Arsenic
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
Add the following:
1 INTRODUCTION
This chapter describes four analytical procedures for the evaluation of the levels of arsenic (As). Use Procedure 1, Procedure 2, Procedure 3, or Procedure 4 as indicated in the individual monograph. Procedure 3 or Procedure 4 can be used in all circumstances, provided that suitability is demonstrated by meeting the Requirements for Procedure Validation. (Official 1-Jun-2023)
Change to read:
2 PROCEDURES
2.1 PROCEDURE 1 AND PROCEDURE 2
Both Procedure 1 and Procedure 2 are (Official 1-Jun-2023) designed to determine the presence of trace amounts of arsenic by converting the arsenic in a substance under test to arsine, which is then passed through a solution of silver diethyldithiocarbamate to form a red complex. The red color so produced is compared, either visually or spectrophotometrically, to the color produced similarly in a control containing an amount of arsenic equivalent to the limit given in the individual monograph. Limits are stated in terms of arsenic. The content of arsenic does not exceed the limit given in the individual monograph.
These procedures differ (Official 1-Jun-2023) only in the preliminary treatment of the test substance and the standard. Generally,
Procedure 1 (Official 1-Jun-2023) is used for inorganic materials, and Procedure 2 (Official 1-Jun-2023) is used for organic materials.
Apparatus
The apparatus (see Figure 1) consists of an arsine generator (a) fitted with a scrubber unit (c) and an absorber tube (e) with standard-taper or ground glass ball-and-socket joints (b and d) between the units. However, any other suitable apparatus embodying the principle of the assembly described and illustrated may be used.
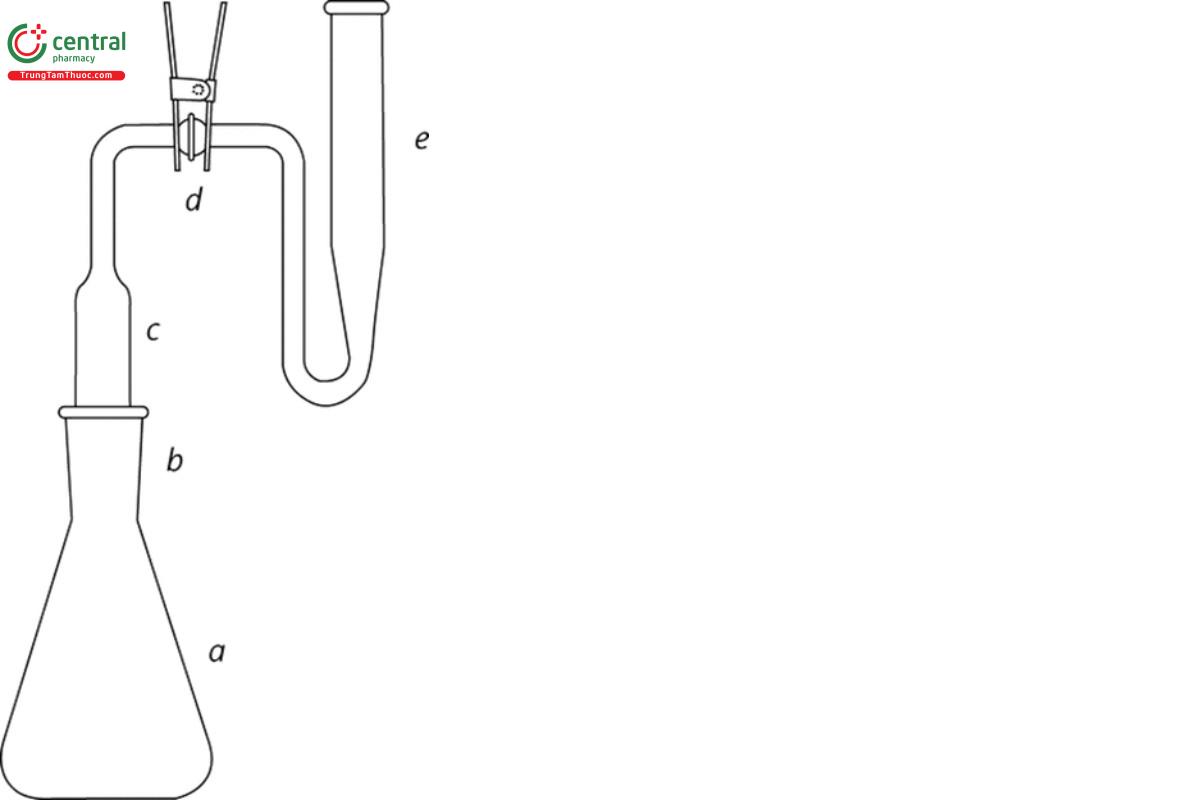
Arsenic trioxide stock solution: Dissolve 132.0 mg of arsenic trioxide, previously dried at 105° for 1 h and accurately weighed, in 5 mL of sodium hydroxide solution (1 in 5) in a 1000-mL volumetric flask. Neutralize the solution with 2 N sulfuric acid, add 10 mL more of 2 N sulfuric acid, then add recently boiled and cooled water to volume, and mix.
Standard arsenic solution: Transfer 10.0 mL of Arsenic trioxide stock solution to a 1000-mL volumetric flask, add 10 mL of 2 N sulfuric acid, then add recently boiled and cooled water to volume, and mix. Each milliliter of Standard arsenic solution contains the equivalent of 1 µg of arsenic. Keep this solution in an all-glass container, and use within 3 days.
Procedure 1: Colorimetry (Official 1-Jun-2023)
Standard preparation: Pipet 3.0 mL of Standard arsenic solution into a generator flask, and dilute with water to 35 mL.
Test preparation: Unless otherwise directed in the individual monograph, prepare as follows. Transfer to the generator flask the quantity, in grams, of the test substance calculated as follows:
Result = 3.0/L
L = limit of arsenic (ppm)
Dissolve in water and dilute with water to 35 mL.
Analysis: (Official 1-Jun-2023) Treat the Standard preparation and the Test preparation similarly as follows. Add 20 mL of 7 N sulfuric acid, 2 mL of potassium iodide TS, 0.5 mL of stronger acid stannous chloride TS, and 1 mL of isopropyl alcohol, and mix. Allow to stand at room temperature for 30 min. Pack the scrubber tube (c) with two pledgets of cotton that have been soaked in saturated lead acetate solution, freed from excess solution by expression, and dried in vacuum at room temperature, leaving a 2-mm space between the two pledgets. Lubricate the joints (b and d) with a suitable stopcock grease designed for use with organic solvents, and connect the scrubber unit to the absorber tube (e). Transfer 3.0 mL of silver diethyldithiocarbamate TS to the absorber tube. Add 3.0 g of granular zinc (#20 mesh) to the mixture in the flask, immediately connect the assembled scrubber unit, and allow the evolution of hydrogen and the color development to proceed at room temperature for 45 min, swirling the flask gently at 10-min intervals. Disconnect the absorber tube from the generator and scrubber units, and transfer the absorbing solution to a 1-cm absorption cell. Any red color produced by the Test preparation does not exceed that produced by the Standard preparation. If necessary or desirable, determine the absorbance at the wavelength of maximum absorbance between 535 and 540 nm, with a suitable spectrophotometer or colorimeter, using silver diethyldithiocarbamate TS as the blank.
Interfering chemicals: Metals or salts of metals, such as chromium, cobalt, copper, mercury, molybdenum, nickel, palladium, and silver,
may interfere with the evolution of arsine. Antimony, which forms stibine, produces a positive interference in the color development with silver diethyldithiocarbamate TS; when the presence of antimony is suspected, the red colors produced in the two silver diethyldithiocarbamate solutions may be compared at the wavelength of maximum absorbance between 535 and 540 nm, with a suitable colorimeter, since at this wavelength the interference due to stibine is negligible.
Procedure 2: Colorimetry (Official 1-Jun-2023)
[CAUTION—Some substances may react with explosive violence when digested with hydrogen peroxide. Exercise safety precautions at all times.]
[NOTE—If halogen-containing compounds are present, use a lower temperature while heating the test specimen with sulfuric acid, avoid boiling the mixture, and add the Hydrogen peroxide with caution, before charring begins, to prevent loss of trivalent arsenic.]
[NOTE—If the test substance reacts too rapidly and begins charring with 5 mL of sulfuric acid before heating, use instead 10 mL of cooled dilute sulfuric acid (1 in 2), and add a few drops of the hydrogen peroxide before heating.]
Standard preparation: Pipet 3.0 mL of Standard arsenic solution into a generator flask, add 2 mL of sulfuric acid, mix, and add the total amount of 30 percent hydrogen peroxide used in preparing the Test preparation. Heat the mixture to strong fuming, cool, cautiously add 10 mL of water, and again heat to strong fumes. Repeat this procedure with another 10 mL of water to remove any traces of hydrogen peroxide. Cool, and dilute with water to 35 mL.
Test preparation: Unless otherwise directed in the individual monograph, transfer to a generator flask the quantity, in grams, of the test substance calculated as follows:
Result = 3.0/L
L = limit of arsenic (ppm)
Add 5 mL of sulfuric acid and a few glass beads, and digest in a fume hood, preferably on a hot plate and at a temperature not exceeding 120°, until charring begins. (Additional sulfuric acid may be necessary to wet some specimens completely, but the total volume added should not exceed 10 mL.) Cautiously add, dropwise, 30 percent hydrogen peroxide, allowing the reaction to subside and again heating between drops. Add the first few drops very slowly with sufficient mixing, in order to prevent a rapid reaction. Discontinue heating if foaming becomes excessive. When the reaction has abated, heat cautiously, rotating the flask occasionally to prevent the specimen from caking on glass exposed to the heating unit. Maintain oxidizing conditions at all times during the digestion by adding small quantities of the hydrogen peroxide solution whenever the mixture turns brown or darkens. Continue the digestion until the organic matter is destroyed, gradually raising the temperature of the hot plate until fumes of sulfur trioxide are copiously evolved, and the solution becomes colorless or retains only a light straw color. Cool, add cautiously 10 mL of water, mix, and again evaporate to strong fuming, repeating this procedure to remove any trace of hydrogen peroxide. Cool, cautiously add 10 mL of water, wash the sides of the flask with a few milliliters of water, and dilute with water to 35 mL.
Analysis: (Official 1-Jun-2023) Proceed as directed for Analysis in Procedure 1. (Official 1-Jun-2023)
Interfering chemicals: See Interfering chemicals in Procedure 1.
2.2 PROCEDURE 3 AND PROCEDURE 4
Both Procedure 3 and Procedure 4 are ICP-based procedures and can be used for the determination of arsenic. Procedure 3 can be used for the determination of arsenic by inductively coupled plasma atomic (or optical) emission spectroscopy (ICP–AES or ICP–OES).
Procedure 4 can be used for the determination of arsenic by ICP–MS.
Before initial use, the analyst should verify that the procedure is appropriate for the instrument and sample used (procedural verification) by meeting the Requirements for Procedure Validation.
Where a monograph specifies a limit for arsenic concentration, the value listed in the monograph should be used as the J value for the purposes of this test.
System standardization and suitability evaluation using applicable reference materials should be performed on the day of analysis. Sample preparation: Forms of sample preparation include neat, direct aqueous solution, direct organic solution, and indirect solution. The selection of the appropriate sample preparation depends on the material under test and is the responsibility of the analyst. When a sample preparation is not indicated in the monograph, an analyst may use any appropriately validated preparation procedure. In cases where spiking of a material under test is necessary to provide an acceptable signal intensity, the blank should be spiked with arsenic using, where possible, the same spiking solution. [NOTE—All liquid samples should be weighed.]
Closed vessel digestion: This sample preparation procedure is designed for samples that must be digested in a concentrated acid using a closed vessel digestion apparatus. Closed vessel digestion minimizes the loss of volatile impurities. The choice of a concentrated acid depends on the sample matrix. The use of any of the concentrated acids may be appropriate, but each introduces inherent safety risks. Therefore, appropriate safety precautions should be used at all times. [NOTE—Weights and volumes provided may be adjusted to meet the requirements of the digestion apparatus used.]
An example procedure that has been shown to have broad applicability is as follows. Dehydrate and predigest 0.5 g of the primary sample in 5 mL of freshly prepared concentrated acid. Allow to sit loosely covered for 30 min in a fume hood. Add an additional 10 mL of concentrated acid, and digest using a closed vessel technique until digestion or extraction is complete. Repeat, if necessary, by adding an additional 5 mL of concentrated acid. [NOTE—Follow the manufacturer’s recommended procedures to ensure safe use.]
Reagents: All reagents used for the preparation of sample and standard solutions should be free of elemental impurities, in accordance with Plasma Spectrochemistry 〈730〉.
Procedure 3: ICP–OES
Standardization solution 1: 1.5J of arsenic in a matched matrix Standardization solution 2: 0.5J of arsenic in a matched matrix
Sample stock solution: Prepare as directed in Sample preparation. Allow the sample to cool, if necessary.
Sample solution: Dilute the Sample stock solution with an appropriate solvent to obtain a final arsenic concentration of not more than 1.5J. Blank: Matched matrix
Elemental spectrometric system
(See 〈730〉.) Rinse: Use diluent.
Standardization: Standardization solution 1, Standardization solution 2, and Blank System suitability
Sample: Standardization solution 1
Suitability requirements
Drift: Compare results obtained from Standardization solution 1 before and after the analysis of the Sample solution.
Suitability criteria: Not more than 20% for arsenic. [NOTE—If samples are high in mineral content, rinse the system well before introducing the Sample in order to minimize carryover.]
Analysis: Analyze according to the manufacturer's suggestions for program and wavelength. Calculate and report results on the basis of
the original sample size. [NOTE—Appropriate measures must be taken to correct for matrix-induced interferences (e.g., wavelength overlaps).]
Procedure 4: ICP–MS
Follow Procedure 3 except for Detector and Analysis.
[NOTE—An instrument with a cooled spray chamber is recommended. (A collision cell or reaction cell may also be beneficial.)] Detector: Mass spectrometer
Analysis: Analyze according to the manufacturer's suggestions for program and mass-to-charge ratio. Calculate and report results based on the original sample size. [NOTE—Appropriate measures must be taken to correct for matrix-induced interferences (e.g., argon chloride interference with arsenic determinations).] (Official 1-Jun-2023)
Add the following:
3 REQUIREMENTS FOR PROCEDURE VALIDATION
The following section defines the validation parameters and the acceptance criteria for performance-based procedures. Meeting these requirements must be demonstrated experimentally using an appropriate system suitability procedure and reference materials. Any alternative procedure (e.g., an atomic-absorption-based procedure) that has been validated and meets the acceptance criteria that follow is considered to be suitable for use.
Meeting these validation acceptance criteria is sufficient to demonstrate that the procedure will produce comparable results to those obtained using the procedure prescribed in the monograph.
3.1 ACCURACY
Standard solutions: Prepare solutions containing arsenic at concentrations ranging from 50% to 150% of J using appropriate reference materials.
Test samples: Spike the material under test with the appropriate reference materials before any sample preparation steps (digestion or solubilization). Prepare three replicate samples at concentrations ranging from 50% to 150% of J for arsenic.
Acceptance criteria
Spike recovery: 70%–150% for the mean of three replicate preparations at each concentration
3.2 PRECISION
Repeatability
Test samples: Six independent samples of material under test (taken from the same lot) spiked with appropriate reference materials for arsenic at the indicated concentration
Acceptance criteria
Relative standard deviation: Not more than 20% (N = 6) for arsenic Intermediate Precision (Ruggedness)
Analysis: Perform the Repeatability analysis again on a different day, with different instrumentation, with a different analyst, or a combination thereof. Combine the results of this analysis with the Repeatability analysis so the total number of analyses is 12.
Acceptance criteria
Relative standard deviation: Not more than 25% (N = 12) for arsenic
3.3 SPECIFICITY
The procedure must be able to unequivocally assess (see Validation of Compendial Procedures 〈1225〉) arsenic in the presence of components that may be expected to be present, including matrix components.
3.4 LIMIT OF QUANTITATION, RANGE, AND LINEARITY
Demonstrated by meeting the Accuracy requirement. (Official 1-Jun-2023)
Add the following:
4 GLOSSARY
Concentrated acid: Concentrated ultra-pure nitric, sulfuric, hydrochloric, or hydrofluoric acid or aqua regia.
Aqua regia: Aqua regia is a mixture of concentrated hydrochloric and nitric acids, typically at ratios of 3:1 or 4:1.
Matched matrix: Solutions having the same solvent composition as the Sample solution. In the case of an aqueous solution, a matched matrix would indicate that the same acids and acid concentrations are used in both preparations.
Target limit or target concentration: The acceptance value for the elemental impurity being evaluated, in this case arsenic. Where a monograph specifies a threshold limit, this shall become the target limit or target concentration of arsenic for the material. Exceeding the target limit indicates that a material under test exceeds the acceptable value. The determination of compliance is addressed in other chapters.
J: The concentration (w/w) of the element of interest, in this case arsenic, at the target limit, appropriately diluted to the working range of the instrument.
Appropriate reference materials: Where "appropriate reference materials" are specified in the chapter, certified reference materials (CRMs) from a national metrology institute (NMI), or reference materials that are traceable to the CRM of an NMI should be used. An example of an NMI in the United States is the National Institute of Standards and Technology (NIST). (Official 1-Jun-2023)


