d 4-[(Methylamino) acetyl]-1,2-phenylene dipivalate.
e 4-(2-[Ethyl(methyl)amino]-1-hydroxyethyl)-1,2-phenylene dipivalate.
6 SPECIFIC TESTS
LOSS ON DRYING (731)
Analysis: Dry in a suitable vacuum drying tube over phosphorus pentoxide at 60° for 6 h.
Acceptance criteria: NMT 1.0%
7 ADDITIONAL REQUIREMENTS
PACKAGING AND STORAGE: Preserve in tight containers.
USP REFERENCE STANDARDS (11).
USP Adrenalone Hydrochloride RS
3',4'-Dihydroxy-2-(methylamino)acetophenone hydrochloride.
C9H11NO3 · HCI 217.65
USP Dipivefrin Hydrochloride RS
USP Digivefrin Related Compound ERS
4-[(Methylamino)acetyl)-1,2-phenylene dipivalate hydrochloride.
C19H27NO5 · HCI 385.88
USP Epinephrine Bitartrate RS C9H13NO3 · H4C6O6 333.29


