Determination of Zeta Potential by Electrophoretic Light Scattering
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
The zeta potential determination test method is based on the International Organization for Standardization (ISO) 13099-2:2012, Colloidal systems—Methods for zeta-potential determination—Part 2: Optical methods, as well as the scientific and technological developments that followed.
Zeta potential, denoted by the Greek letter ζ (hence the name), is a physicochemical characteristic of colloidal systems (suspensions and emulsions) that describes the electric potential difference between the mobile dispersion medium and the stationary layer of the dispersion medium attached to the dispersed particle. It is expressed in units of millivolts (mV). Zeta potential is often used to infer surface properties and to assess colloidal stability. Zeta potential cannot be directly measured but may be derived from experimentally measurable properties, including electroacoustic, electrokinetic, and electrophoretic phenomena.
This chapter provides procedures for the measurement of electrophoretic mobility based on light scattering phenomena, method development, and control of instrument performance. It does not address specific requirements of zeta potential measurement, or the acceptance value for particular products.
2 PRINCIPLE
A representative sample dispersed at an adequate concentration in a suitable dispersion medium is placed in a cell equipped with a pair of electrodes to apply an electric potential. Although cell configuration can vary considerably, typically cells consist either of a cylindrical or rectangular capillary with electrodes at both ends, a cuvette, or another type of vessel into which a pair of electrodes are inserted at a known fixed distance. The cell is illuminated by a monochromatic coherent light beam, usually generated by a laser. Because of the applied electric potential, the particles that bear a net electric charge are attracted toward the oppositely charged electrode, a process known as electrophoresis. In the case of charged capillary walls, the dispersion medium stream flows along the capillary wall, a process known as electroosmosis. The overall movement of particles in the cell is the result of both processes. The electrophoretic light scattering technique uses a photoelectric detector to measure the Doppler shift (observed change in the frequency of a wave when the measuring apparatus and the wave source are in relative motion) of the scattered light. The Doppler shift is used to determine the velocity and direction of particle movement from which the electrophoretic mobility (µ) is calculated. Zeta potential is derived from the electrophoretic mobility using the Henry function, which can be approximated by the Smoluchowski equation or Hückel equation according to the relative thickness of the electrical double layer compared to the hydrodynamic radius of the particle.
For discussion of the theory and principles of measurements, see Analytical Methodologies Based on Scattering Phenomena— Electrophoretic Light Scattering (Determination of Zeta Potential) 〈1430.4〉, which is a helpful, but not mandatory resource.
Change to read:
3 INSTRUMENT
The instrument should be located in a controlled environment with reduced dust, (USP 1-May-2023) electrical noise, vibrations, temperature fluctuations, humidity, and direct light. The instrument should contain a rigid internal optical system fixed on a bench with a vibration damp setup, and should be installed on a rigid table or bench.
An example of a setup of a zeta potential instrument is given in 〈1430.4〉 , Figure 1. Other possibilities for optical arrangements are shown in 〈1430.4〉 , Figure 2.
As shown in those figures, a typical zeta potential measurement instrument is comprised of the following components:
1. A sample cell to contain the test suspension between two electrodes so that an electric field can be applied
2. A suitable laser to illuminate the particles within the sample
3. A suitable detector to capture the scattered light and Doppler shifts in the scattered light when the electric field is applied
4. A software and computer system to process the collected scattering data and compute the mobility and zeta potential using appropriate models and algorithms
Typically, the Doppler shifts that occur during electrophoresis are quite small (ranging from a few to several hundred Hertz) and cannot be measured directly. To overcome this issue, interferometers are used to combine light that is frequency shifted by the sample with a reference beam. When the scattered light and the reference beam interfere, the result is a beat. The frequency of the beat is the frequency difference between the two underlying beams, which is the Doppler shift induced by the particles.
In practice, however, a simple interferometer cannot determine the sign of the Doppler shift and thus cannot determine the sign of the zeta potential. The measured beat frequency is always positive, whether the Doppler shift is positive or negative.
To overcome this problem, modern zeta potential instruments use a frequency modulator in the reference beam. This modulator causes a controlled Doppler shift at the frequency of the reference light. This means that the measured Doppler shift is the sum of the Doppler shift induced by the modulator plus the Doppler shift induced by the motion of the particles. Thus, the use of a frequency modulator enables the determination of the sign (positive or negative voltage) of the zeta potential.
Change to read:
4 DEVELOPMENT OF THE METHOD
4.1 Sample Considerations—Effect of Physicochemical Parameters of the Colloidal System on the Zeta Potential
4.1.1 PH
Depending on particle surface chemistry, a change of the dispersion's pH affects the zeta potential. When the pH changes, the ion concentration changes, and it affects the composition of the double layer on the surface of the particles. This, in turn, changes the electrophoretic mobility and the resulting zeta potential.
4.1.2 CONDUCTIVITY
The conductivity of the dispersion should be appropriate to avoid (USP 1-May-2023) heating and electrolysis. (USP 1-May-2023) It is recommended to use a 10 mM saline or similar media. High conductivity media may result in increased Joule–Thomson heating, and thus a reduced voltage should be used. Typically, the conductivity should be within 1–5 millisiemens per cm (mS/cm).
4.1.3 CONCENTRATION RANGE OF THE COLLOIDAL SYSTEM
The effect of the dispersed material concentration on the zeta potential must be evaluated, and the concentration range of a constant zeta potential established (see Determination of the Concentration Range).
4.1.4 CONCENTRATION OF OTHER ADDED COMPONENTS IN THE SYSTEM
The effects of other system components that need to be part of the colloidal system in a particular application must be evaluated.
4.1.5 VISCOSITY
The electrophoretic mobility of the moving particles is related to the dispersion medium viscosity as shown in 〈1430.4〉 , Equation 3. The dispersion medium's viscosity should preferably be lower than 10 mPa · s, so that the system does not deviate from Newtonian fluid. For high concentration dispersions, the viscosity of the dispersion medium is used, and consideration is given to other factors.
4.2 Sample Preparation
SAMPLE STABILITY CONSIDERATIONS
The following two aspects of sample stability are considered when performing zeta potential measurements:
1. Based on the nature of the dispersion being tested, the electrodes in the sample cell and the dispersion itself may change after a zeta potential measurement is performed. Therefore, when reusing the same sample volume or cell for replicate zeta potential testing, the data should be reviewed carefully to ensure the measurements are acceptable. With certain dispersions, a fresh dispersant aliquot may need to be tested for each replicate measurement.
2. The zeta potential determination of a concentrated dispersion may require further dilution of the dispersion using filtered dispersion
medium prior to the measurement, in order to lower the particle concentration, and to avoid measurement artifacts due to multiple scattering or inter-particle interactions. It is important to ensure that the diluting dispersion medium has the same electrolyte composition and pH as the dispersion being tested, especially when the goal of the study is to understand the formulation properties of that particular medium. However, the resulting conductivity of the media needs to be within the workable range as shown under Conductivity. If the diluted dispersion is prepared well before measurement, the stability of this diluted dispersion should be evaluated by measuring the zeta potential over the expected measurement time, to ensure that there is no time-dependent behavior on the measured zeta potential. Testing of serial dilutions bracketing the expected target dilution factor may also be useful.
4.3 Determination of the Concentration Range
A reliable measurement requires reaching an acceptable signal-to-noise ratio in the detector. To achieve this feat, the dispersed material concentration in the colloidal system being measured must exceed a certain level. Likewise, it must be below a maximum level in order to avoid multiple scattering and ▲colloidal particles▲ (USP 1-May-2023) interaction. The concentration range is influenced by the width of the laser beam, the path length between the electrodes, the optical properties of the materials, and the sensitivity of the detector. Therefore, measurements must be performed at different dispersion concentrations to determine the appropriate concentration range for any specific sample being tested.
4.4 Method Validation
Typically, the validity of an analytical procedure is assessed by the evaluation of its specificity, linearity, range, accuracy, precision, robustness, and quantitation limit (QL) or detection limit (DL) as needed (see Validation of Compendial Procedures 〈1225〉). In zeta potential analysis by electrophoretic light scattering, specificity as defined by the International Council for Harmonisation (ICH) is not applicable because it is not possible to discriminate different components of a sample. The range, linearity, DL, and QL as defined in ICH Q2 guidelines and 〈1225〉 are also not applicable. Exploring a linear relationship between concentration and response, or a mathematical model for interpolation is not applicable to this procedure since Zeta potential should be a constant value, and should be constant for the optimized concentration range. The DL and QL are also not applicable because the concentration range of the method for the zeta potential measurement represents a different concept from the minimum concentration above which the property could be detected or quantified.
Rather than evaluating linearity, DL, and QL, this method requires the definition of a concentration range and measurement conditions within which the result of the measurements does not vary significantly. Concentrations below that range produce an error due to poor signal-to-noise ratio, while concentrations above that range produce an error due to multiple scattering and colloidal particles (USP 1-May-2023) interaction (e.g., flocculation, aggregation). Evaluation of accuracy is not applicable to the actual sample, but it should be confirmed through an appropriate verification of instrument calibration using appropriate reference materials (see Verification of Calibration). Precision may be assessed by means of a repeatability determination and intermediate precision determination (on different days or with different analysts) using the same instrument.
The feasible repeatability of the method depends mainly on the characteristics of the material and dispersion medium, whereas the required repeatability depends on the purpose of the measurement. Mandatory limits cannot be specified in this chapter, as repeatability (different sample preparations) may vary appreciably from one substance to another. However, it is good practice to aim for a variance lower than 10% as acceptance criteria for repeatability, and lower than 15% for intermediate precision.
The robustness assessment should address the sensitivity of the method to other likely sources of variation, and should establish a set of acceptable measurement conditions (see Measurement Procedure) and sample considerations (see Sample Considerations—Effect of Physicochemical Parameters of the Colloidal System on the Zeta Potential).
5 MEASUREMENT PROCEDURE
5.1 Precautions
The instructions provided in the instrument manual should be followed:
Never look into the direct path of the laser beam or its reflections
Check the instrument set-up (e.g., warm-up time, required measuring range and lens position, angle of analysis, temperature equilibration)
Avoid air bubbles, evaporation of liquid, or other inhomogeneities in the suspension (emulsion) being tested
5.2 Dispersion Media
It is critical to ensure that the sample and the sample cell/cuvette are compatible with the dispersion medium. The electrophoretic mobility of the sample is dependent on the chemical characteristics of the dispersion medium. Specifically, the pH and the ion type and concentration need to be controlled if the sample is diluted prior to measurement.
5.3 Measurement Cell
To avoid contamination, the measurement cell must be thoroughly rinsed prior to and after measurement. It is important to control and monitor the temperature of the measurement cell because the dispersion medium viscosity is a function of temperature, and has a significant impact on the measured results.
5.4 Sample Inspection
It is important to visually inspect the sample prior to analysis to check for large particles or sedimentation, which can negatively impact the results and/or reproducibility.
5.5 Preparation
It is important to avoid changing the electrophoretic mobility of the sample during the sample preparation process. Many materials such as pipettes, spatulas, or syringes may have ions on their surface that can contaminate the sample. Disposable plastic materials are recommended if they are chemically compatible with the sample.
5.6 Measurement of the Light Scattering of Dispersed Sample(s)
REPLICATES
The number of replicate measurements (with individual sample preparations) to be performed depends on the required measurement precision. It is recommended to set this number in a substance-specific method. Typically, three consecutive measurements are performed.
6 DATA PROCESSING AND INTERPRETATION
Data processing is done by spectrum analysis, or autocorrelation function, or modulated Brownian motion power spectrum method. For a more detailed information on data processing, see 〈 1430.4〉, 5. Data Analysis.
6.1 Conversion of Doppler Shifts into Electrophoretic Mobility
The relationship between observed Doppler frequency shifts (Δω) of scattered light and corresponding electrophoretic mobility, µ, is shown in Equation 1 and Equation 2 for a reference beam optic alignment and a cross beam optic alignment, respectively:
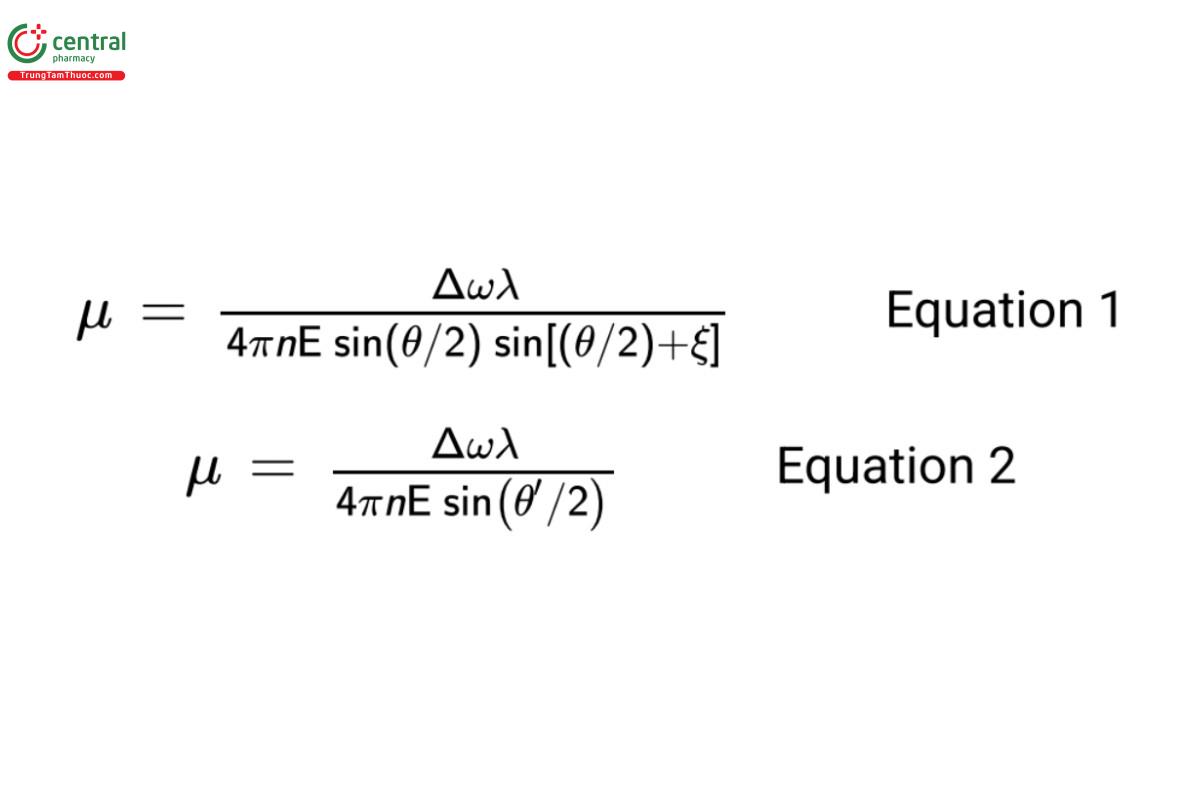
6.2 Calculation of Zeta Potential
For large solute particles (the hydrodynamic radius is large compared to the thickness of the electrical double layer), the Smoluchowski equation is used to calculate ζ from µ:

For small solute particles (the hydrodynamic radius is small compared to the thickness of the electrical double layer), the Hückel equation is used to calculate ζ from µ:
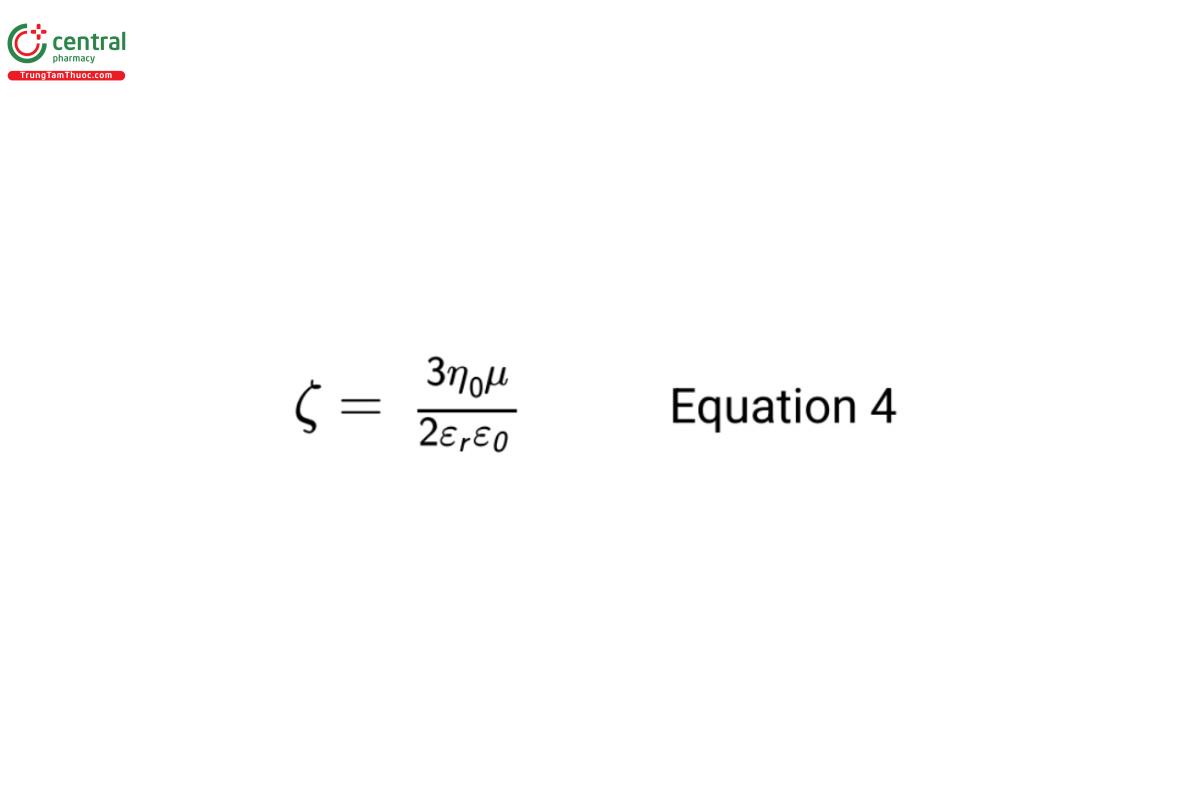
For more detailed information on these equations, see 〈 1430.4〉, 5.2 Calculation of Zeta Potential.
Change to read:
7 CONTROL OF THE INSTRUMENT PERFORMANCE
Use the instrument according to the manufacturer’s instructions and carry out the prescribed qualifications at an appropriate frequency for the use of the instrument and substances to be tested.
7.1 Verification of Calibration
The method is based on first principles and no calibration is needed. Verification is usually done with a standard sample.▲▲ (USP 1-May- 2023) In general, it is not necessary to do a standard measurement each time a sample is being measured. Rather, it is recommended to run the standard sample "regularly" at predefined intervals.
7.2 Qualification of the System
The performance of the instrument must be qualified at regular time intervals or as frequently as appropriate. This can be achieved using any suitable standard sample as mentioned in the previous paragraph.
The qualification of the system is based on the concept that the equipment, electronics, software, and analytical operations constitute an integral system, which can be evaluated as an entity. Thus, the entire measurement procedure is examined, including sample collection, sample dispersion, sample transport through the measuring zone, and the measurement and deconvolution procedure. It is essential that the total operational procedure is fully described.
An instrument meets the requirement if the coefficient of variation for the mean electrophoretic mobility values from each measurement for a standard sample is less than 10%, provided that the absolute value of the standard sample mobility is higher than 2 × 10−8 m2/V · s.


