〈1025〉 Pancreatin
If you find any inaccurate information, please let us know by providing your feedback here

Tóm tắt nội dung
This article is compiled based on the United States Pharmacopeia (USP) – 2025 Edition
Issued and maintained by the United States Pharmacopeial Convention (USP)
1 INTRODUCTION
Pancreatin is a pancreatic enzyme preparation containing amylase, protease, and lipase enzymes isolated from the pancreas gland of the hog, Sus scrofa L. var. domesticus Gray (Fam. Suidae). The pancreas is a secretory organ that plays a crucial role in the digestive process by producing bicarbonate to neutralize the acidic environment in the duodenum, hormones to regulate various catabolic functions, and a variety of digestive enzymes to degrade food in the small intestine. Pancreatin and pancreatin-containing medicinal products are used to aid digestion and absorption of food (carbohydrates, fat, and proteins) in patients with exocrine pancreatic insufficiency (EPI) caused by cystic fibrosis, chronic pancreatitis, and other conditions that might cause a deficiency in the secretion of pancreatic enzymes.
Pancreatic enzyme products (PEPs) of porcine origin have been marketed in the United States for the treatment of EPI before the enactment of the Federal Food, Drug, and Cosmetic Act of 1938. Supplemental pancreatic enzymes are available in prescription and non-prescription forms. Since 2004, the U.S. Food and Drug Administration (FDA) has required that all pancreatic enzyme drug products marketed in the United States obtain FDA approval via a New Drug Application (NDA). Although over-the-counter pancreatic enzyme products are available without a prescription, they are classified as dietary supplements rather than drugs. Pancreatin and Pancrelipase share similar functions and indications; however, pancrelipase, which is available only as a prescription drug, contains more of the active lipase enzyme and also more purified pancreatic extract than pancreatin does.
This chapter describes best practices related to the sourcing and manufacturing of pancreatin raw materials used in both pancreatin and pancrelipase drug products; these best practices help to ensure the safety and efficacy of the drug products made from this active pharmaceutical ingredient (API). A list of applicable regulatory guidance documents is provided in Appendix 1: Regulatory Bibliography.
2 PANCREAS COLLECTION AND PANCREATIN PRODUCTION
The animal-sourced raw material (pancreas glands) intended for pharmaceutical processing is a by-product of meat production and is collected in slaughterhouses that are approved by the national competent authority and inspected by the relevant veterinary authority. The animals from which pancreatin is derived must fulfill the requirements for the health of animals suitable for human consumption.
An example of a typical manufacturing process is summarized in the description and flow chart (Figure 1) below.
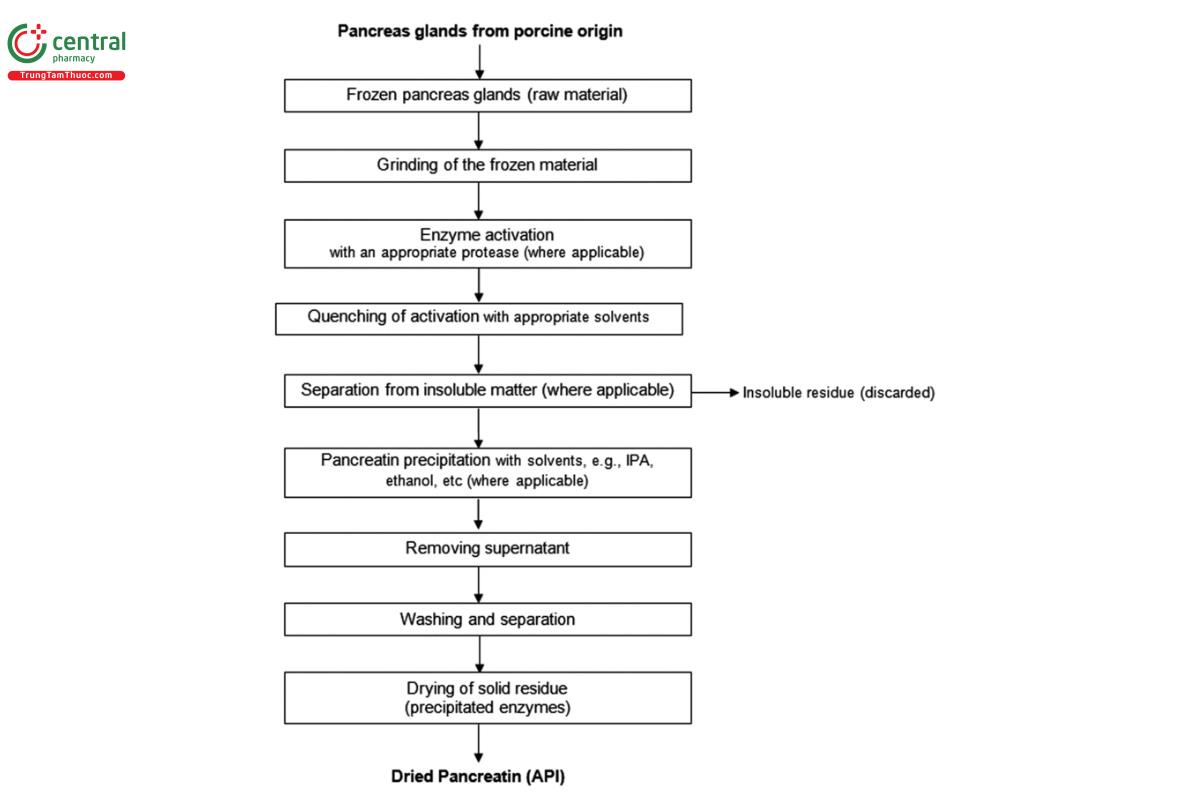
The pancreatic glands should be kept frozen to prevent a loss of enzymatic activity during holding and transport. After the manufacturer accepts the frozen materials, the pancreatin is extracted and purified under conditions that reduce microbiological load and other potential impurities. To accomplish this, the frozen pancreatic glands are macerated into fine slurry and treated with activators to convert the inactive pancreatic enzyme precursors (zymogens) into active enzymes. The activation is carried out under controlled conditions; factors such as time, temperature, ionic strength, or concentration are controlled as defined by each manufacturer's process. Once the activation is complete, the activation step is stopped by the addition of solvents such as acetone, isopropyl alcohol, or other enzyme-compatible solvents. After separation from insoluble matter (where applicable), the mixture is combined with solvent to precipitate the pancreatin. The supernatant is separated, and the solid residue is washed with solvent. Finally, the pancreatin is dried at appropriate temperature and vacuum. The biological activity may be adjusted and/or stabilized by adding suitable fillers, such as lactose; sucrose containing not more than 3.25% starch; pancreatin of lower digestive power; microcrystalline cellulose; maltodextrin; or sodium chloride.
3 QUALITY CONTROL
Each lot of pancreatin is subject to appropriate quality control testing. Quality control testing needs to address the requirements of the applicable monograph, as well as other identified quality parameters. Such tests should include appearance, identification, purity, and activity of the pancreatin. Process- and product-related impurities, such as residual solvents from extraction and precipitation and fat, should be considered.
Water is critical for enzyme activity and stability, and thus the limit of water content should be specified and monitored by appropriate analytical methods such as loss on drying (LOD) (see chapter (731) Loss on Drying). The activity assays are applied to confirm that pancreatin meets predefined limits for activity levels of lipase, amylase, and proteases, which are considered critical quality attributes.
Because pancreatin is of biological origin, quality control includes microbial testing as well as testing for the absence of certain adventitious agents that are pathogenic to humans.
4 LABELING
Product should be labeled in conformance to monograph and regulatory requirements but also should conform to customer requirements as applicable. The label contains information such as the name and address of the manufacturer, manufacturing date, retesting date, expiration date, lot number, storage conditions and specific precautions (e.g., "protect from moisture"), and a statement indicating the intended use.
5 CERTIFICATION AND DOCUMENTATION
The product must be accompanied by required certificates and documentation, as applicable. This documentation should include a certificate of analysis (COA) or other certificate documenting conformance to monograph requirements and quality control test results; a certificate of origin indicating the animal species; and certifications related to adventitious agents, including, where applicable, Transmissible Spongiform Encephalopathy/Bovine Spongiform Encephalopathy statements. Other pertinent and required information, such as retest or expiration date, storage, and packaging recommendations, should also be included as applicable.
The manufacturer/supplier should also be able to provide documentation for regulatory purposes containing the following information:
- Information on the regulatory agency's approval/license number and the full address of the pancreatin manufacturing site.
- A statement that the animal-sourced raw materials intended for pharmaceutical processing were collected and delivered under oversight of the responsible/competent authorities.
- A list of the countries of origin of the collected raw materials.
- A statement that during transportation, the pancreas glands can be identified for animal by-products intended for pharmaceutical purposes by appropriate documentation according to the corresponding legal regulations (such as animal health certificates from official veterinarians or a technical trade documentation).
- A statement that the pancreas glands are collected in approved slaughterhouses, and the animals from which pancreas glands are collected have undergone inspection in compliance with the current applicable legislation and were declared as suitable for human consumption, or no visible signs of diseases transmittable to humans or animals were detected at the time of slaughter.
6 TESTING AND CONTROL OF ADVENTITIOUS INFECTIOUS AGENTS
6.1 Overview
Although there have been no reports documenting any infectious illness subsequent to the use of pancreatin-derived medicinal products, there is a theoretical risk of porcine pathogen transmission from pancreatin.
The safety of porcine-derived PEPs should be enhanced by implementation of multiple complementary and/or overlapping strategies for adventitious agent containment, clearance, and control. Manufacturers should take an ongoing, risk-based approach to enhancing the safety of these products with regard to adventitious agents that includes incorporation of risk assessment, risk mitigation, and process materials management strategies. Testing should be included, when appropriate, to provide maximum possible assurance of a sufficiently low risk of harm that is significantly outweighed by the therapeutic benefits and concurrent assurance of availability of the drug to patients.
6.2 Risk Assessment Strategies
The FDA's "Guidance for Industry: Exocrine Pancreatic Insufficiency Drug Products" encourages a risk-based approach to the potential for viral contamination of pancreatic enzyme products, in agreement with ICH Q5A(R1) and chapter (1050) Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin. The adventitious-agent risk profile of biological products in general is contingent upon a variety of factors including the origin of the biological, the type of raw materials used, manufacturing processes, and the route of administration. As sourcing and manufacturing may vary between manufacturers, each manufacturer should establish and implement an individual, full adventitious-agent risk assessment.
Although the scope of ICH Q5A(R1) covers the viral safety evaluation of biotechnology products derived from cell lines of human and animal origin, the principles and risk assessment approaches can provide the basis for a risk evaluation strategy for the pancreatin products. Applying the principles of ICH Q5A(R1), the risk minimization strategy to protect patients against inadvertent adventitious agent exposure should reflect a combination of three components:
- Sourcing: Use of diligent sourcing to limit adventitious agents' access to the manufacturing process. Because pancreatin material is a by-product of the meat industry, it is important to ensure that pancreatin API is produced only from animals suitable for human consumption.
- Clearance: Incorporation of robust clearance steps into the manufacturing process. The efficacy of these strategies depends on the adventitious agents' resistance to the type of physical and chemical inactivation used.
- Testing: The control and testing of adventitious agents at suitable stages of the manufacturing process to provide assurance that any remaining load of potentially harmful adventitious agent is at sufficiently low levels. This is accomplished by using suitable screening assays against a relevant test virus panel. In this part of the risk assessment, the potential of porcine viruses to pose a risk to humans should be taken into account.
To identify the potential adventitious agents that might be present in a pancreatin preparation, manufacturers should identify adventitious agents that are present in the pig and assess the probability of their presence in the starting materials and API. Chapters (61) Microbiological Examination of Nonsterile Products: Microbial Enumeration Tests and (62) Microbiological Examination of Nonsterile Products: Tests for Specified Microorganisms address testing for specific microbiological contamination, as defined in the relevant pancreatin-related product monographs. Additionally, manufacturers should specifically evaluate the potential presence of porcine viruses. In this evaluation, the manufacturing steps capable of removing or inactivating viruses should be identified, and the efficiency of the process for removing and/or inactivating viruses should be demonstrated by viral validation studies that follow applicable guidelines. Once a list of critical viruses potentially present in the starting material and/or in the API is established, manufacturers will decide on the appropriate process stages for viral testing and the level of viral testing to be performed on a batch-to-batch basis. Tests for specific viruses should be developed and validated, and acceptance criteria should be established and used in making accept/reject decisions about pancreatin API batches. The potential zoonotic or non-zoonotic character of the virus should be taken into account when setting acceptance criteria.
6.3 Identification of Relevant Virus Panel Test
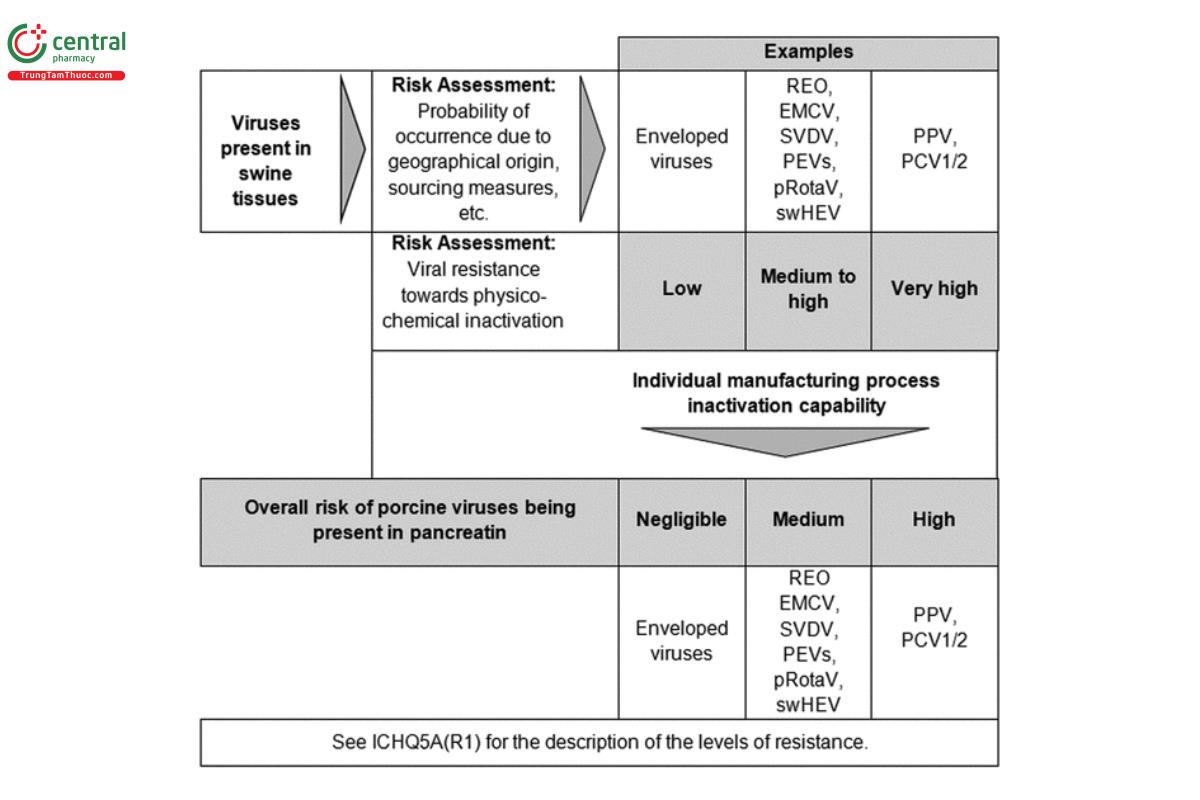
The risk assessment strategy described above requires the identification of potential viral contaminants of porcine-derived starting materials. Table 1 gives an overview of enveloped and non-enveloped viruses known to be present in pigs that may present a contamination risk when using pigs deemed fit for human consumption as source animals. The risk assessment should address at least the viruses listed; however, depending on the origin of animals and the manufacturing process capability, the list may be adapted, and additional viruses may be considered (see the example in Figure 2). Manufacturers should implement systems to identify emergence of potentially relevant new viruses.
Table 1. Hazard Identification: Viruses Known to be Present in Pigs
| Enveloped Viruses | Non-Enveloped Viruses |
| Classical swine fever virus | Encephalomyocarditis virus (EMCV) |
| African swine fever virus (ASFV) | Swine vesicular disease virus (SVDV) |
| Inuenza virus | Foot and mouth disease virus (FMDV) |
| West Nile virus (WNV) | Reoviruses (REO) |
| Vesicular stomatitis virus (VSV) | Swine hepatitis E virus (swHEV) |
| Eastern equine encephalitis virus (EEEV) | Porcine rotaviruses (pRotaV) |
| Rabies virus (RABV) | Porcine enteroviruses (PEVs) |
| Porcine reproductive and respiratory syndrome virus (PRRSV) | Porcine enteroviruses (PEVs) |
| Transmissible gastroenteritis virus (TGEV) | Porcine circovirus types 1 and 2 (PCV1/2) |
| Pseudorabies virus (SuHV-1) | |
| Nipah virus | |
| Porcine endogenous retroviruses (PERVS) |
6.4 Virus Clearance by Manufacturing Process Steps
Demonstration of viral clearance is a critical component of ensuring the overall safety of pancreatin-derived products. The objective of virus-clearance evaluation studies is not only to evaluate the ability of the manufacturing process to clear known viral contaminants and to estimate quantitatively the overall level of virus reduction obtained by the manufacturing process but also to estimate the capability of the manufacturing process steps to clear viruses in general. Viral clearance studies for the manufacturing process of pancreatin should be performed in accordance with the applicable, current guideline, ICH Q5A(R1).
Current pancreatin production processes are considered to be effective for inactivating enveloped viruses, following the results of virus clearance studies. However, non-enveloped viruses are more resistant to physico-chemical inactivation, making their inactivation more variable. Examples of viruses that are classified as having a high viral resistance toward treatment and that show moderate to limited inactivation include PPV and PCV1/2 (see Figure 2).
6.5 Viral Testing
A testing strategy is needed when the ability of the process to remove or inactivate a specific virus to appropriate levels has not been demonstrated. The potential zoonotic or non-zoonotic character of the virus should be taken into account when setting acceptance criteria, in terms of both assay sensitivity and specification limit setting. API batches that test positive for zoonotic viruses should be rejected. As a part of quality control, viral testing should be performed on each lot of API. The tests used should allow exclusion of any detectable load of potentially harmful levels of adventitious agent. The 9 CFR describes testing requirements for porcine biological products including live virus vaccines and antibody products but does not specifically address pancreatin. Applying the principles of 9 CFR, assays to detect porcine viruses can, for example, be based on cell culture monitoring for cytopathogenic effects for an appropriate incubation time, hemadsorption testing, virus-specific staining techniques, or appropriate combinations thereof (also see chapter (1237) Virology Test Methods). In addition to the technologies and viruses covered by 9 CFR, new molecular biology-based technologies may be used, and other viruses with zoonotic potential that are identified may require testing. Examples of specific viruses not covered by the current virus testing guide may include swHEV.
The definition of the test virus panel, selection of the appropriate process stage, suitability of the test method, and test method sensitivity should be justified by the manufacturer. All tests for specific viruses should be developed and validated in compliance with current guidance, for example, chapters (1225) Validation of Compendial Procedures and (1033) Biological Assay Validation, as applicable, and acceptance criteria should be established and used for accept/reject decisions of pancreatin API batches.
7 CHARACTERIZATION OF PANCREATIN
7.1 Description and Physico-Chemical Properties
Pancreatin is a slightly brownish to tan amorphous powder with a raw meat odor and taste. Pancreatin is partly soluble in water, forming a weak turbid solution, and insoluble in alcohol and ether. The following conditions may degrade pancreatin: mineral acids, alkali hydroxides, oxidizing agents, many metallic salts, and high humidity and temperatures. Pancreatin-containing solutions should be filtered with caution, due to potential retention of the lipase and proteases on the filter. The enzymatic activity reaches a maximum in neutral-to-weakly alkaline solutions. The activity decreases quickly in acidic or strong alkaline solutions. The same applies to boiling of pancreatin-containing aqueous solutions. In non-enteric-coated formulations, exposing the product at a pH of 4.5 or less is not recommended, because a nearly complete loss of lipolytic activity has been observed.
Because pancreatin is of biological origin, other components are present in addition to enzymatically active proteins. These other components include proteins, amino acids, peptides, nucleic acids and fragments thereof, tissue components, fat, and inorganic substances. These components may have an impact on the quality of the final material.
7.2 Protein and Enzyme Contents
Pancreatin contains different digestive enzymes, most of which are produced and stored as zymogens (inactive precursors) in the pancreatic acinar cells. Under physiological conditions, pancreatic zymogens are transformed into active enzymes once the pancreatic secretion reaches the upper small intestine. An intestinal protease, enterokinase, triggers the activation process by cleaving the zymogen of trypsin, which further activates the other proteases. During the production process, the enzymes present in pancreatin are activated by trypsin (see Figure 1).
Table 2 summarizes some of the important characteristics of the main pancreatic enzymes found in pancreatin.
Table 2. Characteristics of Known Enzymes Present in Porcine Pancreatin
| Name | E.C. Number | UniProtKB/ Swiss-Prot Accession Number | Substrates | Produced as a Precursor | Molecular Mass (kDa) | Sequence Length | Isoelectric Point |
| Trypsin | 3.4.21.4 | P00761.1 | Proteins | Yes | 23.8 (zymogen), 23.5 (activated) | 231 a.a. (zymogen), 223 a.a. (activated) | 9.3 (zymogen), 10.5 (activated) |
| Chymotrypsin | 3.4.21.1 | G1ARD6_PIG | Proteins | Yes | 29.1 (zymogen), 25.6 (activated) | 268 a.a. (zymogen), 221 a.a. (activated) | 8.7 |
| Elastase | 3.4.21.36 | P00772.1 | Proteins | Yes | 25.9 (activated) | 250 a.a. (zymogen), 240 a.a. (activated) | 8.5 |
| Carboxypeptidase A1 | 3.4.17.1 | P09954 | Proteins | Yes | 34.7 (activated) | 308 a.a. (activated) | Not known |
| Carboxypeptidase B | 3.4.17.2 | P09955.5 | Proteins | Yes | 34.7 (activated) | 305 a.a. (activated) | 6.0 |
| Kallikrein, glandular | 3.4.21.35 | P00752.4 | Proteins | Yes | 25–28 kDa | 246 a.a. (zymogen), 239 a.a. (activated) | 4.2–4.3 |
| Triacylglycerol lipase | 3.1.1.3 | P00591.2 | Triglycerides, diglycerides | No | 50.1 (two glycosylation isoforms, lipases A and B) | 450 a.a. (mature) | 4.9 (lipase A), 5.0 (lipase B) |
| Colipase | P02703.3 | No enzyme activity, cofactor of pancreatic lipase | Yes | 10.3 (porcine procolipase A), 10.1 (porcine procolipase B) | 93 a.a. (procolipase A), 95 a.a. (procolipase B) | Not known | |
| Phospholipase A2 | 3.1.1.4 | P00592 | Phospholipids | Yes | 14.7 (zymogen), 14 (activated) | 146 a.a. (zymogen), 123 a.a. (activated) | 4.4–4.5 |
| Cholesterol esterase, also named carboxyl ester lipase (CEL), carboxyl ester hydrolase (CEH) or bile-salt stimulated lipase (BSSL) | 3.1.1.13, 3.1.1.1 | Complete Amino acid sequence still unknown in pig | Cholesterol esters, vitamin esters, monoglycerides, phospholipids, galactolipids some activity on triglycerides | No | Inconsistent values in the literature ranging from 65 to 98 kDa. While proteolytic forms have been identified, the exact mass is still unknown. | Complete amino acid composition still unknown in pig | 4.2–4.8 |
| α-Amylase | 3.2.1.1 | P00690.3 | Polysaccharides | No | 55.3 | 496 a.a. (mature) | 5.95 (amylase I), 5.45 (amylase II) |
7.2.1 PANCREATIC PROTEASES
Proteases are enzymes that digest proteins into smaller peptide fragments and amino acids by hydrolyzing the peptide bonds. Pancreatin contains five major proteases: trypsin, elastase, chymotrypsin, carboxypeptidase A1, and carboxypeptidase B. Trypsin, chymotrypsin, and elastase are classified as both serine proteases and endopeptidases because they cleave peptide bonds at the C-terminal side of an amino acid and also have a catalytically important serine residue in their active sites. Carboxypeptidases catalyze hydrolysis of the amino acids from the C-terminal end position in polypeptides and thus are classified as exopeptidases. Carboxypeptidases sequentially release residues from the C-terminus of proteins and peptides with a well-defined specificity.
Trypsin acts specifically on the C-terminal side of the positively charged amino acid residues Lysine and arginine. Trypsinogen is secreted by the pancreas as an inactive precursor and discharged into the duodenum where enterokinase converts trypsinogen to active trypsin. Enterokinase is secreted in the duodenum by cells of the duodenal mucosa. Enterokinase removes a terminal octapeptide from trypsinogen and yields a polypeptide chain of active trypsin cross-linked by six disulfide bridges. Trypsin contains one high-affinity calcium binding site that is required for enzyme stability.
Chymotrypsin acts preferentially on the C-terminal side of tyrosine, phenylalanine, and tryptophan residues. It is secreted as an inactive zymogen, chymotrypsinogen, which undergoes proteolytic processing by trypsin to form the active enzyme. Chymotrypsin binds one calcium ion per molecule.
Elastase acts on small, neutral amino acid residues, such as glycine and alanine, but also hydrolyses amides and esters and is distinctive in that it acts upon Elastin. Elastase is produced as a zymogen and the activated form contains four disulfide bridges. Elastase binds one calcium ion per molecule.
Carboxypeptidase A1 is an exopeptidase hydrolyzing the peptide bond adjacent to the C-terminal end of a polypeptide chain, thus releasing the C-terminal amino acid. It cleaves aromatic and bulky aliphatic amino acid residues and shows little or no action with aspartic acid, glutamic acid, Arginine, lysine, and proline amino acid residues. It contains one zinc ion per molecule. The zinc ion is essential for activity, if removed during dialysis it must be replaced. Thus, carboxypeptidase A1 is also classified as a metalloprotease.
Carboxypeptidase B catalyzes the hydrolysis of the basic amino acids lysine, arginine, and ornithine from the C-terminal end of a polypeptide chain. It may also have a function in the further degradation of products of tryptic digestion. The enzyme binds one zinc ion per molecule, which is a necessary functional part of the enzyme for activity, and thus carboxypeptidase B is also classified as a metalloprotease.
7.2.2 PANCREATIC LIPASE AND COLIPASE
Pancreatic lipase (PL, also known as triacylglycerol acyl hydrolase) is a glycoprotein and produced directly as an active enzyme by the pancreas. Two glycosylation isoforms of PL, lipase A and lipase B, are present in pancreatin. These two isoforms have identical amino acid compositions but differ slightly in their glycosylation patterns, with lipase A as more acidic than lipase B. PL is a water-soluble enzyme that acts on insoluble lipid substrates, triglycerides, at the lipid-water interface. Its activity is dependent on the substrate-specific surface accessible to the enzyme, and it increases with the state of emulsification of the lipids. PL preferentially hydrolyzes ester bonds at the C-1 and C-3 positions of triglycerides and exhibits a broad spectrum of fatty acid chain length specificity. Therefore, PL is active against a wide variety of the triglycerides that are typically present in the diet. It also acts on diglycerides, but its activity on monoglycerides is very weak. It mainly converts triglycerides into monoglycerides and free fatty acids; the more polar lipolysis products are absorbed in the small intestine. In the presence of various amphiphiles such as bile salts at micellar concentrations, the PL adsorption at the lipid-water interface can be hindered, thus decreasing lipolytic activity.
A small protein cofactor also produced by the pancreas, colipase, helps PL to anchor to interfaces in the presence of competitive amphiphiles, and thus restores PL activity. Colipase is also produced by the pancreas as a precursor, procolipase. Procolipase is activated by trypsin.
7.2.3 PANCREATIC PHOSPHOLIPASE A2
Phospholipase A2 (also known as type IB secretory PLA2) is a heat-stable, water-soluble enzyme that catalyzes the calcium-dependent hydrolysis of the 2-acyl groups in 3-sn-phosphoglycerides. It is produced as a precursor that is activated by trypsin.
7.2.4 PANCREATIC AMYLASE
Porcine pancreatic a-amylase (1,4-α-D-glucan glucanohydrolase) catalyzes the hydrolysis of internal 1,4-α-D glucosidic linkages in polysaccharides containing three or more 1,4-α-linked D-glucose units to yield a mixture of dextrins, maltose, and glucose. Amylase exists in two forms (I and II) that have similar enzymatic properties but that differ in their isoelectric points. Both molecular forms of amylase are glycoproteins that contain fucose, galactose, mannose, and different contents of glucosamine. Both amylases consist of a single polypeptide chain with four disulfide bridges and contain a tightly bound calcium ion.
8 ENZYME ACTIVITY MEASUREMENTS
Although pancreatin contains a variety of enzymes, it is usually characterized by measuring the activities of the three main enzyme classes, lipase, protease, and amylase.
8.1 Lipase Activity
The lipase activity of pancreatin on triglycerides with long-chain fatty acids is mainly due to PL, which requires the presence of its specific cofactor, colipase, to act on triglyceride emulsions in the presence of bile salts that are competitors for lipase adsorption at the surface of lipid droplets. Colipase and lipase form an active complex with a 1-to-1 stoichiometry. Commercial preparations of pancreatin usually contain colipase; however, it is recommended to check that the API manufacturing process provides enough colipase for the lipase activity. This control should be part of a proper characterization to be performed during process validation of the API.
Besides PL, pancreatin also contains pancreatic carboxyl ester lipase (CEL), which hydrolyzes several lipid substrates. CEL activity on triglycerides is usually considered very low, compared with that of PL, and its contribution to pancreatin lipase activity is negligible, as measured by the USP lipase assay.
The lipase assay described in the USP Pancreatin monograph is based on the enzyme's rate of digestion of olive oil emulsified with acacia (also known as gum arabic). The activity of the test sample is calculated by comparing it to a standard preparation of enzyme with known activity. Olive oil contains triglycerides with long-chain fatty acids that are representative of dietary triglycerides. Lipase activity is measured on the basis of the titration of the free fatty acids released from olive oil upon lipolysis by the lipase present in pancreatin. This titration by sodium hydroxide is performed at a constant pH of 9.0 by pH- or potentiostatic titration, at which long-chain fatty acids are totally ionized. It is worth noting that this pH value does not correspond to physiological conditions (the mean pH of the small intestine contents is close to 6.0 during a meal), but it allows measurement of optimum lipase activity in vitro. Because the USP lipase assay solution contains USP Bile Salts Reference Standard (RS), the detection of enzyme activity requires that both lipase and colipase are present in pancreatin. One USP Unit of lipase is defined as the amount of enzyme that, under the defined conditions with the defined substrate, liberates 1 µmole of fatty acid per minute.
Other lipase assays are available for monitoring lipase activity in pancreatin. When using lipase assays with non-natural substrates, such as tributyrin, it is recommended to confirm that the pancreatin sample tested is also active on long-chain triglycerides, particularly using the USP lipase assay with olive oil-acacia emulsion.
8.2 Protease Activity
The proteolytic activity of pancreatin on polypeptides and proteins is due to the inherent enzymes trypsin, chymotrypsin, elastase, carboxypeptidase A1, and carboxypeptidase B. The two major pancreatic proteases are trypsin and chymotrypsin, and along with elastase, these endopeptidases generate small polypeptides from larger proteins. The further action of the exopeptidases carboxypeptidase A1 and B leads to single amino acids during digestion.
The USP spectrophotometric assay for protease activity from pancreatin is based on the enzyme's rate of digestion of casein under test conditions. The activity of the test sample is calculated by comparing it to a standard preparation of enzyme with known activity. Casein is composed of α (s1) and α (s2)-caseins, β-casein, and casein, which are phosphorylated on serine residues and lack disulfide bridges. The conformation of casein is similar to that of denatured globular proteins with little or no tertiary structure. It is recommended that users evaluate suppliers of the casein substrate for consistency of dispersion.
The hydrolysis of casein by pancreatic proteases generates single amino acids and small peptides, and their release can be quantified by measuring their absorption at 280 nm. Before this measurement, non-hydrolyzed proteins and large peptides have to be separated by a selective precipitation with trichloroacetic acid, followed by filtration. The filtrate is then used for the spectrophotometric assay of protease activity, using tyrosine as a calibrant. One USP Unit of protease activity is contained in the amount of pancreatin that hydrolyzes casein at an initial rate such that the amount of peptides liberated per minute and not precipitated by trichloroacetic acid gives the same absorbance at 280 nm as 15 nmol of tyrosine.
Other assays are available for monitoring the activity of individual and total proteases in pancreatin, and the unit assignments are specific for each substrate. N-Acetyl-L-tyrosine ethyl ester is commonly used for both titrimetric and spectrophotometric (λ = 237 nm) assays of chymotrypsin activity. Similarly, N-benzoyl-L-arginine ethyl ester is commonly used for both titrimetric and spectrophotometric (λ = 253 nm) assays of trypsin activity. In both cases, the titrimetric assay is based on ester hydrolysis by proteases and the release of acid groups, whereas the spectrophotometric assay is based on the chromogenic properties of these acids. Toluene-sulfonyl-L-arginine methyl ester is another chromogenic substrate (λ = 247 nm) used for measuring trypsin activity. These substrates are, however, not proteins, and the assays involve the cleavage of a carboxylic ester bond instead of a peptide bond.
Other assays using chromogenic peptides as substrates have been developed for improving specificity and sensitivity. More specific and sensitive protease assays have been developed using short synthetic peptide substrates (3-5 amino acid residues) with a chromogenic group (4-nitroaniline) coupled to the C-terminal end by an amide bond. The chromogenic group is specifically removed by proteases and is measured photometrically. The change in the absorbance at 405 nm is directly proportional to the protease activity. Specific substrates are commercially available for trypsin (carbobenzoxy-valyl-glycyl-arginine-4-nitril-anilide acetate) and chymotrypsin (methyl-O-succinoyl-arginyl-prolyl-tyrosine-4-nitril-anilide chlorhydrate). Fluorescein isothiocyanate (FITC)-labeled casein is also used as a general protease substrate. The assay is based on the quenching of the fluorescein label bound to casein. When FITC-casein is digested into smaller peptides by proteases, fluorescence at 530 nm (excitation at 485 nm) is increased and can be measured to determine protease activity.
8.3 Amylase Activity
The glycolytic activity of pancreatin is due to α-type amylases. Such enzymes catalyze the hydrolysis on internal α-1,4-glucan links in polysaccharides containing three or more α-1,4-linked D-glucose units, yielding a mixture of maltose and glucose. The two major isoforms (I and II) of porcine amylases have identical enzymatic properties. In the last decades, several methods have been developed for assaying amylase activity; many of these are based on the detection of starch hydrolysis, as this polymer is the natural amylase substrate. Starch consists of two types of molecules, amylose (usually 20%-30%) and amylopectin (usually 70%-80%). Both polymers result from the assembly of Glucose units connected via α-1,4-glucan links; in addition, in amylopectin, about 1 residue in every 20 or so, is also linked α-(1→6), forming branch-points.
The USP Amylase assay in the Pancreatin monograph is based on the enzyme's rate of digestion of starch, and the activity of the test sample is calculated by comparing it to a standard preparation of enzyme with known activity. Starch is hydrolyzed by amylase; the reducing groups resulting from the hydrolysis react with iodine in alkaline solution; and the excess iodine is titrated with thiosulphate. One USP Unit of amylase activity is defined as the amount of pancreatin that decomposes starch at an initial rate such that 0.16 µEq of glycosidic linkage is hydrolyzed per minute under the conditions of the assay.
9 APPENDIX 1: REGULATORY BIBLIOGRAPHY
Code of Federal Regulations, title 9 part 113 section 46. Detection of cytopathogenic and/or hemadsorbing agents. US Government Printing Office; 2006. p. 640. Available from: http://www.gpo.gov/fdsys/pkg/CFR-2006-title9-vol1/pdf/CFR-2006-title9-vol1-chapl-subchapE.pdf Code of Federal Regulations, title 9 part 113 section 47. Detection of extraneous viruses by the fluorescent antibody technique. US Government Printing Office; 2006. p. 640-641. Available from: http://www.gpo.gov/fdsys/pkg/CFR-2006-title9-vol1/pdf/CFR-2006-title9-vol1-chapl-subchapE.pdf
Code of Federal Regulations, title 9 part 113 section 53. Requirements for ingredients of animal origin used for production of biologics. US Government Printing Office; 2006. p. 644-645. Available from: http://www.gpo.gov/fdsys/pkg/CFR-2006-title9-vol1/pdf/CFR-2006-title9-vol1-chapl-subchapE.pdf
EMEA. CHMP/BWP/398498/2005. Guideline on virus safety evaluation of biotechnological investigational medicinal products. 24 Jul 2008. p. 1-9. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003795.pdf
EMEA. CHMP/EWP/9147/2008-corr*. Guideline on the clinical development of medicinal products for the treatment of cystic fibrosis. 22 Oct 2009. p. 1-27. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/12/WC500017055.pdf
FDA. Guidance for industry. Exocrine pancreatic insufficiency drug products-submitting NDAs, April 2006. Available from: http://www.fda.gov/downloads/Drugs/GuidanceCompliance RegulatoryInformation/Guidances/ucm071651.pdf
FDA. Guidance for industry. ICH Q7A: good manufacturing practice guidance for active pharmaceutical ingredients. Aug 2001. p. 1-52. Available from: http://www.fda.gov/downloads/Drugs/.../Guidances/ucm073497.pdf
FDA. Guidance for industry. ICHQ10 pharmaceutical quality system. Apr 2009. p. 1-19. Available from: http://www.fda.gov/downloads/Drugs/Guidances/ucm073517.pdf
ICH. ICH harmonised tripartite guideline. Viral safety evaluation of biotechnology products derived from cell lines of human or animal origin Q5A (R1). 23 Sep 1999. p. 1-27. Available from: http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q5A_R1/Step4/Q5A_R1_Guideline.pdf Uniprot Consortium. Available from: www.uniprot.org
10 APPENDIX 2: LITERATURE BIBLIOGRAPHY
Auld DS. Carboxypeptidase A. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 812-821.
Auld DS. Carboxypeptidase A2. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 821-851.
Avilés FX, Vendrell J. Carboxypeptidase B. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 831-833.
Bieth JG. Pancreatic elastase. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 1504-1508.
Bieth JG. Pancreatic elastase II. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 1508-1509.
Bieth JG. Pancreatic endopeptidase E. In: Barrett AJ, Rawlings ND, Woessner JF, et al., editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 1510-1511.
Borgström B. On the interactions between pancreatic lipase and colipase and the substrate, and the importance of bile salts. J Lipid Res. 1975;16(6):411-417.
Gráf L, et al. Chymotrypsin. In: Barrett AJ, Rawlings ND, Woessner JF, editors. Handbook of proteolytic enzymes. 2nd ed. Amsterdam: Elsevier; 2004. p. 1495-1501.
Lowe ME. The triglyceride lipases of the pancreas. J Lipid Res. 2002;43(2):2007-2016.
Desnuelle P, Sjöström H, Noren O, editors. In: Molecular and cellular basis of digestion. Amsterdam: Elsevier; 1986. p. 539.
Peschke GJ. Active components and galenic aspects of enzyme preparations. In: Lankisch PG, editor. Pancreatic enzymes in health and disease. 1st ed. Berlin: Springer-Verlag:1991. p. 55-64.
Verger R. Pancreatic lipase. In: Lipases. Borgström B, Borockman HL, editors. Amsterdam: Elsevier; 1984. p. 84-150. [review]
Verger R, de Haas GH, Sarda L, Desnuelle P. Purification from porcine pancreas of two molecular species with lipase activity. Biochim Biophys Acta. 1969;188(2):272-282.


